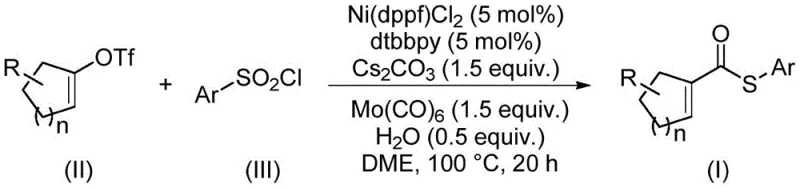
Advanced Nickel-Catalyzed Synthesis of Alpha,Beta-Unsaturated Thioesters for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex molecular architectures, particularly those involving sulfur-containing motifs. Patent CN114773242A introduces a groundbreaking preparation method for alpha,beta-unsaturated thioester compounds, addressing critical limitations in existing synthetic routes. These compounds are pivotal intermediates in organic synthesis, serving as key precursors for Diels-Alder reactions, conjugate additions, and cascade cyclizations found in various bioactive natural products. The disclosed technology leverages a nickel-catalyzed thiocarbonylation strategy that replaces hazardous thiols with stable arylsulfonyl chlorides, marking a significant shift towards safer and more sustainable chemical manufacturing processes for high-value intermediates.
Historically, the synthesis of alpha,beta-unsaturated thioesters has been plagued by significant operational and economic challenges that hinder large-scale adoption. Conventional approaches predominantly rely on condensation reactions or transition metal-catalyzed thiocarbonylation using precious metals like rhodium, platinum, or palladium. While effective, these noble metal catalysts are prohibitively expensive and subject to volatile market pricing, creating supply chain instability for procurement managers. Furthermore, traditional thiocarbonylation methods necessitate the use of thiols as sulfur sources, which are notorious for their repulsive odors and tendency to poison catalysts, leading to reduced turnover numbers and increased waste generation. These factors collectively inflate the cost of goods sold and complicate environmental compliance in industrial settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on precious metal catalysts such as palladium and rhodium creates a substantial bottleneck in the cost reduction in pharmaceutical intermediates manufacturing. These metals not only carry a high initial capital cost but also require rigorous removal steps to meet stringent residual metal specifications mandated by regulatory bodies for API production. Additionally, the use of thiols introduces severe health and safety risks, requiring specialized containment equipment and scrubbing systems to manage emissions. The susceptibility of these catalysts to poisoning by sulfur species further diminishes reaction efficiency, often necessitating higher catalyst loadings or extended reaction times, which negatively impacts throughput and energy consumption in commercial reactors.
The Novel Approach
The innovative method described in the patent circumvents these issues by utilizing an earth-abundant nickel catalyst system paired with arylsulfonyl chlorides. This strategic substitution eliminates the need for malodorous thiols, thereby simplifying facility requirements and enhancing worker safety. By employing molybdenum hexacarbonyl as a dual-purpose reagent acting as both the carbonyl source and reducing agent, the process achieves high atom economy. The reaction operates under relatively mild conditions at 100°C in ethylene glycol dimethyl ether, demonstrating exceptional functional group tolerance. This allows for the synthesis of diverse derivatives without extensive protecting group strategies, streamlining the overall synthetic route for complex molecules.
Mechanistic Insights into Nickel-Catalyzed Thiocarbonylation
The core of this technological advancement lies in the intricate catalytic cycle driven by the nickel complex. The reaction initiates with the reduction of the nickel(II) precatalyst, likely facilitated by the molybdenum species, to generate the active nickel(0) species. This low-valent nickel center undergoes oxidative addition with the arylsulfonyl chloride, cleaving the sulfur-chlorine bond to form a nickel-sulfur intermediate. Subsequently, carbon monoxide is released from the molybdenum hexacarbonyl and inserts into the nickel-sulfur bond, forming an acyl-nickel species. The final step involves the reaction of this acyl intermediate with the alkenyl trifluoromethanesulfonate, followed by reductive elimination to release the desired alpha,beta-unsaturated thioester product and regenerate the catalyst. This mechanism avoids the formation of toxic nickel tetracarbonyl, ensuring a safer reaction profile.
From an impurity control perspective, the choice of arylsulfonyl chlorides as electrophiles offers distinct advantages over traditional nucleophilic sulfur sources. The reaction pathway minimizes the formation of disulfide byproducts, which are common contaminants when using thiols. The use of cesium carbonate as a base ensures efficient neutralization of acidic byproducts without promoting unwanted side reactions such as hydrolysis of the sensitive thioester linkage. The broad substrate scope, accommodating electron-donating and electron-withdrawing groups on the aromatic ring, indicates a robust catalytic system that maintains activity across diverse electronic environments. This reliability is crucial for R&D directors aiming to develop scalable processes for varied analog libraries.
How to Synthesize Alpha,Beta-Unsaturated Thioesters Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The procedure involves charging a sealed vessel with the nickel catalyst, ligand, molybdenum source, base, and substrates in a single pot. The simplicity of the operation reduces the risk of human error and facilitates automation. For detailed procedural specifics regarding stoichiometry and workup, refer to the standardized guide below which outlines the precise execution parameters validated in the patent examples.
- Combine nickel catalyst (Ni(dppf)Cl2), ligand (dtbbpy), molybdenum hexacarbonyl, cesium carbonate, water, alkenyl trifluoromethanesulfonate, and arylsulfonyl chloride in a sealed tube.
- Add ethylene glycol dimethyl ether (DME) as the solvent to ensure proper dissolution of all reactants.
- Heat the reaction mixture to 100°C and stir for 20 hours, followed by filtration, silica gel mixing, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, this technology represents a paradigm shift towards more resilient and cost-efficient sourcing strategies. The substitution of expensive precious metals with nickel drastically lowers the raw material cost baseline, providing a buffer against market fluctuations in palladium and rhodium prices. Moreover, the use of commercially available and stable starting materials like arylsulfonyl chlorides ensures a consistent supply chain, reducing the risk of production delays caused by specialty reagent shortages. The simplified post-treatment process, involving basic filtration and chromatography, reduces solvent consumption and waste disposal costs, contributing to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant cost driver from the bill of materials. Nickel is abundant and inexpensive compared to palladium or rhodium, leading to substantial cost savings per kilogram of product. Additionally, the dual role of molybdenum hexacarbonyl reduces the total number of reagents required, further optimizing the material cost structure. The avoidance of specialized thiol handling infrastructure also translates into lower capital expenditure for facility upgrades.
- Enhanced Supply Chain Reliability: The starting materials, including alkenyl trifluoromethanesulfonates and arylsulfonyl chlorides, are widely available from global chemical suppliers. This ubiquity ensures that production schedules are not held hostage by the lead times of exotic reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing batch-to-batch consistency and reducing the rate of rejected lots.
- Scalability and Environmental Compliance: The reaction operates in a common solvent, ethylene glycol dimethyl ether, which is manageable in large-scale reactors. The absence of noxious thiol vapors simplifies废气 treatment systems and improves the overall environmental profile of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the process attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this nickel-catalyzed methodology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption in their own pipelines.
Q: Why is this nickel-catalyzed method superior to traditional thiol-based synthesis?
A: Traditional methods often rely on unpleasant-smelling thiols which can poison catalysts and pose safety hazards. This novel approach utilizes arylsulfonyl chlorides as a stable, odorless sulfur source, significantly improving operational safety and catalyst longevity.
Q: What serves as the carbonyl source in this reaction mechanism?
A: Molybdenum hexacarbonyl (Mo(CO)6) acts as a dual-function reagent, serving as both the carbonyl source for the thioester formation and as a reducing agent to regenerate the active nickel(0) species during the catalytic cycle.
Q: Does this process support a wide range of functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating substituents such as alkyl, methoxy, trifluoromethoxy, and halogens on the aryl ring, making it highly versatile for complex intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology for the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha,beta-unsaturated thioester delivered meets the highest industry standards for downstream application.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global market.
