Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine-Guided Palladium Catalysis for Commercial Scale
Advanced Synthesis of 2-Alkynyl Indoles via Primary Amine-Guided Palladium Catalysis for Commercial Scale
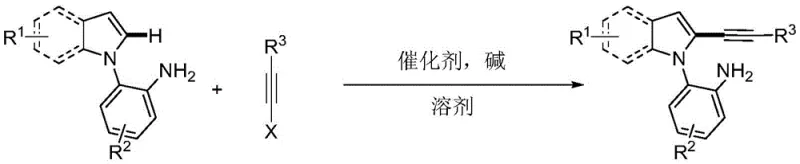
The pharmaceutical and fine chemical industries are constantly seeking more efficient, selective, and environmentally benign routes to construct complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN108864164B, which discloses a novel synthesis method for primary amine-guided 2-alkynyl indole compounds. This technology addresses long-standing challenges in regioselective functionalization by leveraging a unique directing group strategy. Unlike traditional methods that often struggle with mixtures of isomers or require harsh halogenation steps, this approach utilizes a palladium-catalyzed cross-coupling reaction between 2-(1H-indol-1-yl)aniline derivatives and alkyne halides. The process is notable for its use of water as a primary solvent, marking a substantial shift towards green chemistry in the production of high-value pharmaceutical intermediates. For R&D directors and procurement managers alike, this represents a pathway to higher purity profiles and reduced operational expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of functionalized indole derivatives has relied heavily on classical cross-coupling reactions preceded by C-H halogenation. This multi-step sequence is inherently inefficient, generating stoichiometric amounts of salt waste and requiring rigorous purification between steps to remove halogenated byproducts. Furthermore, direct C-H activation strategies, while atom-economical in theory, have faced significant hurdles regarding regioselectivity. Due to the higher electron cloud density at the C3-position of the indole ring compared to the C2-position, electrophilic metallation naturally favors the C3-site. Achieving exclusive C2-alkynylation without protecting groups or complex templating systems has remained a formidable challenge, often resulting in difficult-to-separate isomeric mixtures that compromise the purity specifications required for active pharmaceutical ingredients (APIs).
The Novel Approach
The methodology described in the patent data revolutionizes this landscape by introducing a primary amine as a powerful directing group. By coordinating the palladium catalyst with the amino group on the adjacent aniline ring, the reaction trajectory is steered specifically towards the C2-position of the indole core. This intramolecular coordination forms a transient six-membered palladacycle, effectively overriding the innate electronic bias of the indole ring. The result is a highly regioselective transformation that constructs a series of highly functionalized 2-alkynyl indole compounds in a single pot. Moreover, the ability to conduct this reaction in water or water-toluene mixtures at moderate temperatures (80-110°C) eliminates the need for expensive, toxic organic solvents, thereby simplifying the workup procedure and enhancing the safety profile of the manufacturing process.
Mechanistic Insights into Pd-Catalyzed C2-Alkynylation
The core of this technological advancement lies in the precise mechanistic pathway facilitated by the palladium salt catalyst. Under the promotion of a base, such as cesium pivalate or potassium acetate, the 2-(1H-indol-1-yl)aniline substrate coordinates with the palladium center. This coordination is the critical determinant of selectivity; the nitrogen lone pair of the primary amine guides the metal to the proximal C2-H bond, facilitating C-H activation to form a stable six-membered ring palladium intermediate. This cyclometalated species is far more stable than any potential C3-activated species, ensuring that subsequent steps proceed exclusively at the desired position. Following this activation, the alkyne halide undergoes oxidative addition to the palladium center, followed by reductive elimination to release the final 2-alkynyl indole product and regenerate the active catalyst species.
From an impurity control perspective, this mechanism offers distinct advantages. The high specificity of the directing group minimizes the formation of regioisomers, which are often the most difficult impurities to remove during crystallization or chromatography. Additionally, the use of well-defined palladium salts, such as palladium chloride or palladium acetate, allows for tight control over the catalytic cycle, reducing the risk of homocoupling side reactions common in alkyne chemistry. The broad substrate tolerance demonstrated in the patent examples, accommodating various substituents like fluoro, chloro, methyl, and methoxy groups on both the indole and aniline rings, indicates a robust catalytic system that maintains fidelity even with sterically demanding or electronically diverse substrates. This reliability is crucial for maintaining consistent quality in commercial batches.
How to Synthesize 2-Alkynyl Indole Efficiently
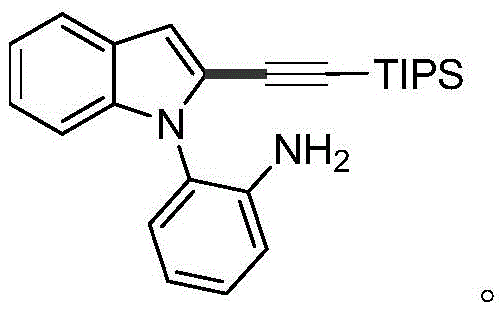
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the precise charging of the 2-(1H-indol-1-yl)aniline compound, the alkyne halide (typically triisopropylsilylpropargyl bromide), and the palladium catalyst into a reactor. The choice of base and solvent ratio is critical; while water is the preferred green solvent, mixtures with toluene can be utilized to improve solubility for certain substrates. The reaction is typically stirred at 100°C for approximately 24 hours to ensure complete conversion. Detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below for technical reference.
- Charge a reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium salt catalyst, base, and water solvent.
- Stir the reaction mixture at a temperature range of 80-110°C for 12-24 hours to facilitate the cross-coupling reaction.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this primary amine-guided synthesis offers tangible strategic benefits beyond mere technical novelty. The shift towards aqueous media and the use of readily available starting materials directly address key pain points in the supply of complex pharmaceutical intermediates. By eliminating the need for pre-functionalized halogenated indoles, the supply chain becomes less dependent on specialized upstream vendors, thereby reducing lead times and mitigating supply disruption risks. Furthermore, the simplified downstream processing associated with water-based reactions translates into significant operational efficiencies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of expensive organic solvents and the reduction of waste treatment costs. Traditional methods often require large volumes of dichloromethane or DMF, which are costly to purchase and dispose of. By utilizing water as the main solvent, the process drastically cuts down on raw material expenses and environmental compliance fees. Additionally, the high atom economy of the direct cross-coupling reaction means less raw material is wasted in the form of byproducts, leading to substantial cost savings in the overall cost of goods sold (COGS) without compromising on yield.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically 2-(1H-indol-1-yl)aniline derivatives and alkyne halides, are synthesized from commodity chemicals like o-iodoaniline and indole. These precursors are widely produced and available from multiple global suppliers, ensuring a stable and competitive supply base. This diversity in sourcing options prevents bottlenecks that often occur with proprietary or highly specialized reagents. Consequently, manufacturers can secure long-term supply agreements with greater confidence, ensuring continuity of production for critical drug substances.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety hazards, particularly when dealing with exothermic reactions in volatile organic solvents. This method operates at moderate temperatures and uses non-flammable water, significantly lowering the safety risk profile during scale-up from pilot plant to commercial tonnage. The reduced generation of hazardous organic waste simplifies the permitting process for new manufacturing lines and ensures compliance with increasingly stringent environmental regulations. This ease of scale-up allows for rapid response to market demand fluctuations, making it an ideal candidate for commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the method for industrial partners.
Q: How does this method achieve C2-selectivity over C3-alkynylation?
A: The method utilizes the primary amine group on the aniline ring as a directing group. This coordinates with the palladium catalyst to form a stable six-membered palladacycle intermediate, which kinetically favors activation and functionalization at the C2 position of the indole ring, overcoming the natural electron density preference for the C3 position.
Q: What are the environmental benefits of using water as a solvent?
A: Using water eliminates the need for volatile organic compounds (VOCs), significantly reducing hazardous waste disposal costs and fire risks. It aligns with green chemistry principles, simplifies downstream processing, and lowers the overall environmental footprint of the manufacturing process compared to traditional organic solvents.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates at moderate temperatures (80-110°C) and uses commercially available, inexpensive starting materials like o-iodoaniline derivatives and indoles. The robustness of the palladium catalyst system and the use of water as a medium make it highly suitable for scale-up from laboratory to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
The development of primary amine-guided 2-alkynyl indole synthesis represents a significant leap forward in heterocyclic chemistry, offering a blend of high selectivity and environmental sustainability. At NINGBO INNO PHARMCHEM, we recognize the potential of this technology to streamline the production of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 2-alkynyl indole intermediate meets the exacting standards required by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in palladium-catalyzed cross-coupling can accelerate your drug development timeline and optimize your manufacturing budget.
