Revolutionizing Pharmaceutical Intermediate Production Through Scalable Synthesis of High-Purity Trifluoromethyl Imidazole Compounds
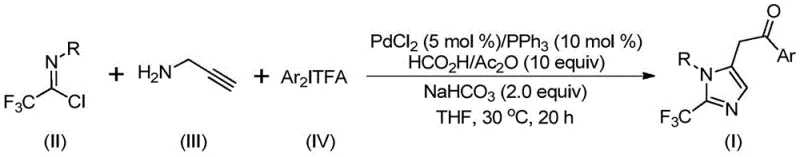
Patent CN111423381B presents a transformative methodology for synthesizing structurally diverse 2-trifluoromethyl substituted imidazole compounds that serve as critical building blocks in modern pharmaceutical chemistry due to their prevalence in bioactive molecular frameworks. This innovative process employs a palladium-catalyzed carbonylation reaction operating under exceptionally mild conditions at precisely thirty degrees Celsius with reaction durations optimized between eighteen to twenty hours. The strategic selection of commercially accessible starting materials including trifluoroethylimidoyl chloride and propargylamine enables scalable production from laboratory-scale validation through industrial implementation while maintaining stringent purity requirements essential for drug development pipelines. By eliminating transition metal catalysts requiring complex removal procedures found in conventional approaches, this patent delivers a sustainable manufacturing pathway that addresses fundamental industry challenges in cost-effective intermediate production without compromising quality standards required by global regulatory authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethyl imidazole compounds typically require harsh reaction conditions including elevated temperatures exceeding one hundred degrees Celsius or cryogenic environments below minus twenty degrees Celsius that significantly increase energy consumption and operational complexity. These methods often employ expensive transition metal catalysts requiring extensive purification steps to remove trace metal residues that could compromise final product quality for pharmaceutical applications. Furthermore, conventional approaches exhibit narrow substrate scope with poor functional group tolerance that limits structural diversity and necessitates multiple specialized synthetic pathways for different derivative compounds. The inherent inefficiencies manifest as low yields typically below sixty percent with extended reaction times exceeding forty-eight hours that create substantial bottlenecks in manufacturing throughput while increasing production costs through excessive solvent usage and waste generation.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation process operating under ambient temperature conditions that dramatically simplifies thermal management requirements while enhancing operational safety profiles across manufacturing facilities. By utilizing readily available diaryl iodonium salts as coupling partners instead of hazardous reagents common in traditional routes, this approach eliminates multiple purification steps previously required to remove toxic byproducts. The strategic design incorporates formic acid/acetic anhydride as a carbon monoxide surrogate that generates controlled CO release during reaction progression without requiring specialized pressurized equipment typically needed for gaseous carbon monoxide handling. This innovation enables seamless scalability from gram-scale laboratory validation directly to multi-kilogram production runs while maintaining consistent product quality through simplified process control parameters that reduce operator dependency and minimize batch-to-batch variability.
Mechanistic Insights into Pd-Catalyzed Carbonylation for Trifluoromethyl Imidazole Synthesis
The reaction mechanism proceeds through a sophisticated sequence where initial intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate generates a trifluoroacetamidine intermediate that undergoes spontaneous isomerization prior to palladation of the alkyne amine component. This critical step forms an alkenyl palladium species that subsequently isomerizes to an alkyl palladium intermediate capable of undergoing carbonylation using carbon monoxide released from the formic acid/acetic anhydride system under mild thermal conditions. The resulting acyl palladium complex then engages with diaryl iodonium salts through oxidative addition to form tetravalent palladium species that ultimately undergo reductive elimination to yield the target imidazole products with precise regioselectivity at the two-position. This cascade process demonstrates exceptional functional group tolerance due to the mild reaction environment that preserves sensitive substituents while enabling diverse structural modifications through strategic substrate design.
Impurity control is achieved through multiple built-in mechanisms including precise stoichiometric control of sodium bicarbonate that prevents over-reaction pathways while maintaining optimal pH conditions throughout the transformation sequence. The inherent selectivity of the palladium catalyst system minimizes side reactions such as homocoupling or hydrolysis that commonly plague alternative synthetic approaches by favoring specific coordination geometries during key transition states. Post-reaction processing employs standard column chromatography purification techniques that effectively separate minor impurities from the desired product stream without requiring specialized equipment or hazardous solvents. This integrated approach ensures consistent production of high-purity intermediates meeting pharmaceutical industry standards through robust process design rather than relying on end-stage corrective measures that increase manufacturing complexity and cost.
How to Synthesize Trifluoromethyl Imidazole Compounds Efficiently
This patented methodology provides a streamlined pathway for producing high-purity trifluoromethyl imidazole intermediates through carefully optimized reaction parameters that balance efficiency with operational simplicity. The process leverages commercially available starting materials with well-defined molar ratios that ensure complete conversion while minimizing waste generation during manufacturing operations. Detailed standardized synthesis steps are provided below to facilitate seamless technology transfer from laboratory development to commercial production environments while maintaining consistent quality outcomes across all scale levels.
- Prepare the reaction mixture by combining palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate (2.0 equiv), formic acid/acetic anhydride (10 equiv), trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in tetrahydrofuran solvent with precise molar ratios ensuring optimal reactivity.
- Conduct the carbonylation reaction at a controlled temperature of exactly 30°C for precisely twenty hours to achieve complete conversion while maintaining stringent purity standards required for pharmaceutical applications.
- Execute post-reaction processing including filtration through silica gel matrix followed by column chromatography purification to isolate high-purity trifluoromethyl imidazole compounds meeting global regulatory specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain functions by addressing critical pain points associated with traditional intermediate production methods through inherent process efficiencies that translate directly into operational benefits without requiring additional capital investment or specialized infrastructure modifications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts requiring complex removal procedures significantly reduces raw material expenses while decreasing downstream processing costs associated with metal residue purification. Utilization of inexpensive starting materials including readily available diaryl iodonium salts derived from common boronic acids creates substantial cost savings through simplified supply chain logistics and reduced dependency on specialized chemical vendors.
- Enhanced Supply Chain Reliability: The use of commercially accessible raw materials with multiple qualified suppliers ensures consistent availability regardless of regional supply disruptions while enabling flexible sourcing strategies that mitigate single-point failure risks. The robust nature of the process allows seamless transfer between manufacturing sites without revalidation requirements due to its inherent tolerance to minor equipment variations across different production facilities.
- Scalability and Environmental Compliance: The ambient temperature operation eliminates energy-intensive heating or cooling requirements while reducing solvent consumption through optimized reaction concentrations that minimize waste generation per unit output. This environmentally conscious design facilitates straightforward scale-up from laboratory validation directly to commercial production volumes without requiring specialized equipment modifications or additional environmental control systems.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations based on patent specifications and provide insights into implementation strategies that maximize value realization from this innovative manufacturing approach.
Q: How does this method overcome limitations of conventional imidazole synthesis techniques?
A: The patented process eliminates harsh reaction conditions and expensive catalysts required by traditional methods through a mild palladium-catalyzed carbonylation approach operating at ambient temperature. This innovation significantly enhances operational safety while reducing purification complexity and associated costs without compromising yield or purity.
Q: What advantages does substrate flexibility provide for pharmaceutical intermediate manufacturing?
A: The broad functional group tolerance enables pharmaceutical manufacturers to design diverse trifluoromethyl imidazole derivatives with tailored properties by simply modifying aryl groups on starting materials. This flexibility accelerates drug discovery pipelines without requiring new synthetic routes or additional capital investment.
Q: How does this process ensure reliable supply chain continuity for critical intermediates?
A: By utilizing commercially available and inexpensive raw materials with straightforward scalability from laboratory to industrial production volumes, this method guarantees consistent supply continuity while meeting stringent quality specifications demanded by global pharmaceutical clients through robust process control mechanisms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Imidazole Supplier
This patented technology represents a significant advancement in pharmaceutical intermediate manufacturing that aligns perfectly with NINGBO INNO PHARMCHEM's extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our specialized expertise in complex heterocyclic synthesis ensures seamless technology transfer from laboratory development through full-scale commercial implementation while delivering consistent product quality that meets global regulatory requirements across all major markets.
We invite your technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs which will include detailed route feasibility assessments and specific COA data demonstrating how this innovative process can optimize your supply chain operations while enhancing product quality standards.
