Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Commercial Scale-Up
Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine Guidance for Commercial Scale-Up
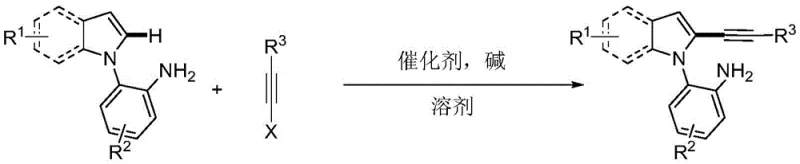
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high precision and minimal environmental impact. A significant breakthrough in this domain is detailed in Chinese Patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology addresses a long-standing challenge in organic synthesis: the selective functionalization of the C2 position of the indole ring, which is traditionally difficult due to the higher electron density at the C3 position. By leveraging a primary amine as a directing group, this process enables the efficient cross-coupling of 2-(1H-indol-1-yl)aniline derivatives with alkyne halides under mild conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing routes for high-value pharmaceutical intermediates.
The core innovation lies in the utilization of simple, commercially available starting materials to generate highly functionalized indole derivatives. The reaction system employs a palladium salt catalyst and a cesium-based base, operating effectively at temperatures between 80°C and 110°C. Crucially, the process is compatible with water as a solvent, aligning perfectly with the principles of green chemistry. This capability not only reduces the reliance on volatile organic compounds (VOCs) but also simplifies the downstream workup procedures. For supply chain managers, the implication is clear: a safer, more scalable process that mitigates regulatory risks associated with hazardous solvent usage while maintaining high atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-substituted indoles has been fraught with synthetic hurdles. Traditional approaches often rely on pre-functionalized indole precursors, such as 2-haloindoles, which require multi-step preparation and generate stoichiometric amounts of waste. Furthermore, direct C-H activation strategies, while attractive in theory, frequently suffer from poor regioselectivity. The inherent electronic bias of the indole nucleus favors electrophilic attack or metallation at the C3 position, leading to mixtures of regioisomers that are difficult and costly to separate. Many existing transition-metal catalyzed methods necessitate the use of expensive, air-sensitive ligands or harsh reaction conditions that are incompatible with sensitive functional groups. These limitations result in lower overall yields, increased production costs, and significant safety concerns during large-scale operations, making them less ideal for the rigorous demands of modern API manufacturing.
The Novel Approach
In stark contrast, the method disclosed in CN108864164B offers a streamlined solution by exploiting the coordinating ability of a proximal primary amine group. This directing group effectively overrides the natural electronic preferences of the indole ring, guiding the palladium catalyst exclusively to the C2 position. The reaction proceeds with excellent regioselectivity, eliminating the formation of C3-alkynylated byproducts. Moreover, the protocol is remarkably tolerant of diverse functional groups, including halogens, ethers, and nitriles, allowing for the late-stage diversification of the molecular scaffold. The use of water as a solvent is a game-changer, drastically reducing the cost of goods sold (COGS) related to solvent purchase and disposal. This approach transforms a complex synthetic challenge into a robust, operationally simple procedure suitable for industrial application.
Mechanistic Insights into Pd-Catalyzed C-H Alkynylation
The success of this transformation hinges on a sophisticated yet elegant catalytic cycle driven by the synergy between the palladium center and the primary amine directing group. Initially, the palladium catalyst coordinates with the nitrogen atom of the primary amine moiety on the 2-(1H-indol-1-yl)aniline substrate. This coordination facilitates the activation of the proximal C2-H bond through a concerted metalation-deprotonation (CMD) pathway or a similar mechanism, resulting in the formation of a stable six-membered palladacycle intermediate. This cyclometalated species is the key to achieving high regioselectivity, as it locks the metal center in close proximity to the C2 carbon, preventing reaction at the more reactive C3 site. The stability of this intermediate ensures that the subsequent steps proceed efficiently without competing side reactions.
Following the formation of the palladacycle, the alkyne halide undergoes oxidative addition to the palladium center. This step introduces the alkynyl fragment into the coordination sphere of the metal. Subsequently, a reductive elimination event occurs, forging the new carbon-carbon bond between the indole C2 position and the alkyne moiety. This final step releases the desired 2-alkynyl indole product and regenerates the active palladium catalyst, allowing the cycle to continue. The choice of base, typically a cesium salt like cesium pivalate, plays a critical role in neutralizing the acid generated during the C-H activation step and maintaining the catalytic turnover. This mechanistic understanding underscores the reliability of the process, providing R&D teams with confidence in its reproducibility and scalability for complex molecule synthesis.
How to Synthesize 2-Alkynyl Indole Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory or plant equipment without the need for specialized high-pressure reactors or inert atmosphere gloveboxes for every step. The procedure involves charging a reactor with the indole-aniline substrate, the triisopropylsilyl-protected alkyne halide, a palladium source such as palladium tetranitrate tetrafluoroborate, and a base like cesium pivalate. Water serves as the primary reaction medium, creating a heterogeneous or homogeneous mixture depending on the specific substrates used. The reaction is heated to a moderate temperature range of 80°C to 110°C and stirred for a period of 12 to 24 hours. Upon completion, the product is isolated through a straightforward extraction with ethyl acetate, followed by drying and purification via column chromatography. Detailed standardized synthesis steps are provided in the guide below.
- Charge the reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium catalyst, base, and water solvent.
- Heat the reaction mixture to 80-110°C and stir for 12-24 hours to facilitate the cross-coupling reaction.
- Cool the mixture, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical feasibility. The shift towards water-based solvent systems represents a significant reduction in operational expenditures related to solvent procurement, storage, and hazardous waste treatment. By eliminating the need for large volumes of toxic organic solvents, manufacturers can achieve drastic cost savings while simultaneously enhancing workplace safety and environmental compliance. Furthermore, the use of readily available starting materials, such as simple o-iodoaniline derivatives and indoles, ensures a stable and resilient supply chain. This reduces the risk of production delays caused by the scarcity of exotic reagents, thereby securing continuity of supply for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high atom economy and the elimination of expensive, specialized ligands often required in traditional C-H activation protocols. By utilizing common palladium salts and inexpensive inorganic bases, the raw material costs are significantly minimized. Additionally, the simplified workup procedure, which avoids complex quenching steps or extensive solvent exchanges, reduces labor and utility costs. The ability to run the reaction in water further lowers the energy burden associated with solvent recovery and distillation, contributing to a leaner and more cost-efficient manufacturing profile that directly improves the bottom line for bulk production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly into supply chain resilience. The tolerance for various functional groups means that a single set of reaction conditions can be applied to a wide library of substrates, simplifying inventory management and reducing the need for multiple specialized process lines. The starting materials are commodity chemicals with established global supply networks, mitigating the risk of bottlenecks. This flexibility allows manufacturers to respond rapidly to changing market demands or custom synthesis requests, ensuring that lead times for high-purity pharmaceutical intermediates are kept to a minimum without compromising on quality or consistency.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the use of water as a solvent, which offers superior heat transfer properties and reduces the risk of thermal runaway compared to flammable organic solvents. The green nature of the process aligns with increasingly stringent environmental regulations, future-proofing the manufacturing facility against tighter emission standards. The reduction in hazardous waste generation simplifies disposal logistics and lowers compliance costs. This makes the technology not only scientifically sound but also commercially sustainable for long-term, large-scale production of complex fine chemicals and API precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the key advantage of using a primary amine as a directing group in this synthesis?
A: The primary amine acts as a powerful coordinating group that directs the palladium catalyst specifically to the C2 position of the indole ring, overcoming the natural electronic preference for C3 functionalization and ensuring high regioselectivity.
Q: Can this reaction be performed in environmentally friendly solvents?
A: Yes, the patented method explicitly utilizes water as the primary solvent or in a mixture with toluene, significantly reducing the environmental footprint compared to traditional organic solvent-heavy processes.
Q: What types of substituents are tolerated on the indole and aniline rings?
A: The method demonstrates broad substrate applicability, tolerating various groups such as methyl, fluoro, chloro, methoxy, and cyano on the indole ring, as well as methyl groups on the aniline ring, without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the primary amine-guided alkynylation technology for the next generation of pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise to translate this academic innovation into a commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our infrastructure is designed to handle complex heterocyclic chemistry safely and efficiently, guaranteeing a consistent supply of high-quality materials for your drug development programs.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Whether you require custom synthesis of novel indole derivatives or scale-up of existing intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this green, cost-effective methodology can enhance your supply chain efficiency and reduce your overall manufacturing costs.
