Advanced Synthesis of 2-Alkynyl Indole Compounds via Primary Amine Guidance
The pharmaceutical and fine chemical industries are constantly seeking efficient, scalable, and environmentally benign routes to construct complex heterocyclic scaffolds, particularly indole derivatives which serve as privileged structures in drug discovery. Patent CN108864164B, published in early 2021, introduces a groundbreaking synthetic methodology for the preparation of primary amine-guided 2-alkynyl indole compounds. This technology represents a significant leap forward in transition metal-catalyzed C-H bond functionalization, specifically addressing the long-standing challenge of regioselective alkynylation at the indole C2-position. By leveraging a simple primary amine as a directing group and employing water as a green solvent, this process offers a robust alternative to traditional multi-step syntheses. For R&D directors and procurement managers alike, understanding the mechanistic elegance and operational simplicity of this patent is crucial for evaluating its potential in reducing the cost of goods sold (COGS) for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of functionalized indole derivatives has relied heavily on pre-functionalization strategies that are inherently inefficient and wasteful. The classical approach typically involves the initial C-H halogenation of the indole core, often requiring stoichiometric amounts of halogenating agents and generating significant quantities of acidic waste. Following halogenation, a classical cross-coupling reaction, such as the Sonogashira coupling, is employed to introduce the alkyne moiety. This multi-step sequence not only suffers from lower overall atom economy due to the accumulation of byproducts at each stage but also struggles with regioselectivity issues. Since the electron cloud density at the C3-position of the indole ring is naturally higher than that at the C2-position, electrophilic substitution or metallization tends to occur preferentially at C3. Consequently, achieving exclusive C2-functionalization often necessitates the use of bulky protecting groups or specialized, expensive reagents, which drastically increases the complexity and cost of the manufacturing process.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108864164B utilizes a direct C-H activation strategy guided by a primary amine group, effectively bypassing the need for pre-halogenation. The core innovation lies in the use of 2-(1H-indol-1-yl)aniline derivatives as substrates, where the ortho-amino group acts as a powerful directing group to coordinate with the palladium catalyst. This coordination facilitates the selective activation of the proximal C2-H bond over the more reactive C3-H bond. Furthermore, the reaction system is remarkably green, utilizing water as the primary solvent instead of toxic organic solvents like DMF or NMP. This shift not only aligns with stringent environmental regulations but also simplifies the downstream purification process, as the product can be easily extracted into organic phases while inorganic salts remain in the aqueous layer. The combination of high regioselectivity, mild reaction conditions (80-110°C), and the use of commercially available starting materials makes this approach highly attractive for industrial scale-up.
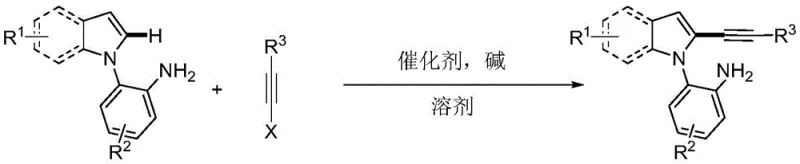
Mechanistic Insights into Pd-Catalyzed C-H Alkynylation
The success of this synthetic route hinges on a sophisticated catalytic cycle driven by palladium species. The mechanism initiates with the coordination of the palladium salt catalyst to the nitrogen atom of the primary amine directing group on the 2-(1H-indol-1-yl)aniline substrate. This coordination is the critical determinant of regioselectivity; it positions the metal center in close proximity to the C2-H bond, facilitating the formation of a stable six-membered palladacycle intermediate via C-H activation. This cyclometallation step effectively overrides the inherent electronic bias of the indole ring that would otherwise favor C3-reactivity. Once the palladacycle is formed, the alkyne halide undergoes oxidative addition to the palladium center. This step inserts the palladium into the carbon-halogen bond of the alkyne precursor, generating a high-valent palladium(IV) or palladium(II) species depending on the specific oxidation state dynamics. Finally, a reductive elimination step occurs, forging the new carbon-carbon bond between the indole C2-position and the alkyne group, thereby releasing the desired 2-alkynyl indole product and regenerating the active palladium catalyst for the next turnover.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or non-directed pathways. Because the C-H activation is directed and intramolecular in nature during the cyclometallation step, the formation of regioisomers (such as C3-alkynylated byproducts) is thermodynamically and kinetically suppressed. Additionally, the use of cesium salts as bases helps to neutralize the hydrogen halide byproduct generated during the reaction without promoting side reactions like hydrolysis of the alkyne halide, which can be a common issue in aqueous media. The tolerance for various functional groups—such as fluoro, chloro, methoxy, and cyano substituents on the indole ring—further indicates that the catalytic cycle is robust against electronic perturbations, ensuring a clean impurity profile that is essential for pharmaceutical applications where strict limits on genotoxic impurities and isomers are enforced.
How to Synthesize 2-Alkynyl Indole Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot plant production. The process involves charging a reactor with the 2-(1H-indol-1-yl)aniline substrate, the alkyne halide (typically a triisopropylsilyl-protected bromoalkyne), a palladium catalyst such as palladium tetranitrate tetrafluoroborate, and a cesium base. The reaction is conducted in water or a water-toluene mixture at temperatures ranging from 80°C to 110°C. Detailed standardized operating procedures regarding specific molar ratios, stirring speeds, and workup protocols are critical for maximizing yield and purity. For the complete step-by-step technical guide, please refer to the section below.
- Charge a reactor with 2-(1H-indol-1-yl)aniline substrate, alkyne halide, palladium salt catalyst, cesium base, and water solvent.
- Stir the reaction mixture at elevated temperatures between 80°C and 110°C for 12 to 24 hours to ensure complete conversion.
- Cool the reaction, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond mere technical novelty. The shift towards a direct C-H functionalization route eliminates entire unit operations associated with traditional synthesis, such as halogenation and subsequent protection/deprotection steps. This reduction in synthetic steps directly correlates to a significant decrease in raw material consumption, solvent usage, and labor hours, driving down the overall manufacturing cost. Furthermore, the use of water as a solvent mitigates the risks and costs associated with handling and disposing of large volumes of flammable and toxic organic solvents, enhancing the safety profile of the facility and reducing insurance and compliance overheads.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of expensive pre-functionalized starting materials. By utilizing simple, commodity-grade 2-(1H-indol-1-yl)anilines and alkyne halides, the bill of materials is optimized. Additionally, the high atom economy of the cross-coupling reaction means that a greater proportion of the input mass ends up in the final product, minimizing waste disposal fees. The ability to use inexpensive palladium salts without the need for complex, proprietary ligands further reduces the catalyst cost, which is often a major expense in noble metal-catalyzed processes. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad substrate scope and the use of readily available reagents. The starting materials, such as o-iodoaniline and indole derivatives, are produced by multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a wide range of functional groups, allows for flexibility in sourcing different substituted anilines without needing to requalify the entire process. This flexibility ensures continuity of supply even if specific grades of raw materials face temporary market shortages, securing the production schedule for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the use of water as a solvent, which offers superior heat transfer properties compared to viscous organic solvents, allowing for better temperature control in large reactors. The mild reaction temperatures (80-110°C) reduce energy consumption and pressure requirements, making the process compatible with standard glass-lined or stainless steel reactors found in most multipurpose plants. Moreover, the green nature of the process simplifies environmental permitting and wastewater treatment, as the aqueous waste stream is easier to treat than mixed organic waste, ensuring long-term regulatory compliance and sustainability goals are met.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this primary amine-guided synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: Why is regioselectivity a challenge in indole alkynylation?
A: Indole compounds naturally possess higher electron cloud density at the C3-position compared to the C2-position, making C3 metallization and subsequent functionalization kinetically favored. Achieving high selectivity for the C2-position typically requires sophisticated directing groups or harsh conditions, which this patent addresses using a primary amine guide.
Q: What are the environmental benefits of using water as a solvent?
A: Utilizing water eliminates the need for volatile organic compounds (VOCs) like DMF or toluene, significantly reducing hazardous waste disposal costs and improving operator safety. It also simplifies the workup process, as the product can often be extracted directly without complex solvent switching.
Q: What types of substituents are tolerated in this synthesis?
A: The method demonstrates broad substrate universality, tolerating various substituents on the indole ring such as methyl, fluoro, chloro, methoxy, and cyano groups, as well as substitutions on the aniline moiety, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkynyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN108864164B can be seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of 2-alkynyl indole intermediate meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cost-effective and green synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this methodology can optimize your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with high-purity, sustainably manufactured intermediates.
