Advanced Iodine-Catalyzed Triazole Synthesis: Scaling Pharmaceutical Intermediates from Lab to Commercial Production
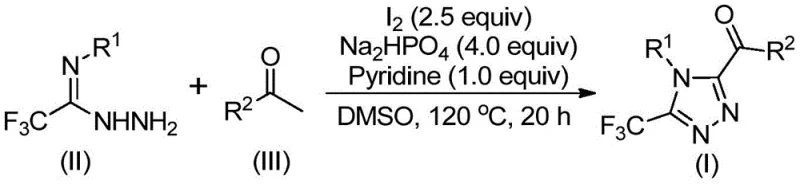
Patent CN113105402B introduces a groundbreaking synthetic methodology for producing high-purity 3,4,5-trisubstituted 1,2,4-triazole compounds through an innovative iodine-catalyzed process that eliminates transition metal requirements while maintaining exceptional functional group tolerance. This novel approach operates under standard atmospheric conditions without requiring anhydrous or oxygen-free environments that typically complicate pharmaceutical intermediate manufacturing processes. The methodology utilizes readily available aryl ketones and trifluoroethylimide hydrazides as starting materials that can be sourced from multiple commercial suppliers globally. By leveraging elemental iodine as a non-toxic catalyst in dimethyl sulfoxide solvent system, the process achieves efficient cyclization to form triazole core structures with simultaneous introduction of trifluoromethyl groups at position five and acyl substituents at position four. This innovation addresses critical limitations in traditional triazole synthesis methods while offering substantial advantages for industrial scale-up operations within pharmaceutical supply chains where purity specifications are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing substituted triazole compounds typically rely on transition metal catalysts such as copper or palladium systems that introduce significant challenges in pharmaceutical manufacturing environments where metal residue limits are strictly regulated by global health authorities. These conventional methods frequently necessitate specialized equipment capable of maintaining strictly anhydrous and oxygen-free conditions throughout the reaction sequence which substantially increases capital investment requirements and operational complexity at commercial scale. The presence of heavy metal catalysts creates additional purification burdens that compromise final product purity profiles while significantly increasing production costs through multiple chromatographic separation steps required to meet regulatory standards. Furthermore, many existing protocols exhibit limited substrate scope with poor functional group tolerance across diverse molecular architectures containing halogen or electron-donating substituents commonly found in pharmaceutical intermediates. These constraints become particularly problematic when scaling processes from laboratory development to commercial manufacturing volumes where process robustness and cost efficiency are critical success factors.
The Novel Approach
The patented methodology overcomes these limitations through an innovative iodine-catalyzed reaction sequence that operates under ambient conditions without requiring specialized equipment or environmental controls beyond standard chemical manufacturing infrastructure. By utilizing elemental iodine as a catalyst in dimethyl sulfoxide solvent system at moderate temperatures between 90°C and 130°C, the process eliminates heavy metal contamination concerns while maintaining excellent reaction efficiency across diverse substrate combinations. The two-stage reaction protocol first generates aryl diketones via iodine/DMSO-mediated oxidation followed by condensation with trifluoroethylimide hydrazides under carefully controlled stoichiometry using sodium dihydrogen phosphate and pyridine as additives at specific molar ratios of 4:1:2.5 respectively. This approach demonstrates remarkable versatility across various substitution patterns including ortho-, meta-, and para-positioned functional groups on both aryl components while maintaining consistent yield characteristics between different substrate combinations as demonstrated in multiple experimental examples within the patent documentation.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with iodine-mediated oxidation of aryl ketones in dimethyl sulfoxide solvent to form α-iodo ketone intermediates that rapidly convert to aryl diketones through Kornblum oxidation at temperatures between 90°C and 110°C over a four-to-six-hour period without requiring additional catalysts or specialized conditions beyond standard heating equipment. This critical transformation step creates the necessary electrophilic centers for subsequent condensation reactions while avoiding side product formation common in alternative oxidation methods. The resulting diketone intermediates then undergo nucleophilic attack by trifluoroethylimide hydrazides (II) in the presence of sodium dihydrogen phosphate buffer system that maintains optimal pH conditions for imine formation followed by dehydration to generate hydrazone intermediates (III). The carefully balanced molar ratio of Na₂HPO₄/pyridine/I₂ creates a synergistic catalytic environment that facilitates the subsequent intramolecular cyclization step through nucleophilic addition followed by ring closure with simultaneous elimination of water molecules.
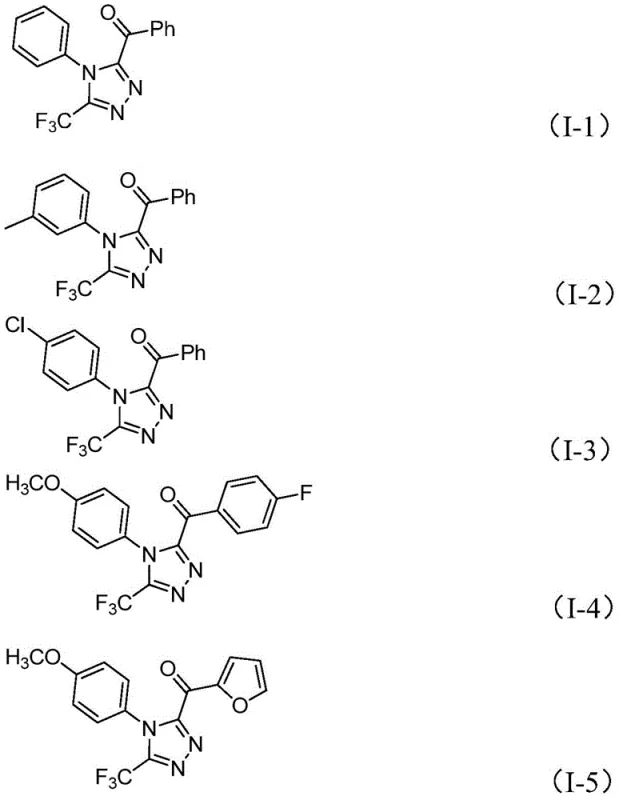
The structural framework of the target compounds (I) features a unique combination of trifluoromethyl group at position five and acyl substituent at position four that significantly enhances physicochemical properties including metabolic stability and membrane permeability while maintaining essential pharmacophore elements required for biological activity in therapeutic applications. This specific substitution pattern improves bioavailability profiles compared to conventional triazole derivatives without introducing additional synthetic complexity or purification challenges during manufacturing operations. The mechanism demonstrates exceptional selectivity with minimal side product formation due to controlled progression through well-defined intermediates that prevent undesired polymerization or decomposition pathways commonly observed in alternative synthetic approaches requiring harsher reaction conditions.
How to Synthesize 3,4,5-Trisubstituted Triazoles Efficiently
This patented synthetic route offers a streamlined approach for producing high-purity triazole intermediates essential for pharmaceutical development programs by leveraging commercially available starting materials and standard laboratory equipment while eliminating complex purification requirements associated with metal-catalyzed processes typically used in heterocyclic chemistry applications. The methodology demonstrates remarkable robustness across diverse substrate combinations containing various functional groups including halogens and electron-donating substituents while maintaining excellent yield characteristics between different molecular architectures as documented in experimental examples within the patent filing. By following the precise reaction sequence detailed in the patent documentation with careful attention to stoichiometric ratios and temperature control parameters manufacturers can achieve consistent product quality across different production scales from laboratory development through commercial manufacturing volumes without requiring significant process reoptimization efforts.
- Prepare aryl diketone intermediates by reacting aryl ethyl ketone with iodine in DMSO solvent at 90-110°C for 4-6 hours under standard atmospheric conditions
- Introduce trifluoroethylimide hydrazide along with sodium dihydrogen phosphate (4 equivalents), pyridine (1 equivalent), and additional iodine (2.5 equivalents) into the reaction mixture
- Conduct cyclization at 110-130°C for 12-20 hours followed by filtration and column chromatography purification to obtain high-purity triazole products
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate production through strategic elimination of costly processing steps while enhancing overall supply chain resilience against market volatility factors affecting traditional synthetic routes requiring specialized materials or equipment configurations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes significant raw material costs while simultaneously reducing downstream purification expenses associated with metal residue removal procedures required by regulatory authorities for drug substance manufacturing operations.
- Enhanced Supply Chain Reliability: Multiple commercial sources exist globally for all required starting materials including aryl ketones and iodine catalysts ensuring robust supply chain continuity even during periods of market volatility or geopolitical disruptions affecting single-source dependencies common in alternative synthetic approaches.
- Scalability and Environmental Compliance: Simplified waste streams resulting from elimination of metal catalysts reduce environmental impact while meeting increasingly stringent regulatory requirements through inherently greener process design that minimizes hazardous waste generation compared to conventional methodologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented triazole synthesis methodology within pharmaceutical manufacturing operations based on detailed analysis of experimental data provided in patent CN113105402B documentation including multiple substrate combinations tested under various reaction conditions.
Q: Does this method require specialized equipment for commercial scale-up?
A: No specialized equipment is required beyond standard chemical manufacturing infrastructure as the process operates under ambient conditions without needing anhydrous or oxygen-free environments.
Q: How does this method address heavy metal contamination concerns in pharmaceutical intermediates?
A: The iodine-catalyzed approach completely eliminates transition metal catalysts that typically cause contamination issues while ensuring compliance with strict regulatory limits for drug substances.
Q: What is the functional group tolerance of this synthetic route?
A: The method demonstrates excellent tolerance across diverse substitution patterns including methyl, methoxy, chloro, bromo, and trifluoromethyl groups at ortho, meta, and para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting trace impurities at parts-per-billion levels required by global regulatory authorities for pharmaceutical intermediates manufacturing operations.
Request our Customized Cost-Saving Analysis to evaluate how this innovative synthesis can optimize your specific manufacturing pathway while meeting all quality requirements; our technical procurement team stands ready to provide detailed COA data and route feasibility assessments tailored to your production needs.
