Scalable Metal-Free Synthesis of Bioactive Trifluoromethyl Triazole Intermediates for Global Pharma Supply Chains
Scalable Metal-Free Synthesis of Bioactive Trifluoromethyl Triazole Intermediates for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113105402A introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural scaffold found in prominent drugs such as Maraviroc and Sitagliptin. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by utilizing a non-metallic iodine promotion system in dimethyl sulfoxide (DMSO). The significance of this technology lies in its ability to construct complex trifluoromethyl-substituted triazoles from cheap, commercially available arylethanones and trifluoroethylimine hydrazides without requiring stringent anhydrous or anaerobic conditions. For R&D directors and supply chain managers, this represents a shift towards more sustainable and cost-effective manufacturing processes that maintain high purity standards while minimizing environmental impact.
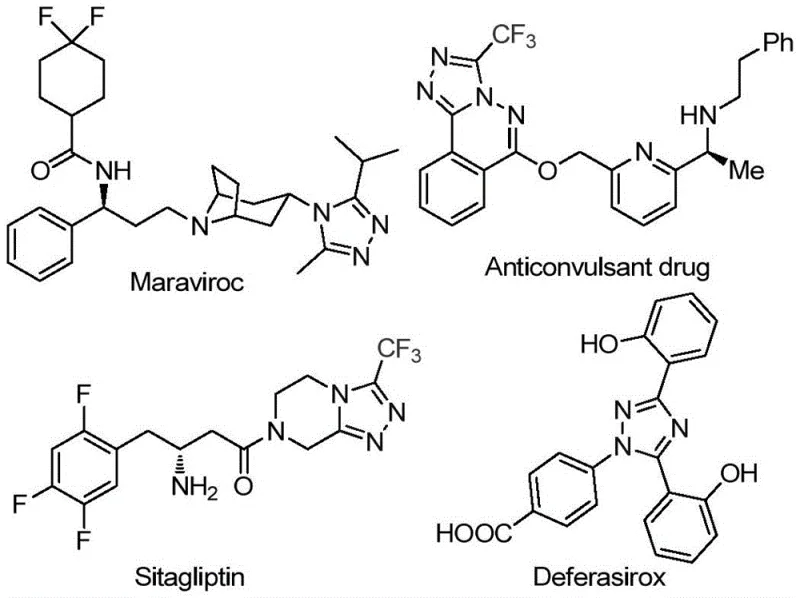
The strategic value of this patent extends beyond mere academic interest; it offers a practical solution for the reliable pharmaceutical intermediate supplier seeking to optimize their portfolio. By leveraging the unique reactivity of iodine in DMSO, the process facilitates a tandem oxidation-cyclization sequence that was previously difficult to achieve with high efficiency. The introduction of the trifluoromethyl group directly into the heterocyclic core during the cyclization step ensures that the final products possess the desired lipophilicity and electronic properties essential for modern drug design. This method not only widens the applicability of triazole synthesis but also provides a versatile platform for generating diverse libraries of compounds for medicinal chemistry campaigns, thereby accelerating the drug discovery timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, especially those bearing both acyl and trifluoromethyl groups, has relied heavily on transition metal catalysis or harsh reaction conditions that pose significant challenges for industrial scale-up. Conventional routes often necessitate the use of expensive palladium or copper catalysts, which not only inflate the raw material costs but also introduce complex downstream processing requirements to remove trace metal residues to meet strict pharmacopeial limits. Furthermore, many existing protocols demand rigorously anhydrous and oxygen-free environments, requiring specialized equipment such as gloveboxes or Schlenk lines, which drastically increases capital expenditure and operational complexity. These factors collectively contribute to longer lead times and higher production costs, creating a bottleneck for the commercial scale-up of complex pharmaceutical intermediates. Additionally, the limited substrate scope of some traditional methods restricts the structural diversity achievable, hindering the optimization of lead compounds in early-stage drug development.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a metal-free system driven by elemental iodine and DMSO, effectively bypassing the limitations associated with heavy metal catalysis. This method operates under relatively mild conditions where the reaction does not need to be carried out under anhydrous and oxygen-free conditions, significantly simplifying the operational protocol. The process begins with the iodination and Kornblum oxidation of arylethanones to generate reactive aryldiketone intermediates in situ, which then undergo tandem cyclization with trifluoroethylimine hydrazide. This one-pot strategy minimizes solvent usage and waste generation, aligning with green chemistry principles. The use of cheap and easily obtainable starting materials like arylethanones further enhances the economic viability of the process. By avoiding toxic heavy metals and simplifying the reaction setup, this novel approach offers a streamlined pathway that is highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation reactions facilitated by the unique properties of the iodine-DMSO system. Initially, the arylethanone substrate undergoes alpha-iodination followed by Kornblum oxidation, mediated by DMSO acting as both solvent and oxidant, to yield an alpha-dicarbonyl species. This highly reactive intermediate is crucial for the subsequent steps. Upon the addition of trifluoroethylimine hydrazide, a dehydration condensation occurs to form a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine acts as a base buffer system, facilitating the deprotonation steps necessary for the final ring closure. The elemental iodine continues to play a catalytic role in promoting the intramolecular cyclization, leading to the formation of the stable 1,2,4-triazole ring. This mechanism ensures high atom economy and minimizes the formation of unwanted byproducts, resulting in a cleaner reaction profile.
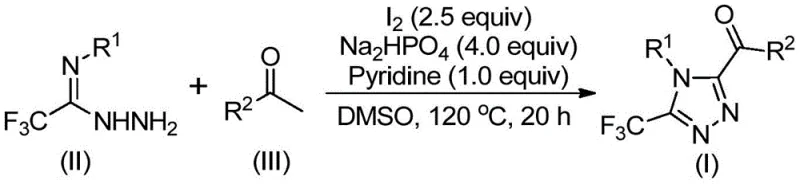
Understanding the impurity profile is vital for ensuring the quality of the final active pharmaceutical ingredient. The controlled addition of reagents and the specific temperature gradients (90-110°C followed by 110-130°C) are designed to maximize the conversion of the intermediate hydrazone to the desired triazole while suppressing side reactions such as over-oxidation or polymerization. The use of pyridine helps to neutralize the hydrogen iodide generated during the reaction, preventing acid-catalyzed decomposition of sensitive functional groups. Post-reaction processing involves simple filtration and silica gel mixing, followed by standard column chromatography, which effectively removes residual iodine and inorganic salts. This robust purification capability ensures that the final product meets stringent purity specifications, making it suitable for direct use in subsequent coupling reactions or biological testing without extensive rework.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for producing high-quality triazole derivatives. The procedure is designed to be user-friendly, requiring standard laboratory glassware and common reagents, which lowers the barrier to entry for adoption. The reaction conditions are optimized to balance reaction rate and selectivity, ensuring consistent results across different batches. Detailed standardized synthesis steps are provided below to guide process chemists in implementing this technology.
- Mix arylethanone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add trifluoroethylimine hydrazide, additional iodine, sodium dihydrogen phosphate, and pyridine to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route offers tangible benefits that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts removes a major cost driver and supply chain vulnerability, as the prices of metals like palladium can be volatile and their sourcing subject to geopolitical instability. Furthermore, the simplified workup procedure reduces the consumption of solvents and adsorbents, leading to substantial cost savings in waste disposal and material handling. The ability to run the reaction without strict inert atmosphere requirements means that existing manufacturing infrastructure can be utilized without significant retrofitting, accelerating the time-to-market for new products.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the replacement of expensive transition metal catalysts with inexpensive elemental iodine and the use of commodity chemicals like arylethanones as starting materials. This shift drastically reduces the raw material cost per kilogram of the final intermediate. Additionally, the simplified purification process, which avoids complex metal scavenging steps, lowers the overall processing costs. The high conversion rates observed in the patent examples suggest that less starting material is wasted, further improving the process mass intensity (PMI) and contributing to a more lean and efficient manufacturing operation.
- Enhanced Supply Chain Reliability: By relying on widely available and stable reagents such as DMSO, iodine, and substituted acetophenones, the supply chain becomes more robust against disruptions. These materials are produced at a global scale and are not subject to the same supply constraints as specialized ligands or organometallic catalysts. The operational simplicity of the method, which tolerates ambient moisture and oxygen to a degree, also reduces the risk of batch failures due to environmental control issues. This reliability ensures a steady flow of high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production schedules with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: The method has been demonstrated to be easily expandable to the gram level and is inherently suitable for kilogram-scale production due to its homogeneous nature and manageable exotherm. The absence of heavy metals simplifies the environmental compliance landscape, as there is no need for extensive wastewater treatment to remove toxic metal ions. This aligns with increasingly strict environmental regulations and corporate sustainability goals. The use of DMSO, while requiring proper recovery systems, is a well-understood solvent in the industry, and the overall greener profile of the reaction makes it an attractive option for companies aiming to reduce their carbon footprint and improve their E-factor.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing pipelines.
Q: What are the primary advantages of this iodine-promoted method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, significantly simplifying the purification process and reducing the risk of metal residue in the final API, which is critical for regulatory compliance.
Q: Does this synthesis require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits described in the patent is that the reaction proceeds efficiently without the need for rigorous anhydrous or oxygen-free environments, lowering equipment and operational costs.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The method demonstrates wide functional group tolerance, successfully accommodating various substituted aryl groups including methyl, methoxy, chloro, and trifluoromethyl substituents at ortho, meta, and para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-promoted triazole synthesis described in patent CN113105402A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole meets the highest industry standards. We are committed to leveraging this innovative chemistry to deliver superior value to our global partners.
We invite you to collaborate with us to explore how this cost-effective and scalable technology can enhance your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the economic benefits of switching to this metal-free route. Please contact our technical procurement team today to request specific COA data for our triazole library and to discuss route feasibility assessments for your target molecules. Let us help you accelerate your path to market with efficient and sustainable chemical solutions.
