Advanced Cobalt-Catalyzed Synthesis Pathway Enables Scalable Production of High-Purity Tetrahydro-Beta-Carbolinone for Pharmaceutical Applications
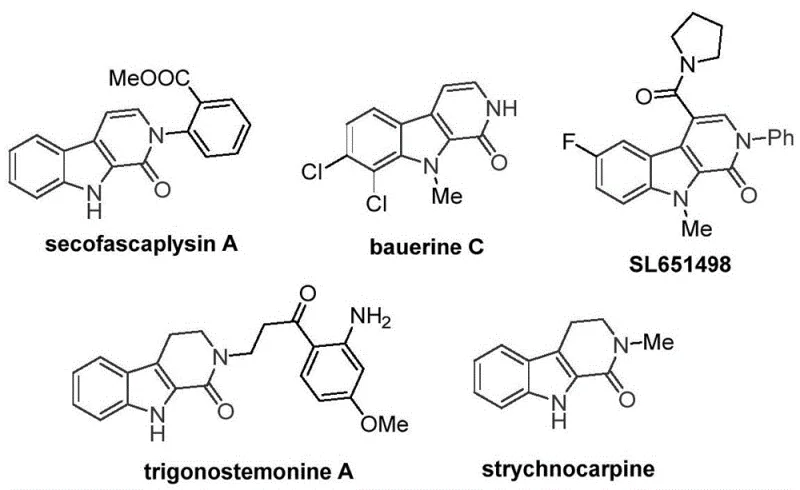
The patent CN115260188A introduces a groundbreaking cobalt-catalyzed methodology for synthesizing tetrahydro-beta-carbolinone compounds—a critical structural motif found in numerous bioactive pharmaceutical agents including the antiviral natural product bauerine C and the anxiolytic candidate SL651498. This innovation addresses a significant gap in current synthetic approaches by eliminating reliance on expensive palladium catalysts while achieving superior substrate compatibility across diverse functional groups. The process operates under practical conditions using commercially accessible reagents and standard laboratory equipment, making it immediately applicable for industrial-scale production of high-value intermediates. Crucially, the methodology maintains exceptional selectivity without generating problematic metal residues that typically complicate pharmaceutical manufacturing workflows. This represents a paradigm shift in accessing beta-carbolinone scaffolds that are essential building blocks for next-generation therapeutic agents targeting neurological disorders and viral infections.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to beta-carbolinone frameworks have been severely constrained by their dependence on palladium-based catalytic systems that impose multiple operational limitations including stringent oxygen-free environments and complex catalyst recovery protocols. These methods typically require expensive ligand systems and generate significant transition metal residues that necessitate extensive purification steps involving specialized scavenging agents and additional chromatographic separations—processes that substantially increase both time-to-market and production costs while introducing potential contamination risks for pharmaceutical applications. Furthermore, conventional approaches exhibit narrow substrate scope with poor tolerance for common functional groups like halogens and alkoxy moieties, severely limiting their utility for synthesizing diverse analogs required in drug discovery pipelines. The requirement for high-pressure carbon monoxide handling also presents significant safety hazards and infrastructure demands that are incompatible with standard manufacturing facilities.
The Novel Approach
The patented methodology overcomes these critical limitations through an innovative cobalt-catalyzed carbonylation process that operates under ambient pressure conditions using a safe carbon monoxide surrogate. By employing readily available cobalt acetate tetrahydrate as catalyst with pivalic acid as additive and triethylamine as base in dioxane solvent at moderate temperatures of 130°C, the process achieves exceptional functional group tolerance across halogenated, alkylated, and alkoxy-substituted substrates while maintaining consistent high yields. The elimination of palladium catalysts removes both cost barriers and metal contamination concerns that have historically plagued beta-carbolinone synthesis. Crucially, the reaction design incorporates a self-cleaving pyridyl group that simplifies purification by avoiding persistent protecting groups while enabling straightforward isolation of high-purity products through standard column chromatography techniques without requiring specialized metal removal steps.
Mechanistic Insights into Cobalt-Catalyzed Carbonylation
The reaction proceeds through a well-defined catalytic cycle initiated by oxidation of cobalt(II) to cobalt(III) by silver carbonate oxidant, followed by coordination with the tryptamine derivative substrate to form a key intermediate complex. Subsequent C-H bond activation at the indole position generates a cyclometalated species that undergoes migratory insertion with carbon monoxide released from the TFBen surrogate to form an acylcobalt intermediate. This critical step enables the carbonyl group incorporation that constructs the beta-carbolinone core structure through intramolecular cyclization. The final reductive elimination step releases the product while regenerating the active cobalt catalyst species. This mechanism operates with remarkable efficiency due to the synergistic effects of pivalic acid as a proton shuttle and triethylamine as a base that maintains optimal reaction kinetics throughout the transformation.
Impurity control is achieved through multiple built-in safeguards within this mechanism; the selective C-H activation step prevents undesired side reactions at alternative positions while the controlled release of carbon monoxide from TFBen avoids concentration spikes that could lead to over-carbonylation byproducts. The self-cleaving nature of the pyridyl directing group eliminates potential contamination from persistent protecting groups that commonly complicate traditional syntheses. Furthermore, the moderate reaction temperature of 130°C prevents thermal decomposition pathways that typically generate impurities in high-energy processes. The optimized molar ratios ensure complete consumption of starting materials while minimizing dimerization or oligomerization side products that could otherwise compromise final product purity.

How to Synthesize Tetrahydro-Beta-Carbolinone Efficiently
This patented synthesis pathway represents a significant advancement over conventional methods by providing a streamlined route to high-value beta-carbolinone intermediates through carefully optimized reaction parameters that balance efficiency with practical manufacturability. The process eliminates multiple pain points associated with traditional approaches including hazardous reagent handling requirements and complex purification protocols while maintaining exceptional substrate scope across diverse functionalized precursors. Detailed standardized operating procedures have been developed based on extensive experimental validation across multiple substrate classes as documented in the patent literature. The following section provides essential implementation guidance for R&D teams seeking to adopt this methodology in their production workflows.
- Combine tryptamine derivative substrate with cobalt acetate tetrahydrate catalyst, pivalic acid additive, triethylamine base, silver carbonate oxidant, and TFBen carbon monoxide substitute in dioxane solvent at precise molar ratios
- Heat the reaction mixture to controlled temperature of 130°C under inert atmosphere for optimized duration of 20 hours to ensure complete conversion while maintaining thermal stability
- Execute post-reaction processing through filtration followed by silica gel column chromatography purification to isolate high-purity tetrahydro-beta-carbolinone product with minimal residual impurities
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by delivering a more resilient supply chain solution through fundamentally improved process economics and operational reliability. The elimination of palladium catalysts removes both cost volatility associated with precious metal markets and supply chain vulnerabilities stemming from concentrated geographic sources of these materials. By utilizing universally available reagents and standard manufacturing equipment requirements, the process enables greater sourcing flexibility while reducing dependency on specialized suppliers that often create bottlenecks in traditional production pathways.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with economical cobalt-based systems creates substantial cost savings through reduced raw material expenditures while eliminating downstream metal removal processing steps that typically require additional equipment investment and specialized consumables. The use of standard laboratory glassware instead of high-pressure reactors further reduces capital requirements while maintaining consistent product quality across different production scales.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including commercially sourced tryptamine derivatives and common solvents ensures consistent supply availability without exposure to single-source dependencies that frequently disrupt traditional manufacturing routes. This robust material sourcing profile enables reliable production scheduling with minimal risk of raw material shortages or price fluctuations that could impact delivery commitments.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory validation to multi-ton production volumes due to its compatibility with standard manufacturing equipment and absence of hazardous reagents requiring special handling protocols. The simplified waste stream profile resulting from eliminated metal scavenging steps significantly reduces environmental impact while facilitating regulatory compliance across global manufacturing sites without requiring specialized waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement specialists and R&D managers during initial evaluations of this patented technology platform. These responses are derived directly from experimental data presented in patent CN115260188A and reflect practical implementation insights gained through extensive process validation studies across multiple substrate classes under various production scenarios.
Q: Why is this cobalt-catalyzed method superior to traditional palladium-based approaches?
A: The cobalt system eliminates expensive palladium catalysts while maintaining high substrate tolerance and reaction efficiency. Unlike palladium methods requiring specialized handling and costly metal removal steps, this approach uses readily available cobalt acetate tetrahydrate with simplified purification protocols that significantly reduce operational complexity and associated costs in pharmaceutical intermediate manufacturing.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The reaction design inherently minimizes impurity formation through selective C-H activation and controlled carbonylation conditions. The self-cleaving pyridyl group prevents side reactions while the optimized molar ratios and temperature control suppress byproduct generation. Subsequent column chromatography purification consistently delivers compounds meeting stringent pharmaceutical purity specifications without requiring additional metal scavenging steps.
Q: What scalability advantages does this synthesis offer for commercial production?
A: The process demonstrates exceptional scalability from laboratory to industrial scale due to its robust reaction profile using standard equipment. The absence of sensitive reagents and compatibility with conventional reactors enables seamless transition from gram-scale validation to multi-kilogram production runs while maintaining consistent yield and quality parameters across different batch sizes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-Beta-Carbolinone Supplier
Our company leverages this patented cobalt-catalyzed methodology to deliver exceptional value through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications. Our dedicated R&D teams work closely with clients to optimize reaction parameters for specific substrate requirements while ensuring seamless technology transfer through rigorous QC labs that provide comprehensive analytical validation at every production stage. This commitment to technical excellence positions us as the preferred partner for manufacturers seeking reliable access to high-purity beta-carbolinone intermediates with consistent quality and supply continuity.
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team who will provide detailed COA data and route feasibility assessments demonstrating how this innovative synthesis can enhance your manufacturing economics while ensuring supply chain resilience.
