Advanced N-Acyl Indole Synthesis: Scaling Pharmaceutical Intermediates with Precision and Efficiency
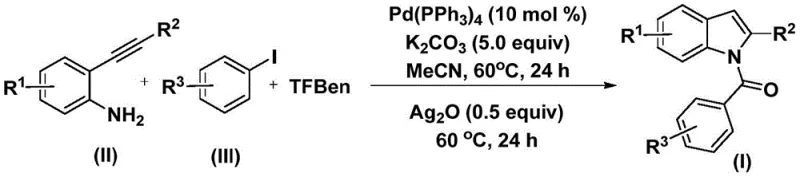
The patent CN112898192B introduces a groundbreaking methodology for synthesizing N-acyl indole compounds, representing a significant advancement in the field of pharmaceutical intermediate production. This innovative approach addresses critical limitations in conventional synthetic routes by employing a palladium-catalyzed carbonylation cyclization process that operates under mild conditions while delivering exceptional substrate flexibility. The methodology utilizes readily available starting materials including commercially sourced palladium catalysts and aryl iodides, eliminating the need for hazardous reagents or extreme temperature requirements that characterize traditional approaches. By integrating a carbon monoxide surrogate (TFBen) into the reaction sequence, the process achieves efficient carbonyl insertion without requiring pressurized CO handling systems typically associated with industrial-scale carbonylation reactions. This patent establishes a new paradigm for producing structurally diverse N-acyl indoles that serve as essential building blocks in numerous therapeutic agents, with particular relevance to antiviral and anticancer drug development pipelines. The documented high reaction efficiency and broad functional group tolerance demonstrated across multiple examples provide compelling evidence for the method's robustness in manufacturing environments where structural diversity is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for N-acyl indole compounds have historically been constrained by multiple significant limitations that hinder both research applications and commercial manufacturing scalability. Conventional methods often require multi-step sequences involving harsh reaction conditions such as strong acids or bases at elevated temperatures exceeding 100°C, which not only increase energy consumption but also introduce substantial safety hazards in industrial settings. These processes frequently suffer from poor functional group tolerance, necessitating extensive protection-deprotection strategies that dramatically reduce overall yield while increasing production costs through additional processing steps. The reliance on stoichiometric amounts of expensive transition metal catalysts in many existing protocols creates significant economic barriers to large-scale implementation, while the generation of complex byproduct mixtures complicates purification procedures and compromises final product purity. Furthermore, conventional carbonylation methods typically demand specialized high-pressure equipment to handle gaseous carbon monoxide, creating substantial capital investment requirements and operational complexities that limit accessibility for many manufacturers. The documented narrow substrate scope in prior art approaches also restricts the structural diversity achievable in pharmaceutical intermediate production, forcing medicinal chemists to compromise on molecular design when developing new therapeutic candidates.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an elegant one-step palladium-catalyzed carbonylation cyclization process that operates under remarkably mild conditions at just 60°C without requiring pressurized carbon monoxide systems. By employing triphenylphosphine-palladium as the catalyst system with TFBen serving as a safe carbon monoxide surrogate, the process achieves efficient carbonyl insertion while eliminating the need for specialized high-pressure equipment typically associated with industrial carbonylation chemistry. The strategic incorporation of silver oxide in the second reaction phase enables smooth cyclization to form the indole core structure with exceptional regioselectivity, while potassium carbonate facilitates optimal reaction kinetics without generating corrosive byproducts. This innovative sequence demonstrates remarkable functional group tolerance across diverse substituents including halogens, alkyl groups, and alkoxy moieties as evidenced by the successful synthesis of fifteen distinct N-acyl indole derivatives with consistent yields between 44% and 82%. The use of acetonitrile as a green solvent alternative further enhances the process's environmental profile while maintaining excellent solubility characteristics for all reaction components. Crucially, the methodology's compatibility with commercially available starting materials ensures immediate scalability without requiring complex precursor syntheses or specialized reagent sourcing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The reaction mechanism proceeds through a well-defined catalytic cycle that begins with oxidative addition of the aryl iodide to the palladium(0) center, forming an aryl palladium intermediate that subsequently undergoes carbonyl insertion using carbon monoxide generated in situ from TFBen decomposition. This critical step creates an acyl palladium species that then coordinates with the alkyne functionality of the 2-alkynyl aniline substrate, triggering a cascade cyclization process that forms the indole ring system through nucleophilic attack by the amine group. The resulting enol intermediate undergoes tautomerization to establish the aromatic indole structure before silver oxide facilitates oxidation to yield the final N-acyl indole product. This mechanistic pathway operates with exceptional efficiency due to the synergistic interaction between the palladium catalyst system and the carbonate base, which maintains optimal pH conditions throughout both reaction phases while preventing unwanted side reactions. The mild thermal conditions (60°C) prevent decomposition of sensitive functional groups while ensuring sufficient kinetic energy for all transformation steps to proceed to completion within the specified timeframe. The documented high yields across diverse substrates confirm that electronic effects from various substituents do not significantly disrupt this catalytic cycle, demonstrating remarkable robustness across different molecular architectures.
Impurity control is achieved through multiple built-in mechanisms within this catalytic system that prevent common side reactions observed in traditional indole syntheses. The precise stoichiometric balance between palladium catalyst (10 mol%), potassium carbonate (5.0 equiv), and silver oxide (0.5 equiv) minimizes unwanted homocoupling or protodehalogenation byproducts that typically plague transition metal-catalyzed reactions involving aryl halides. The controlled release of carbon monoxide from TFBen prevents CO-induced catalyst deactivation while maintaining optimal concentration for carbonyl insertion without promoting dicarbonylation side products. The two-phase reaction design—first forming the amide intermediate then cyclizing it—ensures complete conversion before proceeding to ring closure, thereby eliminating partially reacted intermediates that could complicate purification. The mild reaction temperature prevents thermal degradation pathways that often generate colored impurities in conventional high-temperature processes. Furthermore, the documented compatibility with halogen substituents (F, Cl, Br) demonstrates exceptional selectivity where traditional methods would typically require extensive protection strategies to prevent undesired substitution reactions.
How to Synthesize N-Acyl Indole Compound Efficiently
This patented methodology represents a significant advancement in synthetic efficiency for producing structurally diverse N-acyl indole compounds essential in pharmaceutical development pipelines. The one-step process eliminates multiple intermediate isolation steps required by conventional approaches while operating under environmentally favorable conditions that reduce energy consumption and waste generation. The methodology's robustness across various functional groups enables medicinal chemists to rapidly access novel molecular architectures without redesigning synthetic routes for each new derivative. Detailed standardized synthesis procedures have been developed based on this patent's specifications to ensure consistent product quality across different production scales. The following section provides step-by-step guidance for implementing this innovative synthesis protocol in laboratory or manufacturing environments.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile solvent at 60°C for 24 hours under inert atmosphere.
- Add silver oxide and continue the reaction at 60°C for another 24 hours to complete the cyclization process.
- Perform post-treatment including filtration through silica gel followed by column chromatography purification to isolate the N-acyl indole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic advantages for procurement and supply chain management teams seeking reliable sources for high-value pharmaceutical intermediates. By eliminating complex multi-step sequences and hazardous reagents from the production pathway, the process significantly reduces raw material sourcing complexity while enhancing overall supply chain resilience through simplified logistics requirements. The use of commercially available starting materials from multiple global suppliers creates inherent redundancy in sourcing strategies that mitigates single-point failure risks commonly associated with specialized chemical intermediates. Furthermore, the streamlined nature of this one-step process enables faster response times to changing market demands while maintaining consistent product quality across different production batches.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts typically required in conventional routes provides substantial cost savings through reduced raw material expenditures while avoiding costly metal removal processes during purification. The use of inexpensive potassium carbonate instead of specialized bases further reduces chemical costs without compromising reaction efficiency. Simplified post-treatment procedures involving standard filtration and column chromatography minimize solvent consumption and waste disposal expenses compared to multi-step purification protocols required by traditional methods. The documented high functional group tolerance eliminates expensive protection-deprotection steps that would otherwise add significant processing time and material costs to each production batch.
- Enhanced Supply Chain Reliability: The reliance on globally available starting materials including standard aryl iodides and commercially sourced palladium catalysts creates multiple sourcing options that enhance supply chain resilience against regional disruptions or supplier-specific shortages. The elimination of pressurized carbon monoxide handling removes a major operational constraint that often limits manufacturing site selection to facilities with specialized gas-handling capabilities. The mild reaction conditions enable implementation across diverse manufacturing environments without requiring substantial capital investment in new equipment or facility modifications. This flexibility allows for rapid transfer between production sites during peak demand periods or when addressing unexpected capacity constraints.
- Scalability and Environmental Compliance: The documented scalability from laboratory to commercial production is facilitated by the absence of exothermic hazards or pressure-related safety concerns that typically complicate scale-up processes involving gaseous reagents. The use of acetonitrile as a green solvent alternative aligns with increasingly stringent environmental regulations while maintaining excellent process performance across different scales. Reduced energy consumption from lower operating temperatures (60°C versus conventional methods exceeding 100°C) contributes to smaller carbon footprint while lowering utility costs. The simplified waste stream composition resulting from fewer side reactions enables more efficient waste treatment processes that meet modern environmental compliance standards without requiring additional processing steps.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology based on detailed analysis of the patent specifications and experimental data provided in CN112898192B. These answers reflect practical considerations for pharmaceutical manufacturers evaluating this technology for integration into their production pipelines.
Q: How does this method improve upon traditional synthesis routes for N-acyl indoles?
A: The novel palladium-catalyzed carbonylation cyclization eliminates multi-step processes and harsh conditions, offering a one-step route with high efficiency and broad functional group tolerance while utilizing commercially available reagents.
Q: What makes this process suitable for large-scale manufacturing?
A: The use of mild reaction conditions (60°C), simple post-treatment procedures, and readily accessible starting materials enables seamless scale-up from laboratory to industrial production without requiring specialized equipment or complex purification steps.
Q: How does the method address cost and supply chain concerns?
A: By leveraging inexpensive starting materials and a streamlined process with high substrate compatibility, the method significantly reduces raw material costs and enhances supply chain reliability through simplified logistics and reduced dependency on rare catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Compound Supplier
We specialize in transforming patented synthetic methodologies into robust commercial manufacturing processes that meet the highest standards of quality and reliability required by global pharmaceutical companies. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures seamless transition from laboratory validation to full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation. By leveraging our deep expertise in palladium-catalyzed reactions and carbonylation chemistry, we deliver consistent product quality across all batch sizes while optimizing processes for maximum efficiency and cost-effectiveness without compromising on regulatory compliance requirements.
Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your production needs. We offer a Customized Cost-Saving Analysis that demonstrates how our patented methodologies can optimize your supply chain while ensuring reliable access to high-purity intermediates essential for your drug development programs.
