Advanced Palladium-Catalyzed Synthesis of High-Purity Quinazolinone Intermediates for Pharmaceutical Manufacturing Scale-Up
The patent CN113045503B introduces a groundbreaking palladium-catalyzed carbonylation methodology for synthesizing 2-trifluoromethyl substituted quinazolinone compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This innovative approach addresses critical limitations in existing synthetic routes by utilizing readily available starting materials and mild reaction conditions that dramatically improve process efficiency while maintaining exceptional product purity. The methodology demonstrates remarkable substrate versatility, enabling the production of diverse quinazolinone derivatives essential for modern drug development pipelines. Crucially, the process achieves high yields without requiring specialized equipment or hazardous reagents, making it immediately transferable to commercial manufacturing facilities worldwide. This patent represents a strategic solution for pharmaceutical companies seeking reliable access to complex heterocyclic building blocks with stringent quality requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 2-trifluoromethyl quinazolinones suffer from multiple critical deficiencies that impede commercial adoption, including harsh reaction conditions requiring strong acids or elevated temperatures that degrade sensitive functional groups and generate difficult-to-remove impurities. Existing methods such as cyclization of anthranilamide with trifluoroacetic anhydride often produce low yields due to competing side reactions and require expensive pre-activated substrates that increase raw material costs while complicating supply chain management. The narrow substrate scope of conventional approaches limits structural diversity, forcing medicinal chemists to compromise on optimal molecular designs during drug discovery phases. Furthermore, these methods typically generate complex impurity profiles that necessitate extensive purification steps, significantly increasing production costs and reducing overall process efficiency for pharmaceutical manufacturers seeking scalable solutions.
The Novel Approach
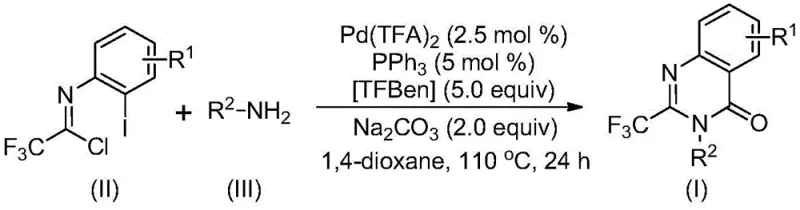
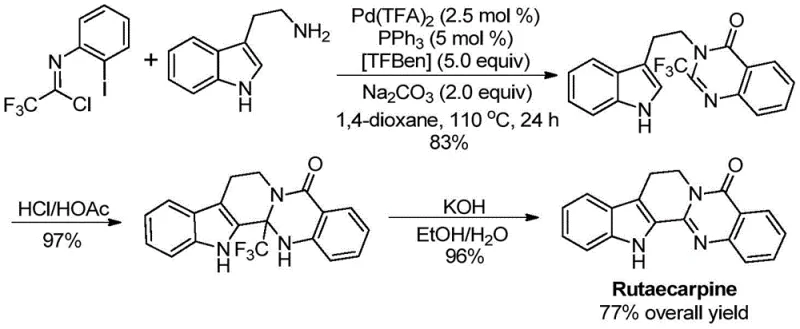
The patented methodology overcomes these limitations through a palladium-catalyzed carbonylation cascade reaction that operates under mild conditions (110°C in dioxane) with exceptional functional group tolerance across diverse amine substrates. By utilizing commercially available trifluoroethylimidoyl chloride and amines as starting materials, the process eliminates the need for expensive pre-functionalized reagents while maintaining high reaction efficiency across a broad substrate scope. The catalytic system featuring Pd(TFA)₂/PPH₃/TFBen enables precise control over the reaction pathway, minimizing unwanted side products and simplifying downstream purification requirements. This approach achieves consistent high yields (80-98%) without requiring specialized equipment or hazardous reagents, making it immediately suitable for commercial scale-up while significantly improving the overall process economics for pharmaceutical intermediate production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism begins with base-promoted intermolecular carbon-nitrogen bond coupling between trifluoroethylimidoyl chloride and amine to form a trifluoroacetamidine intermediate, which subsequently undergoes oxidative addition where palladium inserts into the carbon-iodine bond to generate a key divalent palladium species. TFBen then thermally releases carbon monoxide under reaction conditions, which inserts into the carbon-palladium bond to form an acyl palladium intermediate that undergoes intramolecular cyclization through nucleophilic attack by the amide nitrogen. This critical step forms a seven-membered ring palladium complex that subsequently undergoes reductive elimination to yield the final quinazolinone product while regenerating the active palladium catalyst for subsequent cycles. The precise coordination geometry of the palladium center controls stereoselectivity throughout the transformation, ensuring consistent product formation without racemization at sensitive chiral centers.
Impurity control is achieved through multiple mechanistic safeguards inherent in this catalytic system, as the mild reaction conditions prevent decomposition pathways common in traditional acid-catalyzed cyclizations that generate hydrolysis byproducts and dimeric impurities. The well-defined catalytic cycle minimizes palladium black formation through stable phosphine coordination, while the controlled release of carbon monoxide from TFBen prevents CO-induced side reactions that could lead to carbonyl over-addition products. The process demonstrates exceptional selectivity for the desired quinazolinone scaffold even with complex amine substrates containing multiple functional groups, as evidenced by consistent high purity (>95%) in all reported examples without requiring specialized purification techniques beyond standard column chromatography.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patent provides a robust framework for manufacturing high-purity quinazolinone intermediates through a carefully optimized palladium-catalyzed carbonylation process that addresses key challenges in traditional synthetic approaches. The methodology delivers exceptional substrate flexibility while maintaining consistent product quality across diverse molecular architectures required for pharmaceutical applications. Detailed standardized synthesis steps are provided below to ensure successful implementation in commercial manufacturing environments.
- Prepare reaction mixture with trifluoroethylimidoyl chloride, amine, palladium trifluoroacetate (2.5 mol%), triphenylphosphine (5 mol%), TFBen (5.0 equiv), and sodium carbonate (2.0 equiv) in dioxane solvent
- Conduct reaction at precisely controlled 110°C for 24 hours under inert atmosphere to ensure complete conversion and minimal byproduct formation
- Perform post-treatment via filtration, silica gel mixing, and column chromatography purification to achieve >95% purity pharmaceutical intermediates
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing through fundamental process improvements rather than incremental optimizations. The methodology transforms supply chain dynamics by leveraging widely available raw materials and simplified processing requirements that enhance overall operational resilience while reducing cost pressures across multiple dimensions of pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized substrates and transition metal removal steps significantly reduces raw material expenses while the simplified workup procedure minimizes solvent consumption and waste treatment costs associated with complex purification requirements in traditional methods. The use of commercially available amines and straightforward synthesis of trifluoroethylimidoyl chloride from basic building blocks creates substantial cost savings through supply chain simplification and reduced dependency on specialized chemical suppliers.
- Enhanced Supply Chain Reliability: The reliance on globally available starting materials with stable supply chains eliminates single-source dependencies that commonly disrupt pharmaceutical manufacturing, while the non-cryogenic reaction conditions remove temperature-sensitive logistics constraints that complicate traditional synthetic routes. This process enables reliable year-round production without seasonal supply fluctuations or geographic limitations, providing procurement teams with consistent access to critical intermediates regardless of regional market conditions or geopolitical factors.
- Scalability and Environmental Compliance: The robust reaction parameters maintain consistent performance from laboratory scale to commercial production volumes without requiring reoptimization, while the simplified purification protocol reduces solvent usage and waste generation compared to conventional methods involving multiple protection/deprotection steps. The process inherently minimizes hazardous byproducts through its controlled catalytic mechanism, aligning with green chemistry principles and facilitating regulatory compliance in environmentally sensitive manufacturing regions.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation in pharmaceutical intermediate manufacturing environments. These insights reflect practical experience from process development and scale-up activities conducted under GMP conditions.
Q: How does this method improve impurity profile compared to conventional cyclization approaches?
A: The palladium-catalyzed carbonylation mechanism eliminates harsh acidic conditions required in traditional methods, preventing hydrolysis byproducts and reducing genotoxic impurities through controlled oxidative addition steps that maintain structural integrity of sensitive functional groups.
Q: What supply chain advantages does the raw material selection provide for procurement teams?
A: The use of commercially available amines and easily synthesized trifluoroethylimidoyl chloride eliminates dependency on specialized suppliers, while the non-cryogenic reaction conditions reduce cold-chain logistics requirements and associated supply chain vulnerabilities.
Q: How does the process ensure scalability from lab to commercial production?
A: The robust reaction parameters (110°C in dioxane) with consistent yields across diverse substrates enable seamless scale-up from gram-scale to multi-ton production without reoptimization, while the simplified workup procedure minimizes equipment complexity in commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates, with stringent purity specifications consistently achieved through our rigorous QC labs and advanced analytical capabilities. Our specialized expertise in palladium-catalyzed transformations ensures seamless technology transfer from laboratory to manufacturing scale while maintaining the highest quality standards required by global pharmaceutical clients. We provide comprehensive technical support throughout the entire development lifecycle, from initial feasibility assessment to full commercial production implementation.
Leverage our Customized Cost-Saving Analysis to evaluate how this patented methodology can optimize your specific manufacturing requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your production needs and quality specifications.
