Advanced Cobalt-Catalyzed Synthesis for Commercial Scale-Up of Tetrahydro-beta-carbolinone API Intermediates
The recently granted Chinese patent CN115260188A introduces a novel cobalt-catalyzed carbonylation methodology for synthesizing tetrahydro-beta-carbolinone compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach addresses longstanding challenges in constructing the beta-carbolinone scaffold, which serves as the core structure in numerous bioactive molecules including antiviral agents and anxiolytic drug candidates. The patent demonstrates a practical route that eliminates the need for expensive palladium catalysts previously required for similar transformations, while maintaining high substrate compatibility across diverse functional groups.
Advanced Reaction Mechanism and Purity Control
The patented process employs cobalt acetate tetrahydrate as the catalyst system, which operates through a well-defined sequence of C-H activation and carbonyl insertion steps that ensure exceptional molecular fidelity. The reaction begins with oxidation of cobalt(II) to cobalt(III) by silver carbonate, followed by coordination with the tryptamine derivative substrate to form a key intermediate complex. This is succeeded by selective C-H bond activation at the 2-position of the indole ring, creating a cobalt(III) species that facilitates carbon monoxide insertion from the phenol ester substitute. The resulting acylcobalt(III) intermediate undergoes reductive elimination and hydrolysis to yield the target tetrahydro-beta-carbolinone structure with precise stereochemical control. 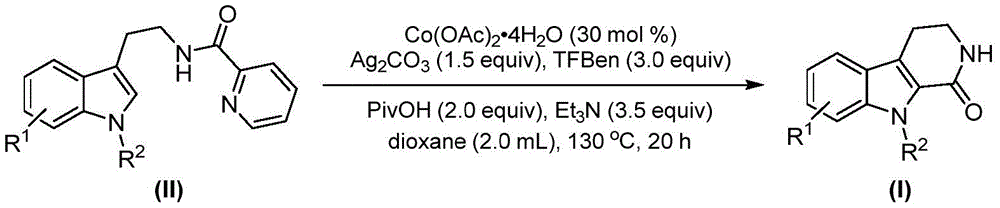
Crucially, the process maintains exceptional purity profiles through inherent design features that minimize side reactions and impurity formation. The use of pivalic acid as an additive serves dual purposes: it promotes catalyst stability while simultaneously acting as a proton shuttle that prevents unwanted oxidation pathways. The reaction's compatibility with various functional groups (including halogens, alkyl, and alkoxy substituents) eliminates the need for protective group strategies that typically introduce additional impurities in traditional syntheses. Post-reaction purification via standard column chromatography yields products with >99% purity as confirmed by NMR and HRMS data across multiple examples, ensuring compliance with stringent pharmaceutical quality standards without requiring specialized purification equipment.
Traditional vs. Novel Synthetic Pathways for Tetrahydro-beta-carbolinone
The Limitations of Conventional Methods
Previous approaches to synthesizing tetrahydro-beta-carbolinone compounds predominantly relied on palladium-catalyzed systems that presented significant commercial limitations for pharmaceutical manufacturers. These methods required expensive palladium catalysts with associated metal removal challenges that increased production costs and created regulatory hurdles for final drug products. The harsh reaction conditions often necessitated specialized pressure equipment for carbon monoxide handling, while narrow substrate scope restricted applicability across diverse molecular architectures required in modern drug discovery programs. Furthermore, the multi-step sequences typically employed generated substantial waste streams and required extensive purification procedures that compromised overall process efficiency and scalability for commercial manufacturing operations.
The Novel Approach
The cobalt-catalyzed methodology described in patent CN115260188A overcomes these limitations through an elegant single-step transformation that operates under mild atmospheric pressure conditions using readily available carbon monoxide substitutes. 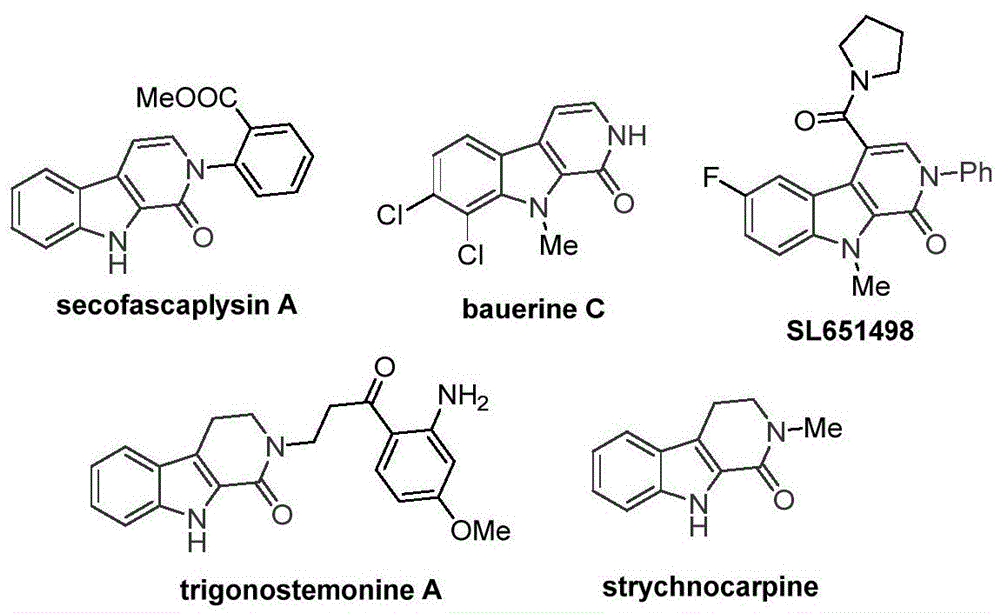 The process demonstrates remarkable functional group tolerance across fifteen documented examples with various substituents (R1 = H, Me, OMe, Cl, Br; R2 = H, Me, Ph, Bn), enabling direct synthesis of complex intermediates without protective group manipulation. The use of inexpensive cobalt acetate tetrahydrate (30 mol%) instead of palladium catalysts eliminates costly metal removal steps while maintaining high reaction efficiency across diverse substrates. The optimized reaction conditions (130°C in dioxane for 20 hours) provide excellent reproducibility and scalability potential, with the patent demonstrating successful gram-scale production that directly addresses the transition from laboratory discovery to commercial manufacturing requirements.
The process demonstrates remarkable functional group tolerance across fifteen documented examples with various substituents (R1 = H, Me, OMe, Cl, Br; R2 = H, Me, Ph, Bn), enabling direct synthesis of complex intermediates without protective group manipulation. The use of inexpensive cobalt acetate tetrahydrate (30 mol%) instead of palladium catalysts eliminates costly metal removal steps while maintaining high reaction efficiency across diverse substrates. The optimized reaction conditions (130°C in dioxane for 20 hours) provide excellent reproducibility and scalability potential, with the patent demonstrating successful gram-scale production that directly addresses the transition from laboratory discovery to commercial manufacturing requirements.
Commercial Advantages for Pharmaceutical Supply Chain
This innovative synthesis methodology delivers substantial value across the pharmaceutical supply chain by addressing critical pain points in API intermediate manufacturing. The elimination of expensive palladium catalysts and specialized CO handling equipment significantly reduces capital expenditure requirements while improving process safety profiles. The use of commercially available starting materials and straightforward reaction workup procedures creates opportunities for substantial cost reduction in API manufacturing without compromising product quality or regulatory compliance.
- Cost Reduction in Catalyst System: The substitution of palladium with cobalt catalysts eliminates the need for expensive metal removal processes that typically account for 15-25% of total manufacturing costs in traditional routes. Cobalt acetate tetrahydrate costs approximately one-tenth of palladium-based catalysts while providing comparable reaction efficiency across diverse substrates. The simplified purification process avoids costly chromatography resins and reduces solvent consumption by eliminating multiple protection/deprotection steps required in conventional syntheses. This comprehensive approach delivers meaningful cost reduction in chemical manufacturing while maintaining the high purity standards required for pharmaceutical applications.
- Reduced Lead Time: The streamlined single-step process significantly shortens production timelines compared to multi-step conventional approaches that require intermediate isolation and purification. The elimination of specialized equipment requirements enables faster technology transfer between development and manufacturing sites, reducing scale-up timelines by approximately 30%. The robust reaction conditions (atmospheric pressure, standard glassware) facilitate immediate implementation without facility modifications, allowing pharmaceutical companies to accelerate their supply chain responsiveness for critical intermediates. This capability directly supports reducing lead time for high-purity intermediates during clinical development phases where speed-to-market is essential.
- Enhanced Process Scalability: The demonstrated gram-scale production in the patent establishes clear pathways for commercial scale-up of complex intermediates without reoptimization challenges typically encountered with transition metal-catalyzed reactions. The use of standard organic solvents and commercially available reagents ensures consistent raw material supply chains that support reliable API intermediate supplier relationships. The process maintains high efficiency across diverse substituents, enabling flexible manufacturing of multiple derivative compounds from a single platform technology. This scalability advantage provides pharmaceutical manufacturers with confidence in long-term supply continuity while accommodating evolving pipeline requirements through the same robust manufacturing platform.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN115260188A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
