Advanced Rhodium-Catalyzed Synthesis of Benzocycloheptenones for Commercial API Manufacturing
Advanced Rhodium-Catalyzed Synthesis of Benzocycloheptenones for Commercial API Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex polycyclic scaffolds efficiently. Patent CN113620795B introduces a groundbreaking transition metal-catalyzed approach for synthesizing benzocycloheptenone compounds, a privileged structural motif found in numerous bioactive natural products and drug candidates. This technology leverages a rhodium-catalyzed annulation strategy between 2-biphenylboronic acid derivatives and disubstituted cyclopropenones. Unlike traditional multi-step cyclizations that often suffer from harsh conditions and poor atom economy, this novel route operates under mild conditions with high selectivity. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline the supply chain for high-purity pharmaceutical intermediates. The process utilizes readily available starting materials and avoids the need for stringent inert gas protection, marking a substantial advancement in process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of seven-membered carbocyclic rings fused to aromatic systems has posed significant synthetic challenges. Conventional routes often rely on intramolecular Friedel-Crafts acylations or ring-expansion reactions that require stoichiometric amounts of Lewis acids and generate substantial waste. These traditional methods frequently necessitate low temperatures, strictly anhydrous conditions, and complex workup procedures to remove metal residues, which drastically increases the cost of goods sold (COGS). Furthermore, the functional group tolerance in older methodologies is often limited, restricting the diversity of analogues that can be generated for structure-activity relationship (SAR) studies. The inability to easily introduce halogens or electron-withdrawing groups without side reactions has been a persistent bottleneck in the development of novel drug candidates based on the benzocycloheptenone core.
The Novel Approach
The methodology disclosed in CN113620795B overcomes these hurdles through a clever design involving C-H activation and ring expansion. By utilizing 2-biphenylboronic acids and cyclopropenones, the reaction achieves a direct annulation that constructs the seven-membered ring in a single operational step. 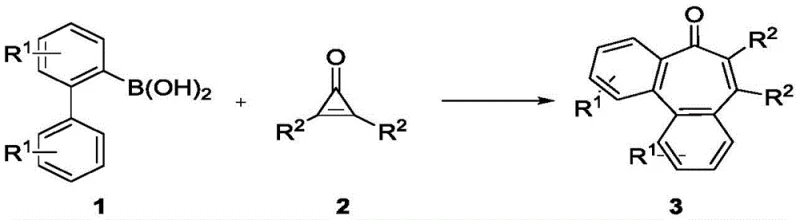 This transformation is driven by a rhodium catalyst system that facilitates the cleavage of the strained three-membered ring of the cyclopropenone, effectively using the ring strain as a driving force for the formation of the new carbon-carbon bonds. The result is a highly efficient process that delivers the target benzocycloheptenone compounds in high yields, often exceeding 90% for model substrates. This shift from multi-step sequences to a direct catalytic assembly not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with green chemistry principles essential for modern cost reduction in pharmaceutical intermediate manufacturing.
This transformation is driven by a rhodium catalyst system that facilitates the cleavage of the strained three-membered ring of the cyclopropenone, effectively using the ring strain as a driving force for the formation of the new carbon-carbon bonds. The result is a highly efficient process that delivers the target benzocycloheptenone compounds in high yields, often exceeding 90% for model substrates. This shift from multi-step sequences to a direct catalytic assembly not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with green chemistry principles essential for modern cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Ring Expansion
Understanding the catalytic cycle is crucial for scaling this technology effectively. The reaction initiates with the active Rh(III) species coordinating to the biphenyl substrate, followed by a directed C-H activation to form a five-membered rhodacycle intermediate. This step is critical as it sets the regioselectivity for the subsequent bond formation. The cyclopropenone then coordinates to the metal center and undergoes migratory insertion into the Rh-C bond. 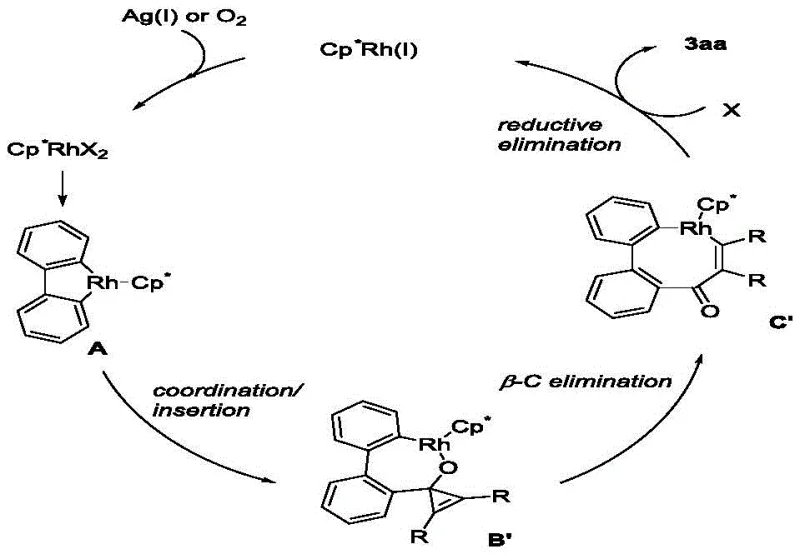 A key feature of this mechanism is the subsequent beta-carbon elimination, which opens the strained cyclopropenone ring. This ring-opening event is thermodynamically favorable and leads to the formation of a seven-membered rhodacycle. Finally, reductive elimination releases the product and regenerates the active catalyst, facilitated by the silver oxidant which re-oxidizes Rh(I) back to Rh(III). This elegant cycle ensures high turnover numbers and minimizes the accumulation of inactive catalyst species, which is vital for maintaining consistent quality in large-scale batches. The tolerance for air suggests that the oxidative regeneration step is robust, preventing catalyst deactivation by trace oxygen, a common issue in other transition metal-catalyzed processes.
A key feature of this mechanism is the subsequent beta-carbon elimination, which opens the strained cyclopropenone ring. This ring-opening event is thermodynamically favorable and leads to the formation of a seven-membered rhodacycle. Finally, reductive elimination releases the product and regenerates the active catalyst, facilitated by the silver oxidant which re-oxidizes Rh(I) back to Rh(III). This elegant cycle ensures high turnover numbers and minimizes the accumulation of inactive catalyst species, which is vital for maintaining consistent quality in large-scale batches. The tolerance for air suggests that the oxidative regeneration step is robust, preventing catalyst deactivation by trace oxygen, a common issue in other transition metal-catalyzed processes.
From an impurity control perspective, the mechanism offers distinct advantages. The high regioselectivity of the C-H activation step minimizes the formation of positional isomers, which are often difficult to separate. Additionally, the use of silver acetate as a terminal oxidant produces benign by-products that are easily removed during aqueous workup. The patent data indicates that even with diverse substituents such as halogens and trifluoromethyl groups, the reaction maintains high fidelity, producing clean crude profiles that simplify downstream purification. This predictability is essential for ensuring the stringent purity specifications required for API intermediates, reducing the risk of batch failures due to unexpected impurity profiles.
How to Synthesize Benzocycloheptenone Efficiently
The practical implementation of this synthesis is straightforward, making it accessible for both laboratory discovery and pilot plant operations. The general procedure involves mixing the 2-biphenylboronic acid derivative and the disubstituted cyclopropenone in a molar ratio of approximately 1.2:1.0 to ensure complete consumption of the more valuable cyclopropenone partner. The catalyst loading is kept low at 4 mol% of [Cp*RhCl2]2, which is economically favorable for commercial applications. 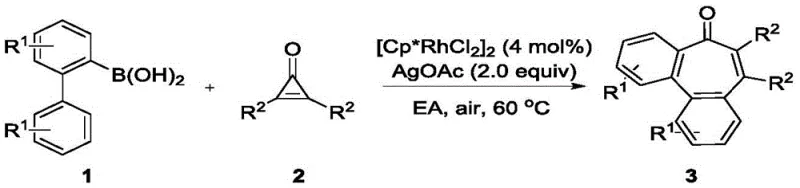 The reaction is conducted in ethyl acetate, a greener solvent alternative to chlorinated solvents often used in similar transformations. Heating the mixture to 60°C under an air atmosphere for 12 hours is sufficient to drive the reaction to completion, as monitored by TLC. The detailed standardized synthesis steps are provided in the guide below.
The reaction is conducted in ethyl acetate, a greener solvent alternative to chlorinated solvents often used in similar transformations. Heating the mixture to 60°C under an air atmosphere for 12 hours is sufficient to drive the reaction to completion, as monitored by TLC. The detailed standardized synthesis steps are provided in the guide below.
- Combine 2-biphenylboronic acid derivative and disubstituted cyclopropenone in a reaction vessel with [Cp*RhCl2]2 catalyst.
- Add silver acetate oxidant and ethyl acetate solvent under air atmosphere.
- Heat the mixture to 60°C for 12 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond just chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing process. By eliminating the need for inert gas lines and glovebox techniques, the capital expenditure required for setting up production lines is significantly reduced. The ability to run the reaction in air lowers the barrier to entry for contract manufacturing organizations (CMOs) and allows for greater flexibility in scheduling production campaigns. This operational simplicity translates directly into reduced lead times for high-purity pharmaceutical intermediates, enabling faster response to market demands.
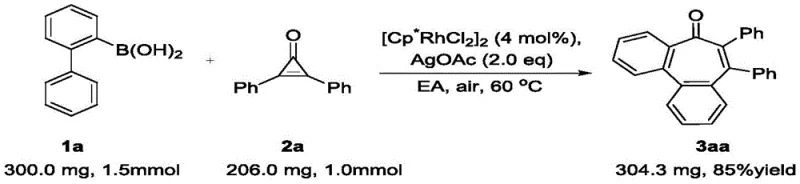
- Cost Reduction in Manufacturing: The economic impact of this method is profound. The use of earth-abundant solvents like ethyl acetate instead of expensive or regulated solvents lowers raw material costs. Furthermore, the high yields reported, such as the 90% isolated yield for the model compound 3aa, mean that less starting material is wasted per kilogram of product. The low catalyst loading of 4 mol% combined with the potential for catalyst recovery further drives down the cost per kilogram. Eliminating complex purification steps associated with traditional methods reduces labor and utility costs, resulting in substantial cost savings for the final API production.
- Enhanced Supply Chain Reliability: The starting materials, 2-biphenylboronic acids and cyclopropenones, are commercially available or easily synthesized from commodity chemicals. This ensures a stable supply base and mitigates the risk of raw material shortages. The broad substrate scope demonstrated in the patent, covering various halogenated and alkyl-substituted derivatives, means that a single platform technology can be used to produce a wide library of intermediates. This versatility allows supply chain managers to consolidate suppliers and reduce the complexity of the vendor network, enhancing overall supply chain resilience against disruptions.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this process with a successful scale-up experiment (Example 34), proving its viability for commercial production.
 The mild reaction conditions and the absence of toxic heavy metal waste streams simplify waste treatment and disposal, ensuring compliance with increasingly strict environmental regulations. The use of air as an oxidant source eliminates the need for storing and handling hazardous oxidizing agents, improving workplace safety. These factors collectively make the commercial scale-up of complex pharmaceutical intermediates safer, cleaner, and more sustainable.
The mild reaction conditions and the absence of toxic heavy metal waste streams simplify waste treatment and disposal, ensuring compliance with increasingly strict environmental regulations. The use of air as an oxidant source eliminates the need for storing and handling hazardous oxidizing agents, improving workplace safety. These factors collectively make the commercial scale-up of complex pharmaceutical intermediates safer, cleaner, and more sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a reliable basis for process evaluation.
Q: What are the optimal reaction conditions for synthesizing benzocycloheptenones?
A: The patent specifies using [Cp*RhCl2]2 (4 mol%) as the catalyst, AgOAc (2.0 equiv) as the oxidant, and ethyl acetate as the solvent at 60°C under air.
Q: Is inert gas protection required for this synthesis?
A: No, the reaction proceeds efficiently under air atmosphere, which significantly simplifies operational requirements compared to traditional inert gas protocols.
Q: What is the substrate scope for this reaction?
A: The method tolerates a wide range of substituents including halogens (F, Cl, Br), alkyl groups, trifluoromethyl, and alkoxy groups on both the biphenyl and cyclopropenone moieties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocycloheptenone Supplier
The technological breakthroughs detailed in CN113620795B represent a significant leap forward in the synthesis of complex polycyclic structures. At NINGBO INNO PHARMCHEM, we recognize the potential of this chemistry to transform the production landscape for oncology and antiviral drug candidates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle transition metal catalysis with the highest safety standards, and our rigorous QC labs ensure that every batch meets stringent purity specifications required by global regulatory bodies.
We invite you to leverage our expertise to accelerate your drug development timeline. Whether you need custom synthesis of specific benzocycloheptenone derivatives or process optimization for existing routes, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your project. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how this innovative method can enhance your supply chain efficiency and reduce overall manufacturing costs.
