Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-beta-carbolinones for Commercial Scale-up
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for efficient access to biologically active scaffolds. Among these, the tetrahydro-beta-carbolinone skeleton stands out as a privileged structure found in numerous natural products and pharmaceutical candidates, such as the antiviral agent bauerine C and the anxiolytic candidate SL651498. 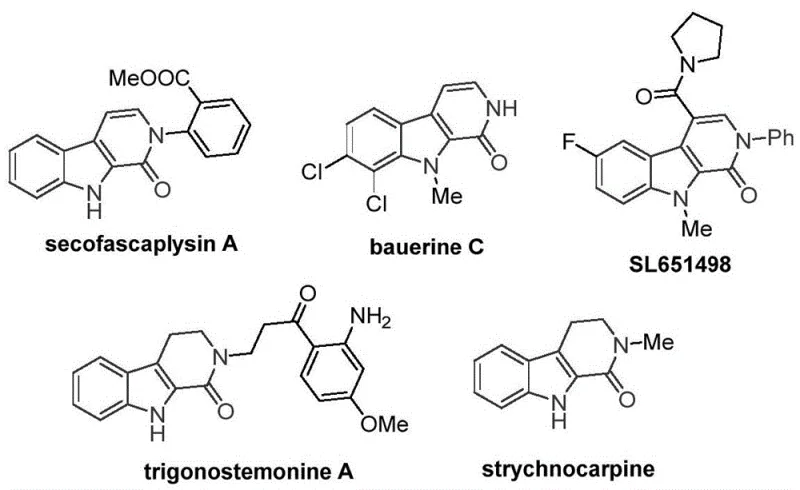 . Despite their therapeutic potential, traditional synthetic routes often rely on expensive transition metals or harsh conditions that limit their commercial viability. A breakthrough in this domain is detailed in Chinese Patent CN115260188A, which discloses a robust preparation method for tetrahydro-beta-carbolinone compounds. This innovation leverages a transition metal cobalt-catalyzed C-H activated carbonylation reaction, utilizing tryptamine derivatives as accessible starting materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards more sustainable and cost-effective manufacturing processes.
. Despite their therapeutic potential, traditional synthetic routes often rely on expensive transition metals or harsh conditions that limit their commercial viability. A breakthrough in this domain is detailed in Chinese Patent CN115260188A, which discloses a robust preparation method for tetrahydro-beta-carbolinone compounds. This innovation leverages a transition metal cobalt-catalyzed C-H activated carbonylation reaction, utilizing tryptamine derivatives as accessible starting materials. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards more sustainable and cost-effective manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrahydro-beta-carbolinone core has been fraught with synthetic challenges that impede large-scale production. Conventional methodologies frequently depend on palladium catalysis for carbonylation reactions. While effective on a small laboratory scale, palladium is a precious metal with volatile pricing and supply chain vulnerabilities, making it less attractive for high-volume commercial manufacturing. Furthermore, many existing protocols require the use of gaseous carbon monoxide, a highly toxic and hazardous reagent that necessitates specialized equipment and rigorous safety protocols, thereby increasing capital expenditure and operational complexity. These factors collectively contribute to higher production costs and longer lead times, creating significant bottlenecks for companies aiming to bring beta-carbolinone-based therapeutics to market efficiently.
The Novel Approach
The methodology described in the patent offers a compelling alternative by replacing precious palladium with earth-abundant cobalt and utilizing a solid carbon monoxide substitute. As illustrated in the general reaction scheme, the process involves reacting a tryptamine derivative with a cobalt catalyst, an oxidant, and a CO surrogate in an organic solvent.  . This approach eliminates the need for handling toxic CO gas, significantly enhancing workplace safety and reducing regulatory burdens. The reaction operates under relatively mild thermal conditions (120-140°C) and demonstrates exceptional substrate compatibility, tolerating various functional groups such as halogens, alkoxy, and alkyl chains. This versatility allows for the rapid generation of diverse compound libraries, accelerating the drug discovery timeline while maintaining a streamlined workflow suitable for industrial application.
. This approach eliminates the need for handling toxic CO gas, significantly enhancing workplace safety and reducing regulatory burdens. The reaction operates under relatively mild thermal conditions (120-140°C) and demonstrates exceptional substrate compatibility, tolerating various functional groups such as halogens, alkoxy, and alkyl chains. This versatility allows for the rapid generation of diverse compound libraries, accelerating the drug discovery timeline while maintaining a streamlined workflow suitable for industrial application.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams optimizing process parameters. The reaction initiates with the oxidation of the cobalt(II) catalyst, typically cobalt acetate tetrahydrate, by silver carbonate to generate a reactive cobalt(III) species. This high-valent cobalt intermediate then coordinates with the tryptamine derivative, facilitating the critical C-H bond activation at the 2-position of the indole ring. This step forms a stable cobalt(III) metallacycle, setting the stage for the subsequent carbonylation event. The carbon monoxide required for the ring closure is not introduced as a gas but is released in situ from the decomposition of 1,3,5-tricarboxylic acid phenol ester (TFBen). This controlled release ensures a steady concentration of CO, promoting efficient insertion into the cobalt-carbon bond to form an acylcobalt(III) intermediate.
The final stage of the catalytic cycle involves reductive elimination and hydrolysis of the acylcobalt(III) species, which releases the desired tetrahydro-beta-carbolinone product and regenerates the active cobalt catalyst. This mechanism highlights the dual role of the additives: pivalic acid acts as a proton shuttle to assist in C-H activation, while triethylamine serves as a base to neutralize acidic byproducts. The use of silver carbonate as a terminal oxidant is also pivotal, driving the catalytic turnover by re-oxidizing the reduced cobalt species back to its active state. This intricate balance of reagents ensures high conversion rates and minimizes the formation of side products, resulting in a clean impurity profile that simplifies downstream purification efforts.
How to Synthesize Tetrahydro-beta-carbolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it highly attractive for process chemistry teams. The protocol utilizes common laboratory reagents and standard heating equipment, avoiding the need for high-pressure reactors or glovebox techniques. By adhering to the optimized molar ratios and reaction times specified in the patent, manufacturers can achieve consistent yields across different batches. The following guide outlines the standardized procedure derived from the patent data, ensuring that the critical parameters for successful cyclization are met.
- Charge a reaction vessel with cobalt catalyst (Co(OAc)2·4H2O), base (Et3N), additive (PivOH), oxidant (Ag2CO3), CO substitute (TFBen), and tryptamine derivative in dioxane.
- Heat the mixture to 120-140°C and stir for 16-24 hours to facilitate C-H activation and carbonylation cyclization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target tetrahydro-beta-carbolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this cobalt-catalyzed protocol offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with catalyst replacement. By shifting from palladium to cobalt, companies can decouple their production costs from the fluctuating precious metal markets, leading to more predictable budgeting and improved margin stability. Additionally, the elimination of gaseous carbon monoxide removes the logistical complexities and safety costs associated with storing and transporting hazardous gases, further streamlining the supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the substitution of expensive noble metals with base metals. Cobalt acetate is significantly cheaper than palladium catalysts, and the use of a solid CO surrogate eliminates the need for specialized gas handling infrastructure. This combination results in substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing of the final API. Moreover, the high atom economy and efficient conversion rates minimize waste generation, reducing the costs associated with waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable reagents. Unlike specialized ligands or sensitive organometallic complexes that may have long lead times, cobalt salts, silver carbonate, and the CO surrogate are commodity chemicals with robust global supply networks. This availability ensures continuous production capabilities even during market disruptions. The simplicity of the workup procedure, involving filtration and standard column chromatography, also reduces the dependency on complex purification technologies, making the process easier to transfer between different manufacturing sites.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively on gram scales with the potential for multi-kilogram expansion. The reaction conditions are compatible with standard stainless steel reactors, facilitating a smooth transition from pilot plant to commercial production. From an environmental perspective, the method aligns with green chemistry principles by avoiding toxic gases and reducing heavy metal contamination in the final product. This compliance with stringent environmental regulations simplifies the permitting process and enhances the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this technology, we have compiled a set of questions based on the specific experimental data and scope defined in the patent. These answers provide clarity on the operational boundaries and capabilities of the cobalt-catalyzed system, helping stakeholders make informed decisions about adopting this route for their specific projects.
Q: What are the advantages of using cobalt over palladium for this synthesis?
A: Cobalt catalysts are significantly more earth-abundant and cost-effective than precious palladium catalysts. Furthermore, this method utilizes a solid CO surrogate rather than toxic gas, enhancing operational safety and simplifying the supply chain for large-scale manufacturing.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The process demonstrates excellent functional group tolerance. R1 can be hydrogen, methyl, methoxy, bromo, or chloro substituents, while R2 accommodates hydrogen, alkyl groups, benzyl, phenyl, naphthyl, or allyl groups, allowing for diverse library synthesis.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly states the method is simple to operate with readily available starting materials. The use of standard solvents like dioxane and robust reaction conditions (130°C) supports scalability from gram to multi-kilogram production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-beta-carbolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed technology for the pharmaceutical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydro-beta-carbolinone intermediate meets the highest quality standards required for drug development.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this method can optimize your overall production budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with confidence.
