Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A recent breakthrough detailed in Chinese Patent CN113307790B introduces a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These specific molecular architectures are of immense value due to the prevalence of the 1,2,4-triazole ring in bioactive frameworks and its utility as a bidentate ligand in coordination chemistry. The disclosed methodology represents a significant leap forward in synthetic efficiency, moving away from multi-step, low-yield processes toward a streamlined, metal-free oxidative cyclization strategy. By leveraging a catalytic system composed of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), this invention enables the direct coupling of 2-methylquinoline derivatives with trifluoroethylimide hydrazides. For R&D directors and procurement specialists alike, this development signals a potential paradigm shift in how these high-value pharmaceutical intermediates are sourced and manufactured, offering a route that is not only chemically elegant but also commercially viable for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by significant inefficiencies that hinder commercial scalability. Traditional literature methods typically rely on quinoline-2-formic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target molecule. This linear synthesis is not only time-consuming but also suffers from abysmal overall yields, often reported as low as 17%. Furthermore, these legacy routes frequently demand severe reaction conditions, including strict anhydrous and oxygen-free environments, which impose heavy operational burdens on manufacturing facilities. The requirement for such stringent controls increases energy consumption, complicates reactor design, and elevates the risk of batch failure due to environmental exposure. Additionally, the reliance on precious metal catalysts in some conventional cross-coupling approaches introduces downstream purification challenges, specifically the costly and technically difficult removal of trace heavy metals to meet regulatory standards for API intermediates. These cumulative factors render traditional methods economically unfeasible for the high-volume production required by the global supply chain.
The Novel Approach
In stark contrast, the methodology disclosed in CN113307790B offers a transformative solution by utilizing cheap and readily available 2-methylquinoline and trifluoroethylimide hydrazide as starting materials. This innovative route achieves the construction of the triazole ring through a direct oxidative cyclization promoted by a TBAI/TBHP system. The reaction proceeds efficiently in common organic solvents like DMSO at moderate temperatures ranging from 80°C to 100°C. Crucially, this process eliminates the need for toxic heavy metal catalysts and does not require inert atmosphere conditions, drastically simplifying the operational protocol. As illustrated in the general reaction scheme below, the transformation is direct and atom-economical, converting simple precursors into complex heterocycles with remarkable speed and efficiency.
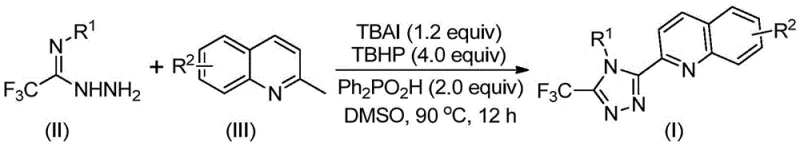
The versatility of this novel approach is further evidenced by its broad substrate scope, allowing for the introduction of diverse substituents on both the quinoline and the aryl rings without compromising yield. Experimental data from the patent indicates that yields can reach as high as 97% for certain substrates, a massive improvement over the historical 17% benchmark. This dramatic increase in efficiency directly translates to reduced waste generation and lower raw material consumption, addressing key sustainability goals in modern chemical manufacturing.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The success of this synthesis lies in the intricate interplay between the iodide catalyst and the peroxide oxidant, which facilitates a cascade of oxidative transformations. The mechanism likely initiates with the oxidation of the methyl group on the 2-methylquinoline substrate by the TBAI/TBHP system, generating a reactive 2-quinoline carbaldehyde intermediate in situ. This aldehyde then undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination activates the hydrazone species, triggering an intramolecular electrophilic substitution that closes the triazole ring. The final aromatization step releases the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole product. This radical-mediated or electrophilic pathway bypasses the need for pre-functionalized starting materials, such as acid chlorides or esters, thereby reducing the step count and associated costs.
From an impurity control perspective, the metal-free nature of this catalytic system is a distinct advantage for high-purity pharmaceutical intermediate production. Traditional transition metal-catalyzed reactions often leave behind trace amounts of palladium, copper, or nickel, which are strictly regulated in drug substances. By utilizing an organic iodide salt (TBAI) as the promoter, the risk of heavy metal contamination is virtually eliminated. This simplifies the downstream purification process, often requiring only standard filtration and column chromatography to achieve high purity specifications. The use of diphenylphosphoric acid as an additive further enhances reaction selectivity, minimizing the formation of side products and ensuring a clean impurity profile that is easier to characterize and control during quality assurance testing.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting involves a straightforward one-pot procedure that minimizes handling risks and equipment requirements. The process begins by charging a reactor with the requisite molar ratios of tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in a solvent such as DMSO. The mixture is then heated to the optimal temperature range of 80-100°C and maintained for 8 to 14 hours to ensure full conversion. The simplicity of the workup procedure, involving filtration followed by silica gel mixing and column chromatography, makes this method particularly attractive for rapid process development and scale-up activities.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP), diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in an organic solvent like DMSO.
- Heat the reaction mixture to 80-100°C and maintain stirring for 8-14 hours to ensure complete oxidative cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from a five-step sequence to a single-pot reaction fundamentally alters the cost structure of manufacturing these valuable intermediates. By collapsing multiple unit operations into one, manufacturers can significantly reduce labor costs, energy consumption, and solvent usage. Furthermore, the elimination of expensive transition metal catalysts removes a major line item from the bill of materials, while simultaneously avoiding the capital expenditure associated with specialized metal scavenging equipment. This streamlined approach allows for a more agile supply chain capable of responding quickly to market demands for complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the use of commodity chemicals rather than specialized reagents. Starting materials like 2-methylquinoline and aromatic amines are produced on a massive industrial scale, ensuring stable pricing and abundant availability. The replacement of precious metal catalysts with inexpensive TBAI and TBHP results in substantial cost savings per kilogram of product. Additionally, the high reaction yields observed (often exceeding 80-90%) mean that less raw material is wasted, directly improving the overall material efficiency and reducing the cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Since the process does not require strict anhydrous or anaerobic environments, it can be performed in standard glass-lined or stainless steel reactors without the need for complex inert gas blanketing systems. This reduces the risk of batch failures due to minor leaks or moisture ingress, leading to more predictable production schedules and reliable delivery times. The use of widely available solvents like DMSO further ensures that production is not bottlenecked by the scarcity of niche reagents, facilitating a smoother flow of materials through the global supply network.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this metal-free protocol aligns well with green chemistry principles. The absence of heavy metals simplifies waste treatment and disposal, lowering the environmental compliance burden and associated fees. The reaction's ability to be scaled from gram-level experiments to multi-kilogram production without significant re-optimization demonstrates its inherent scalability. This ease of scale-up allows manufacturers to rapidly transition from clinical trial supplies to commercial production volumes, ensuring continuity of supply for downstream drug developers without the typical delays associated with process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this metal-free synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-formic acid and five steps with only 17% yield, this novel approach uses cheap 2-methylquinoline in a one-pot reaction with yields up to 97%, eliminating the need for expensive heavy metal catalysts and harsh anhydrous conditions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states the reaction can be easily expanded to gram-scale and potentially larger, utilizing commercially available reagents like TBAI and TBHP without requiring specialized oxygen-free environments, making it highly scalable.
Q: What represents the primary cost driver in this synthesis?
A: The primary cost drivers are minimized because the starting materials (2-methylquinoline and aromatic amines) are inexpensive and widely available, and the catalyst system (TBAI/TBHP) avoids the high costs associated with transition metal removal and disposal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the CN113307790B patent and is well-positioned to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of high-purity pharmaceutical intermediate we deliver meets the highest international standards.
We invite you to collaborate with us to explore how this advanced oxidative cyclization route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in metal-free catalysis can drive value for your organization.
