Advanced Rhodium-Catalyzed Synthesis of Benzocycloheptenones for Commercial Pharmaceutical Manufacturing
Introduction to Novel Benzocycloheptenone Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to construct complex polycyclic scaffolds, particularly seven-membered rings which are prevalent in bioactive natural products and drug candidates. A significant breakthrough in this domain is detailed in Chinese Patent CN113620795B, which discloses a robust transition metal-catalyzed method for synthesizing benzocycloheptenone compounds. This technology leverages a rhodium-catalyzed C-H activation strategy coupled with the unique reactivity of cyclopropenones to forge the seven-membered ring in a single operational step. For R&D directors and process chemists, this represents a paradigm shift from laborious multi-step constructions to a streamlined annulation protocol. The ability to generate these valuable cores from simple 2-biphenylboronic acids and disubstituted cyclopropenones under mild conditions addresses critical bottlenecks in lead optimization and process development. As a reliable pharmaceutical intermediate supplier, understanding such patented methodologies is essential for evaluating the feasibility of scaling these high-value structures for commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of benzocycloheptenone frameworks has been fraught with synthetic challenges that hinder efficient manufacturing. Classical approaches often rely on intramolecular Friedel-Crafts acylations or ring-expansion reactions that require harsh acidic conditions, high temperatures, and specialized precursors that are difficult to source commercially. These conventional pathways frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are arduous to separate, thereby drastically reducing the overall yield and purity of the final API intermediate. Furthermore, many existing methods necessitate the use of stoichiometric amounts of toxic heavy metals or explosive diazo compounds, posing significant safety and environmental hazards in a production setting. The lack of functional group tolerance in older methodologies often requires extensive protecting group strategies, adding unnecessary steps, time, and cost to the synthesis. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, making the supply chain vulnerable to disruptions.
The Novel Approach
In stark contrast, the methodology described in patent CN113620795B offers a transformative solution by utilizing a rhodium(III)-catalyzed oxidative annulation. This novel approach employs 2-biphenylboronic acid derivatives and disubstituted cyclopropenones as building blocks, which undergo a seamless transformation to yield the target benzocycloheptenones with high efficiency. 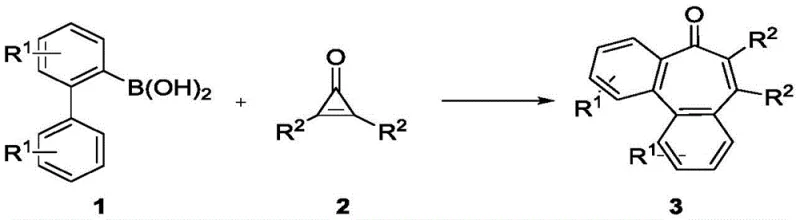 The reaction operates under remarkably mild conditions, typically at 60°C in common solvents like ethyl acetate, and crucially, it proceeds under an air atmosphere without the need for inert gas protection. This simplicity significantly lowers the barrier for implementation in standard chemical manufacturing facilities. The strategic use of cyclopropenones allows for the incorporation of diverse substituents at the 2 and 3 positions of the seven-membered ring, providing unparalleled flexibility for medicinal chemists to explore structure-activity relationships. This one-step protocol not only simplifies the synthetic route but also enhances the overall atom economy, making it a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
The reaction operates under remarkably mild conditions, typically at 60°C in common solvents like ethyl acetate, and crucially, it proceeds under an air atmosphere without the need for inert gas protection. This simplicity significantly lowers the barrier for implementation in standard chemical manufacturing facilities. The strategic use of cyclopropenones allows for the incorporation of diverse substituents at the 2 and 3 positions of the seven-membered ring, providing unparalleled flexibility for medicinal chemists to explore structure-activity relationships. This one-step protocol not only simplifies the synthetic route but also enhances the overall atom economy, making it a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Ring Expansion
Understanding the underlying mechanism is vital for R&D teams aiming to optimize this process for specific substrates. The reaction is initiated by the active cationic Cp*Rh(III) species, which facilitates the directed C-H activation of the 2-biphenylboronic acid substrate. This step forms a stable five-membered rhodacycle intermediate, a critical juncture that dictates the regioselectivity of the subsequent transformation. Following C-H activation, the coordination and insertion of the cyclopropenone into the Rh-C bond occur, driven by the relief of ring strain inherent in the three-membered cyclopropenone system. 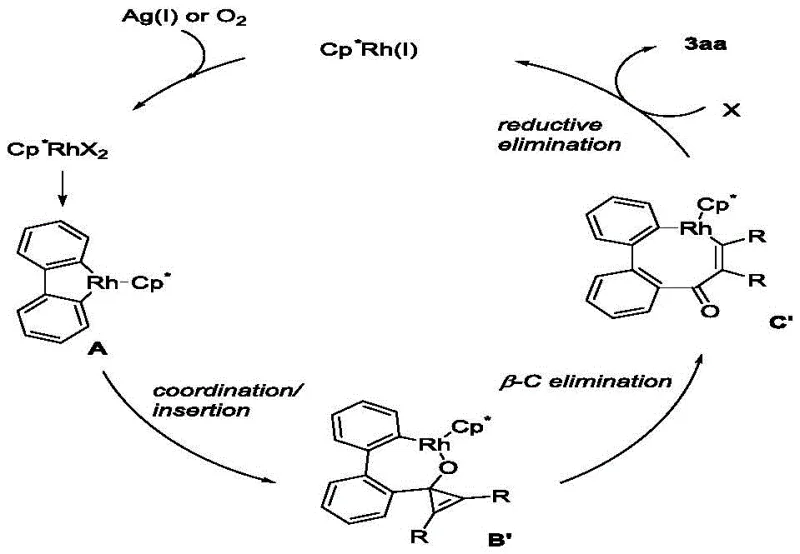 The most distinctive feature of this mechanism is the subsequent beta-carbon elimination step. Unlike typical insertion reactions that preserve the carbon skeleton, the strained cyclopropenone ring undergoes cleavage, effectively expanding the ring system from a transient intermediate to the final seven-membered benzocycloheptenone scaffold. This beta-C elimination is facilitated by the electronic properties of the carbonyl group and the steric environment created by the Cp* ligand. Finally, reductive elimination releases the product and regenerates the Rh(I) species, which is re-oxidized to Rh(III) by the silver salt oxidant to close the catalytic cycle. This mechanistic pathway ensures high selectivity and minimizes the formation of side products, resulting in a clean impurity profile that is highly desirable for regulatory compliance in drug substance production.
The most distinctive feature of this mechanism is the subsequent beta-carbon elimination step. Unlike typical insertion reactions that preserve the carbon skeleton, the strained cyclopropenone ring undergoes cleavage, effectively expanding the ring system from a transient intermediate to the final seven-membered benzocycloheptenone scaffold. This beta-C elimination is facilitated by the electronic properties of the carbonyl group and the steric environment created by the Cp* ligand. Finally, reductive elimination releases the product and regenerates the Rh(I) species, which is re-oxidized to Rh(III) by the silver salt oxidant to close the catalytic cycle. This mechanistic pathway ensures high selectivity and minimizes the formation of side products, resulting in a clean impurity profile that is highly desirable for regulatory compliance in drug substance production.
How to Synthesize Benzocycloheptenone Efficiently
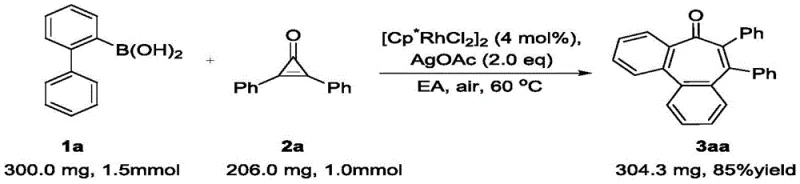
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale discovery and pilot-scale production. The general procedure involves mixing the 2-biphenylboronic acid and the disubstituted cyclopropenone in a molar ratio of approximately 1.2:1.0 to ensure complete consumption of the limiting reagent. The reaction is catalyzed by [Cp*RhCl2]2 at a loading of 4 mol%, with silver acetate (AgOAc) serving as the terminal oxidant in a 2.0 equivalent amount. Ethyl acetate is the preferred solvent due to its favorable safety profile and ability to dissolve the reactants effectively. 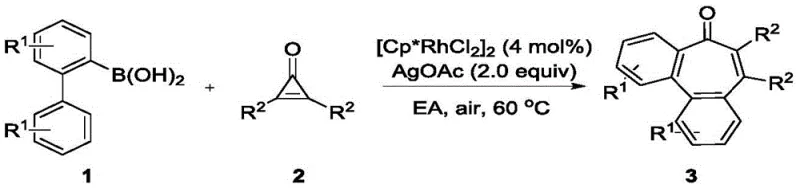 The mixture is heated to 60°C in a sealed tube under ambient air for approximately 12 hours. Upon completion, monitored by TLC, the workup is straightforward: the solvent is removed, and the crude product is purified via silica gel column chromatography using a petroleum ether/dichloromethane system. This standardized protocol has been validated across a wide range of substrates, consistently delivering high yields.
The mixture is heated to 60°C in a sealed tube under ambient air for approximately 12 hours. Upon completion, monitored by TLC, the workup is straightforward: the solvent is removed, and the crude product is purified via silica gel column chromatography using a petroleum ether/dichloromethane system. This standardized protocol has been validated across a wide range of substrates, consistently delivering high yields.
- Combine 2-biphenylboronic acid compound and disubstituted cyclopropenone compound in an organic solvent such as ethyl acetate.
- Add [Cp*RhCl2]2 catalyst (4 mol%) and silver salt oxidant (e.g., AgOAc, 2.0 equiv) to the reaction mixture.
- Heat the sealed reaction tube to 60°C under air atmosphere for 12 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that align with the strategic goals of procurement and supply chain management. The shift towards this Rh-catalyzed method addresses several pain points associated with traditional manufacturing, specifically regarding cost, reliability, and scalability. By simplifying the synthetic route to a single step, the process inherently reduces the consumption of solvents, reagents, and energy, leading to significant operational cost savings. Moreover, the use of commercially available starting materials mitigates the risk of supply chain bottlenecks often associated with exotic or custom-synthesized precursors.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and protecting group manipulations directly translates to lower production costs. The use of earth-abundant solvents like ethyl acetate and the avoidance of cryogenic conditions or high-pressure equipment further reduce capital and operational expenditures. While the rhodium catalyst is a precious metal, its low loading (4 mol%) and the potential for recycling or recovery in large-scale processes ensure that the overall catalyst cost remains manageable. The high isolated yields reported, often exceeding 80-90% for optimized substrates, minimize waste and maximize the throughput of valuable intermediates, enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: The robustness of this reaction under air atmosphere is a critical advantage for supply chain continuity. Unlike sensitive organometallic reactions that require rigorous exclusion of oxygen and moisture, this method can be performed in standard reactors without specialized inert gas lines or gloveboxes. This operational simplicity reduces the likelihood of batch failures due to environmental factors and allows for faster turnaround times. Additionally, the broad substrate scope means that a single manufacturing platform can be adapted to produce a diverse library of benzocycloheptenone derivatives, providing flexibility to respond to changing market demands or client specifications without retooling the entire production line.
- Scalability and Environmental Compliance: The patent data includes successful scale-up experiments, demonstrating that the reaction maintains its efficiency when moving from milligram to gram scales.
 The mild reaction temperature of 60°C is energy-efficient and safe for large-scale operations, reducing the thermal load on cooling systems. Furthermore, the generation of fewer by-products simplifies downstream purification and waste treatment, aiding in compliance with increasingly stringent environmental regulations. The atom-economic nature of the ring-expansion strategy ensures that a high proportion of the starting material mass is incorporated into the final product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process.
The mild reaction temperature of 60°C is energy-efficient and safe for large-scale operations, reducing the thermal load on cooling systems. Furthermore, the generation of fewer by-products simplifies downstream purification and waste treatment, aiding in compliance with increasingly stringent environmental regulations. The atom-economic nature of the ring-expansion strategy ensures that a high proportion of the starting material mass is incorporated into the final product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzocycloheptenone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners and licensees.
Q: What are the key advantages of this Rh-catalyzed method over traditional synthesis?
A: This method utilizes readily available 2-biphenylboronic acids and cyclopropenones under mild conditions (60°C, air atmosphere) to achieve high yields in a single step, avoiding complex multi-step sequences.
Q: Is the reaction sensitive to oxygen or moisture?
A: No, the reaction proceeds efficiently under air atmosphere without the need for inert gas protection or strictly anhydrous conditions, simplifying operational requirements.
Q: What is the substrate scope for this benzocycloheptenone synthesis?
A: The method tolerates a wide range of substituents including halogens (F, Cl, Br), trifluoromethyl groups, alkyl chains, and alkoxy groups on both the biphenyl and cyclopropenone substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocycloheptenone Supplier
The technological advancements presented in patent CN113620795B highlight the immense potential of rhodium-catalyzed C-H activation for constructing complex heterocyclic systems. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative synthetic routes in accelerating drug discovery and development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing the quality and consistency of every batch we produce.
We invite you to leverage our expertise to optimize your supply chain for benzocycloheptenone derivatives. Whether you require custom synthesis services or technology transfer support, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us collaborate to bring your next-generation therapeutics to market faster and more efficiently.
