Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Global Pharmaceutical Manufacturing
Introduction to Next-Generation Triazole Synthesis
The 1,2,4-triazole scaffold represents a cornerstone structural motif in modern medicinal chemistry, serving as the critical pharmacophore in a wide array of blockbuster therapeutics including antiviral agents like Maraviroc, antidiabetic drugs such as Sitagliptin, and iron chelators like Deferasirox. As depicted in the structural overview below, the incorporation of trifluoromethyl groups into these heterocyclic systems further enhances metabolic stability and lipophilicity, making them highly desirable targets for pharmaceutical R&D teams globally. 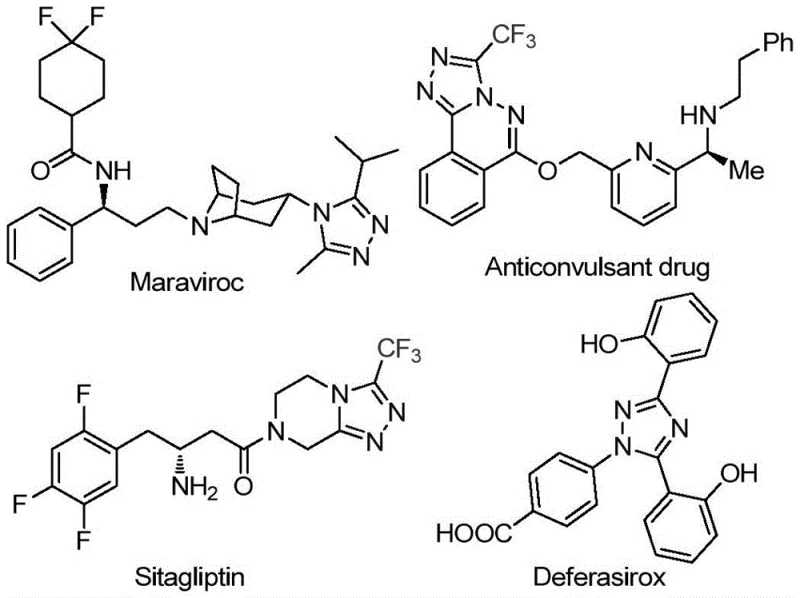 . However, the efficient construction of 3,4,5-trisubstituted 1,2,4-triazoles, particularly those bearing both acyl and trifluoromethyl functionalities, has historically presented significant synthetic challenges. Recent advancements detailed in patent CN113105402B have introduced a transformative approach that addresses these bottlenecks by utilizing a metal-free, iodine-promoted tandem reaction strategy. This innovation not only streamlines the synthetic route but also aligns perfectly with the industry's shift towards greener, more cost-effective manufacturing processes for high-value pharmaceutical intermediates.
. However, the efficient construction of 3,4,5-trisubstituted 1,2,4-triazoles, particularly those bearing both acyl and trifluoromethyl functionalities, has historically presented significant synthetic challenges. Recent advancements detailed in patent CN113105402B have introduced a transformative approach that addresses these bottlenecks by utilizing a metal-free, iodine-promoted tandem reaction strategy. This innovation not only streamlines the synthetic route but also aligns perfectly with the industry's shift towards greener, more cost-effective manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for constructing polysubstituted 1,2,4-triazole rings often rely heavily on the use of transition metal catalysts, such as copper or palladium complexes, which introduce substantial complications in the downstream processing of active pharmaceutical ingredients (APIs). The presence of residual heavy metals necessitates rigorous and expensive purification protocols to meet stringent regulatory limits, thereby inflating the overall cost of goods and extending production lead times. Furthermore, many classical routes require harsh reaction conditions, including strict anhydrous and oxygen-free environments, which demand specialized equipment and increase operational risks during scale-up. The limited substrate scope of older methods often fails to accommodate sensitive functional groups, restricting the chemical diversity available to medicinal chemists during the lead optimization phase. Additionally, the reliance on pre-functionalized starting materials can drive up raw material costs and complicate supply chain logistics for bulk manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113105402B leverages a simple yet powerful iodine-promoted cascade reaction that operates efficiently in dimethyl sulfoxide (DMSO) without the need for exotic catalysts or inert atmospheres. This novel route utilizes readily available aryl ethyl ketones and trifluoroethylimine hydrazides as starting materials, which are commodity chemicals with stable global supply chains. The reaction proceeds through a tandem sequence involving initial iodination and Kornblum oxidation to generate reactive alpha-diketone intermediates in situ, followed immediately by condensation and cyclization. 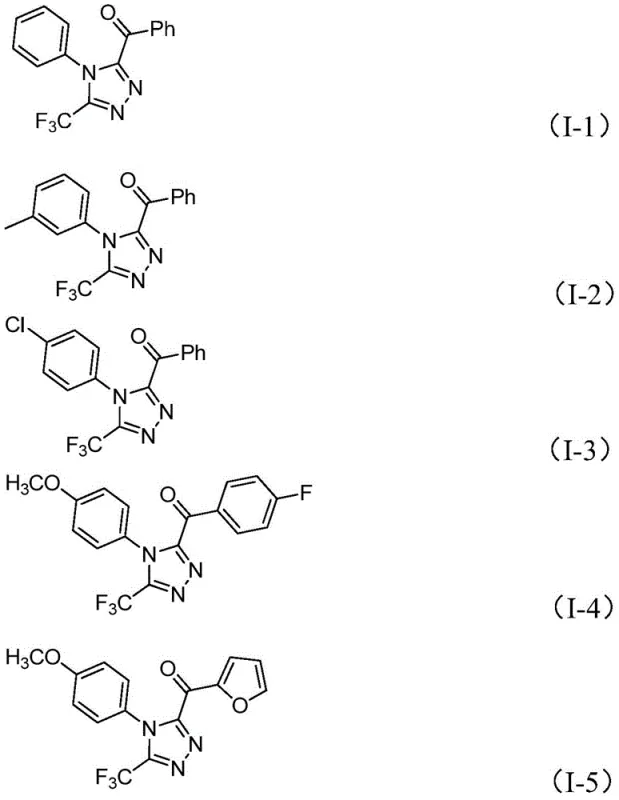 . This one-pot strategy significantly reduces waste generation and solvent consumption compared to multi-step linear syntheses. The robustness of this system allows for easy expansion from gram-scale laboratory experiments to kilogram-level commercial production, offering a reliable pharmaceutical intermediate supplier pathway that balances high purity with economic efficiency.
. This one-pot strategy significantly reduces waste generation and solvent consumption compared to multi-step linear syntheses. The robustness of this system allows for easy expansion from gram-scale laboratory experiments to kilogram-level commercial production, offering a reliable pharmaceutical intermediate supplier pathway that balances high purity with economic efficiency.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this synthetic breakthrough lies in the dual role of elemental iodine, which acts first as an oxidant and subsequently as a promoter for cyclization within the DMSO solvent matrix. The mechanism initiates with the iodination of the aryl ethyl ketone at the alpha-position, followed by a Kornblum oxidation where DMSO acts as the oxygen source to convert the alpha-iodo ketone into an alpha-dicarbonyl species. This oxidative transformation is critical as it generates the electrophilic center necessary for the subsequent nucleophilic attack by the hydrazide nitrogen. The absence of transition metals eliminates the risk of catalyst poisoning by sulfur-containing substrates and removes the need for ligand optimization, simplifying the process development timeline considerably. Following the formation of the hydrazone intermediate via dehydration, the system undergoes an intramolecular cyclization facilitated by the basic environment provided by sodium dihydrogen phosphate and pyridine. 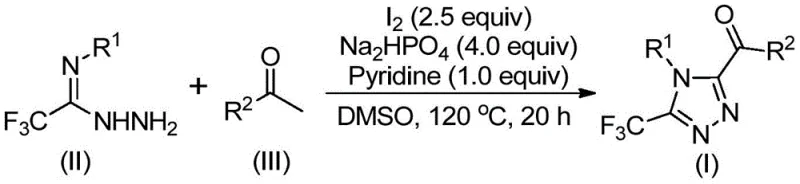 . This final ring-closing step is highly regioselective, ensuring the formation of the desired 3,4,5-substitution pattern with minimal formation of regioisomeric impurities. The entire sequence is thermodynamically driven and kinetically accessible at moderate temperatures ranging from 110°C to 130°C, ensuring high conversion rates while maintaining thermal safety margins for industrial reactors.
. This final ring-closing step is highly regioselective, ensuring the formation of the desired 3,4,5-substitution pattern with minimal formation of regioisomeric impurities. The entire sequence is thermodynamically driven and kinetically accessible at moderate temperatures ranging from 110°C to 130°C, ensuring high conversion rates while maintaining thermal safety margins for industrial reactors.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of metal-complexed byproducts that are notoriously difficult to separate. The primary side reactions are limited to over-oxidation or incomplete cyclization, both of which can be effectively managed by optimizing the stoichiometric ratio of iodine and the reaction duration. The use of sodium dihydrogen phosphate serves as a mild buffer, preventing the degradation of acid-sensitive functional groups on the aromatic rings while facilitating the elimination of water during the condensation phase. This precise control over the reaction microenvironment results in a cleaner crude profile, reducing the burden on downstream purification units such as crystallization or chromatography columns. For R&D directors focused on quality by design (QbD), understanding this mechanistic pathway allows for better prediction of potential genotoxic impurities related to alkyl halides, ensuring that the final API meets ICH M7 guidelines without extensive rework.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical implementation of this synthesis involves a straightforward two-stage heating protocol within a standard glass-lined or stainless steel reactor, making it highly accessible for contract development and manufacturing organizations (CDMOs). The process begins by dissolving the aryl ketone and a portion of elemental iodine in DMSO, followed by an initial heating phase to generate the oxidized intermediate. Subsequently, the trifluoroethylimine hydrazide, base, and remaining iodine are introduced to trigger the cyclization. Detailed standardized operating procedures regarding exact stoichiometry, addition rates, and quenching protocols are essential for reproducibility.
- Mix aryl ethyl ketone and elemental iodine in DMSO solvent and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add trifluoroethylimine hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates directly into tangible operational efficiencies and risk mitigation strategies. By eliminating the dependency on precious metal catalysts, manufacturers can insulate their production costs from the volatile pricing fluctuations of commodities like palladium and platinum, ensuring more stable long-term budgeting. The use of commodity-grade starting materials such as acetophenones and hydrazines ensures a robust supply base with multiple qualified vendors, reducing the risk of single-source bottlenecks that can disrupt API production schedules. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, significantly shortens the overall cycle time from raw material intake to finished intermediate release. This acceleration in throughput allows for greater flexibility in responding to market demand surges and enables just-in-time manufacturing models that minimize inventory holding costs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated ligands results in a direct reduction in raw material expenditure, while the simplified purification process lowers utility and labor costs per kilogram of product. The avoidance of heavy metal removal steps also reduces the consumption of specialized scavenger resins and silica gel, contributing to substantial cost savings in consumables. Additionally, the high atom economy of the tandem reaction minimizes waste disposal fees, further enhancing the overall economic viability of the process for large-scale commercial operations.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the reliance on widely available bulk chemicals rather than specialized custom-synthesized reagents that may have long lead times. The robustness of the reaction conditions, which do not require stringent exclusion of moisture or oxygen, reduces the likelihood of batch failures due to environmental excursions, thereby improving on-time delivery performance. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight production schedules and cannot afford interruptions caused by intermediate shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to proceed efficiently from milligram to gram scales without loss of yield or selectivity, indicating a smooth path to tonnage production. The use of DMSO, a high-boiling polar aprotic solvent, facilitates high-concentration reactions which reduce reactor volume requirements and energy consumption for heating and cooling cycles. Moreover, the absence of toxic heavy metals simplifies wastewater treatment and effluent management, ensuring compliance with increasingly stringent environmental regulations and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metallic promoter, completely eliminating the need for costly palladium or copper catalysts and the associated heavy metal removal steps.
Q: What are the key advantages regarding reaction conditions for scale-up?
A: The process operates under ambient atmospheric conditions without the need for strict anhydrous or oxygen-free environments, significantly simplifying reactor requirements and reducing operational complexity for large-scale production.
Q: Is the substrate scope suitable for diverse pharmaceutical applications?
A: Yes, the method demonstrates high functional group tolerance, successfully accommodating various substituted aryl ketones and hydrazides to produce diverse 3,4,5-trisubstituted scaffolds relevant to drugs like Sitagliptin and Deferasirox.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the fast-paced pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest quality standards required for global regulatory submissions. Our commitment to technical excellence allows us to adapt quickly to specific client needs, whether it involves custom substitution patterns or optimized crystallization forms.
We invite you to engage with our technical procurement team to discuss how this advanced iodine-promoted synthesis can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data for our existing library of triazole derivatives and to request comprehensive route feasibility assessments for your proprietary targets, ensuring a partnership built on transparency and scientific rigor.
