Revolutionizing Thioester Production: A Scalable Nickel-Catalyzed Carbonylation Strategy for Global Supply Chains
Revolutionizing Thioester Production: A Scalable Nickel-Catalyzed Carbonylation Strategy for Global Supply Chains
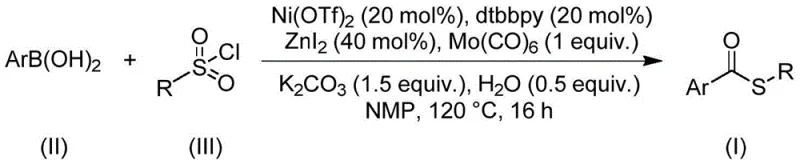
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly for versatile building blocks like thioester compounds. A significant breakthrough in this domain is documented in Chinese Patent CN112239384B, which discloses a novel preparation method for thioester compounds that addresses long-standing challenges in efficiency and substrate tolerance. This patented technology utilizes a sophisticated nickel-catalyzed carbonylation reaction, employing readily available arylboronic acids and sulfonyl chlorides as starting materials to construct the thioester backbone with high precision. The significance of this development cannot be overstated, as thioesters serve as critical scaffolds in numerous bioactive molecules, including well-known pharmaceutical agents such as Timobesone, Pivalopril, and Spironolactone, highlighting the immense commercial potential of optimizing their synthesis.
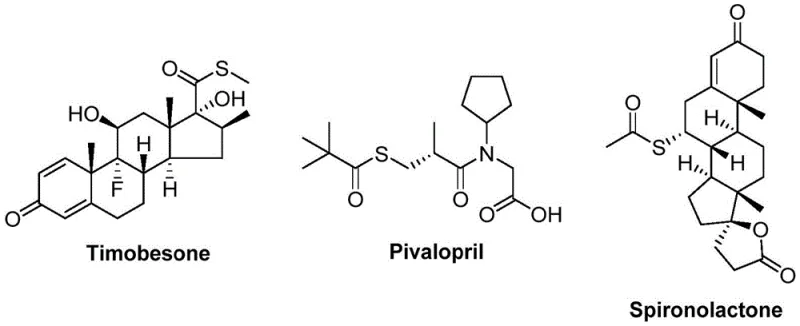
For R&D directors and process chemists, the ability to access these structures through a robust, catalytic pathway represents a paradigm shift from traditional stoichiometric methods. The patent outlines a procedure that operates under relatively mild thermal conditions, specifically around 120°C, utilizing a dual-metal system involving nickel and molybdenum to facilitate the carbonyl insertion. This approach not only streamlines the synthetic route but also significantly enhances the functional group compatibility, allowing for the incorporation of diverse substituents such as halogens, alkyl groups, and alkoxy groups without compromising the integrity of the final product. By leveraging this technology, manufacturers can achieve a level of molecular complexity and purity that is essential for meeting the stringent regulatory standards of modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has relied heavily on the direct acylation of thiols with carboxylic acids or their activated derivatives, a process fraught with significant operational and safety drawbacks. Thiols, while effective nucleophiles, are notoriously malodorous and volatile, posing severe health and safety risks in large-scale manufacturing environments where containment is difficult to maintain perfectly. Furthermore, the sulfur atom in thiols has a high affinity for transition metals, often leading to catalyst poisoning in metal-mediated reactions, which necessitates the use of excessive catalyst loading or forces chemists to resort to non-catalytic, waste-generating stoichiometric reagents. Another critical limitation is the instability of many thiol substrates, which can oxidize easily to disulfides, thereby reducing the overall atom economy and complicating the purification process with difficult-to-remove byproducts. These factors collectively contribute to increased production costs, extended lead times, and a larger environmental footprint, making conventional thiol-based routes increasingly unattractive for sustainable industrial chemistry.
The Novel Approach
In stark contrast, the methodology described in patent CN112239384B introduces a transformative strategy that replaces hazardous thiols with stable, odorless sulfonyl chlorides as the sulfur source. This substitution fundamentally alters the reaction profile, eliminating the issues of odor and catalyst deactivation while opening up new avenues for substrate design. The reaction employs arylboronic acids, which are widely available and stable solids, reacting them with sulfonyl chlorides in the presence of a nickel catalyst and a molybdenum carbonyl source to effectuate the carbonylation. This novel approach allows for a broader scope of substrates, tolerating sensitive functional groups that would otherwise be incompatible with harsh acidic or basic conditions found in traditional acylation methods. Moreover, the process does not require additional external oxidants or reductants, simplifying the reaction setup and reducing the generation of chemical waste, which aligns perfectly with the principles of green chemistry and sustainable manufacturing practices demanded by today's supply chain leaders.
Mechanistic Insights into Nickel-Catalyzed Carbonylation
The core of this innovative synthesis lies in the intricate interplay between the nickel catalyst and the molybdenum carbonyl complex, which together orchestrate the formation of the carbon-sulfur and carbon-carbon bonds necessary for the thioester structure. The reaction mechanism likely initiates with the oxidative addition of the sulfonyl chloride to the low-valent nickel species, generating a reactive nickel-sulfur intermediate. Simultaneously, the molybdenum hexacarbonyl serves as a safe and controllable source of carbon monoxide, releasing CO in situ under the thermal conditions of 120°C to insert into the nickel-carbon or nickel-sulfur bond. This carbonyl insertion step is critical, as it builds the central carbonyl group of the thioester functionality without the need for handling high-pressure CO gas, thereby enhancing operational safety. The subsequent transmetallation with the arylboronic acid, facilitated by the base potassium carbonate and the additive zinc iodide, delivers the aryl group to the metal center, followed by reductive elimination to release the final thioester product and regenerate the active nickel catalyst.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over radical-based or high-energy thermal processes. The use of a well-defined catalytic cycle ensures that side reactions, such as homocoupling of the boronic acid or decomposition of the sulfonyl chloride, are minimized through careful tuning of the ligand environment, specifically using 4,4'-di-tert-butyl-2,2'-bipyridine (dtbbpy). The presence of zinc iodide acts as a crucial promoter, likely assisting in the activation of the sulfonyl chloride or stabilizing intermediate species, which contributes to the high yields observed across various substrate combinations, ranging from 62% to 90% in the reported examples. This high level of selectivity means that the crude reaction mixture contains fewer byproducts, significantly reducing the burden on downstream purification processes like column chromatography or recrystallization, which is a key consideration for maintaining high throughput in commercial production facilities.
How to Synthesize Thioester Compounds Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific protocols regarding reagent ratios and reaction parameters to maximize yield and purity. The process involves charging a reaction vessel with precise amounts of nickel triflate, the bipyridine ligand, molybdenum carbonyl, and additives before introducing the substrates in a polar aprotic solvent like N-methylpyrrolidone (NMP). Maintaining the temperature at 120°C for a duration of 16 hours is critical to ensure complete conversion of the starting materials while avoiding thermal degradation of the product. For a comprehensive understanding of the exact stoichiometry and workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Combine nickel triflate, dtbbpy ligand, molybdenum carbonyl, zinc iodide, potassium carbonate, and water in an organic solvent like NMP.
- Add the arylboronic acid and sulfonyl chloride substrates to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 120°C for 16 hours, then perform filtration and column chromatography to isolate the pure thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed protocol offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience. The shift from malodorous thiols to stable sulfonyl chlorides eliminates the need for specialized scrubbing systems and extensive personal protective equipment associated with handling volatile sulfur compounds, thereby reducing capital expenditure on safety infrastructure. Furthermore, the reliance on commodity chemicals like arylboronic acids and sulfonyl chlorides, which are produced on a massive global scale, ensures a secure and continuous supply of raw materials, mitigating the risk of shortages that often plague niche reagent markets. This stability in sourcing allows for more accurate forecasting and inventory management, essential components of a lean and responsive supply chain strategy in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and hazardous reagents, as well as the reduction in waste disposal costs associated with thiol byproducts. By utilizing a catalytic amount of nickel rather than stoichiometric activators, the material cost per kilogram of product is significantly lowered, while the high atom economy of the carbonylation reaction ensures that a greater proportion of the input mass is converted into valuable product. Additionally, the simplified workup procedure, which often requires only filtration and standard purification, reduces labor hours and solvent consumption, leading to substantial overall cost savings in the manufacturing of complex organic intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups and moisture levels (with the addition of water as a promoter), makes the process less susceptible to variations in raw material quality or environmental fluctuations. This tolerance translates to higher batch-to-batch consistency and fewer failed runs, ensuring that delivery schedules to downstream customers are met reliably. The use of shelf-stable solid reagents also simplifies logistics and storage, as there is no need for refrigerated transport or strict exclusion of air during warehousing, further streamlining the supply chain operations.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the absence of high-pressure gas cylinders, as the carbon monoxide is generated safely in situ from a solid precursor. This inherent safety feature lowers the barrier for scale-up in multipurpose plants that may not be equipped for high-pressure carbonylations. Moreover, the reduced generation of sulfur-containing waste and the avoidance of heavy metal contaminants in the final product simplify the environmental compliance process, making it easier to obtain the necessary permits for commercial production and aligning with increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thioester synthesis technology, providing clarity for stakeholders evaluating its potential for their specific applications. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of using sulfonyl chlorides over thiols in thioester synthesis?
A: Sulfonyl chlorides are odorless, stable solids that do not poison catalysts, unlike thiols which are malodorous and can deactivate transition metal catalysts, leading to higher reaction efficiency and easier handling.
Q: What is the typical yield range for this nickel-catalyzed carbonylation process?
A: According to the patent data, the reaction yields typically range from 62% to 90%, depending on the specific substrate combinations of arylboronic acids and sulfonyl chlorides used.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method uses commercially available raw materials, operates at moderate temperatures (120°C), and avoids expensive oxidants or reductants, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compounds Supplier
As the demand for high-quality thioester intermediates continues to grow in the development of next-generation therapeutics and agrochemicals, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch, thereby safeguarding your downstream processes and final product integrity.
We invite you to collaborate with us to leverage this innovative nickel-catalyzed technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, as well as obtain specific COA data and route feasibility assessments to validate the suitability of this method for your target molecules. Let us help you optimize your supply chain and accelerate your time to market with our reliable thioester compounds supply capabilities.
