Advanced Catalytic Strategy for Scalable Production of 2-Trifluoromethyl Quinazolinone Intermediates
Advanced Catalytic Strategy for Scalable Production of 2-Trifluoromethyl Quinazolinone Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly those exhibiting potent biological activities. Quinazolinone scaffolds are ubiquitous in medicinal chemistry, serving as the core structure for a wide array of therapeutic agents ranging from anticancer drugs to antimalarials. A recent technological breakthrough detailed in patent CN111675662B introduces a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This innovation addresses critical bottlenecks in traditional synthesis by utilizing a cost-effective iron-catalyzed system that operates under relatively mild conditions. The introduction of the trifluoromethyl group is strategically significant, as it markedly enhances the electronegativity, metabolic stability, and lipophilicity of the target molecules, thereby improving their bioavailability and pharmacokinetic profiles. For R&D directors and procurement managers alike, this development represents a pivotal shift towards more sustainable and economically viable manufacturing processes for high-value pharmaceutical intermediates.
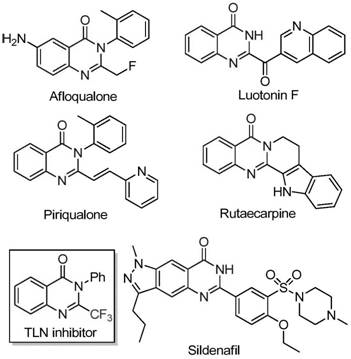
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. While these methods are chemically feasible, they suffer from severe practical limitations that hinder their adoption in large-scale commercial manufacturing. The reaction conditions are often excessively harsh, requiring stringent temperature controls or hazardous reagents that pose safety risks in an industrial setting. Furthermore, the substrate scope is frequently narrow, limiting the structural diversity accessible to medicinal chemists during lead optimization phases. Perhaps most critically for supply chain stakeholders, these conventional routes often exhibit low yields and require expensive starting materials, leading to inflated production costs and inconsistent supply continuity. The reliance on specialized fluorinating agents also complicates waste management and environmental compliance, adding hidden costs to the overall process.
The Novel Approach
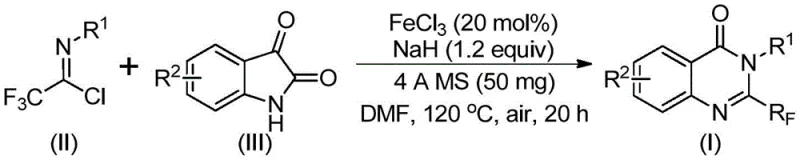
In stark contrast to legacy methodologies, the novel approach disclosed in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as primary building blocks. This strategy leverages a cheap metal iron catalyst, specifically ferric chloride, to drive the cyclization efficiently. The operational simplicity is a major advantage; the reaction proceeds in common organic solvents like DMF and tolerates a wide range of functional groups, allowing for the synthesis of diverse analogues without extensive protection-deprotection sequences. By shifting from precious metal catalysts or aggressive fluorinating agents to an abundant iron-based system, the process drastically simplifies the downstream purification requirements. This not only accelerates the timeline from laboratory bench to pilot plant but also aligns with green chemistry principles by reducing the environmental footprint associated with heavy metal waste. The ability to scale this reaction to the gram level with high conversion rates demonstrates its immediate potential for industrial application.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this synthetic innovation lies in the elegant mechanistic pathway facilitated by the iron catalyst and base promotion. The reaction initiates with an alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This initial coupling generates a trifluoroacetamidine intermediate, which serves as the precursor for the subsequent ring closure. The presence of ferric chloride is crucial, as it catalyzes a decarbonylation and cyclization sequence that isomerizes the intermediate into the final stable quinazolinone structure. This dual-function mechanism ensures high atom economy and minimizes the formation of unwanted byproducts. The use of 4A molecular sieves in the reaction mixture further enhances efficiency by sequestering moisture, which could otherwise deactivate the sodium hydride base or hydrolyze sensitive intermediates. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratio of catalyst to substrate, to maximize throughput.
From an impurity control perspective, the robustness of this catalytic cycle is evident in the clean reaction profiles observed across various substrates. The tolerance for electron-withdrawing and electron-donating groups on the aromatic rings suggests that the electronic nature of the substrate does not significantly impede the catalytic turnover. This is particularly beneficial for manufacturing consistency, as it reduces the risk of batch-to-batch variability caused by minor fluctuations in raw material quality. The final products, such as the specific derivatives shown in the patent examples, are obtained with high purity after standard column chromatography, indicating that the reaction selectivity is inherently high. For quality assurance teams, this implies a simpler analytical workflow and reduced need for complex recrystallization steps to meet stringent pharmaceutical specifications. The mechanistic clarity provided by this patent empowers manufacturers to implement rigorous process controls that ensure the delivery of high-purity intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a production environment requires adherence to specific operational parameters to ensure optimal yield and safety. The process begins with the precise weighing and mixing of ferric chloride, sodium hydride, and molecular sieves in an anhydrous solvent. The addition of the trifluoroethylimidoyl chloride and isatin must be controlled to manage the exothermic nature of the initial base-promoted step. Following the initial stirring period at moderate temperatures, the reaction vessel is heated to facilitate the cyclization. Detailed standardized operating procedures regarding temperature ramping, stirring speeds, and quenching protocols are essential for reproducibility. The following guide outlines the critical stages of this synthesis based on the patented methodology.
- Combine ferric chloride catalyst, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in an organic solvent such as DMF.
- Stir the reaction mixture at 40°C for 8-10 hours to initiate the alkali-promoted bond formation.
- Heat the mixture to 120°C and react for 18-20 hours under air to complete the decarbonylation and cyclization, followed by filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology offers substantial strategic benefits beyond mere chemical efficiency. The primary driver for adoption is the significant reduction in manufacturing costs achieved through the substitution of expensive reagents with commodity chemicals. By eliminating the dependency on precious metal catalysts, companies can insulate their production budgets from the volatility of the precious metals market. Furthermore, the use of widely available starting materials like isatin ensures a stable and reliable supply chain, mitigating the risk of production stoppages due to raw material shortages. The simplified post-treatment process, which involves basic filtration and chromatography, reduces the consumption of solvents and consumables, contributing to lower operational expenditures. These factors collectively enhance the overall cost competitiveness of the final pharmaceutical product.
- Cost Reduction in Manufacturing: The replacement of costly catalysts and specialized fluorinating agents with inexpensive ferric chloride and common chlorides results in a drastic decrease in direct material costs. Additionally, the high reaction efficiency minimizes the loss of valuable intermediates, maximizing the output per unit of input. The elimination of complex heavy metal scavenging steps further reduces the cost associated with waste disposal and purification media. This comprehensive cost optimization allows for more competitive pricing strategies in the global marketplace while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as both isatin derivatives and trifluoroethylimidoyl chlorides are commercially available from multiple vendors. This multi-sourcing capability prevents single-point failures in the supply network and provides leverage in negotiations with suppliers. The robustness of the reaction conditions means that production can be maintained even if there are minor variations in the quality of incoming materials, ensuring consistent delivery schedules to downstream customers. This reliability is crucial for maintaining the continuity of drug development pipelines and commercial manufacturing runs.
- Scalability and Environmental Compliance: The protocol is designed with scalability in mind, having been successfully demonstrated at the gram level with clear pathways for expansion to kilogram and ton scales. The use of iron, a non-toxic and environmentally benign metal, aligns with increasingly strict global environmental regulations regarding heavy metal discharge. Simplified waste streams reduce the burden on effluent treatment plants and lower the regulatory compliance costs associated with hazardous waste management. This sustainability profile enhances the corporate social responsibility standing of the manufacturer and facilitates easier regulatory approvals in key markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They serve to clarify the practical implications of adopting this method for your specific manufacturing needs. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: What are the advantages of using FeCl3 over precious metal catalysts?
A: Using ferric chloride significantly reduces raw material costs compared to palladium or gold catalysts, eliminates the need for expensive heavy metal removal steps, and simplifies waste treatment protocols.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the patent explicitly states the method is expandable to the gram level and provides possibilities for industrial scale application due to simple operation and cheap raw materials.
Q: What is the substrate scope for this reaction?
A: The method demonstrates good functional group tolerance, accommodating various substituents on the aryl ring including alkyl, halogen, methoxy, and nitro groups at ortho-, meta-, or para-positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our facilities are equipped with state-of-the-art reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering consistent quality and supply continuity, leveraging our technical expertise to optimize every step of the manufacturing process for maximum efficiency and cost-effectiveness.
We invite you to collaborate with us to explore the full capabilities of this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target compounds. By partnering with us, you gain access to a reliable supply chain partner dedicated to driving innovation and value in your drug development programs.
