Advanced Palladium-Catalyzed Route for Scalable 2-Trifluoromethyl Imidazole Production
Introduction to Next-Generation Imidazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups, due to their profound impact on drug efficacy. The introduction of a trifluoromethyl moiety into a heterocyclic backbone significantly enhances electronegativity, bioavailability, metabolic stability, and lipophilicity, making these structures highly coveted in modern medicinal chemistry. A groundbreaking development in this domain is detailed in Chinese Patent CN111423381B, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. This technology represents a paradigm shift from traditional, often hazardous synthetic routes to a streamlined, palladium-catalyzed carbonylation process. By leveraging inexpensive and commercially accessible starting materials, this innovation addresses critical pain points in API intermediate manufacturing, offering a pathway that is not only chemically elegant but also economically viable for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant challenges that hinder efficient commercial production. Conventional literature reports predominantly rely on the direct reaction of synthons containing trifluoromethyl groups with suitable substrates, often utilizing reagents such as trifluorodiazoethane. While chemically effective in academic settings, these traditional synthons are characterized by their instability, high cost, and potential safety hazards, which pose severe risks during scale-up. Furthermore, many existing protocols require harsh reaction conditions, including elevated temperatures and pressures, which increase energy consumption and complicate process safety management. The limited availability and high price point of specialized trifluoromethylating agents create a bottleneck in the supply chain, leading to inflated production costs and extended lead times for critical pharmaceutical intermediates. Consequently, there has been an urgent industry demand for a safer, more cost-effective, and operationally simple alternative that does not compromise on yield or substrate compatibility.
The Novel Approach
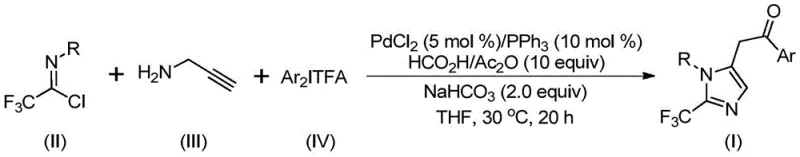
The methodology outlined in patent CN111423381B offers a transformative solution by employing a transition metal palladium-catalyzed carbonylation series reaction. This innovative route utilizes trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the foundational building blocks. Unlike previous methods, this process operates under remarkably mild thermal conditions, specifically at a constant temperature of 30°C, which drastically reduces the energy footprint and eliminates the need for complex heating infrastructure. The reaction system is designed to be highly tolerant of various functional groups, allowing for the synthesis of diversified substituted imidazole compounds through simple substrate design. By replacing dangerous diazo compounds with stable imidoyl chlorides and utilizing a catalytic system that promotes efficient carbon-nitrogen bond formation, this approach ensures high reaction efficiency and excellent substrate compatibility. The simplicity of the operation, combined with the use of cheap and easily obtained raw materials, makes this method exceptionally suitable for industrial expansion.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific API candidates. The reaction proceeds through a sophisticated catalytic cycle initiated by the formation of an intermolecular carbon-nitrogen bond promoted by alkali, yielding a trifluoroacetamidine compound. This intermediate undergoes isomerization, followed by the palladation of the alkyne amine to generate an alkenyl palladium intermediate. Subsequent isomerization converts this species into an alkyl palladium intermediate, setting the stage for the critical carbonylation step. In the presence of carbon monoxide released in situ from the formic acid and acetic anhydride mixture, an acyl palladium intermediate is formed. The cycle culminates with the oxidative addition of the diaryl iodonium salt to produce a tetravalent palladium intermediate, which finally undergoes reductive elimination to release the desired 2-trifluoromethyl-substituted imidazole product. This intricate mechanism ensures high selectivity and minimizes the formation of unwanted by-products, thereby simplifying downstream purification.
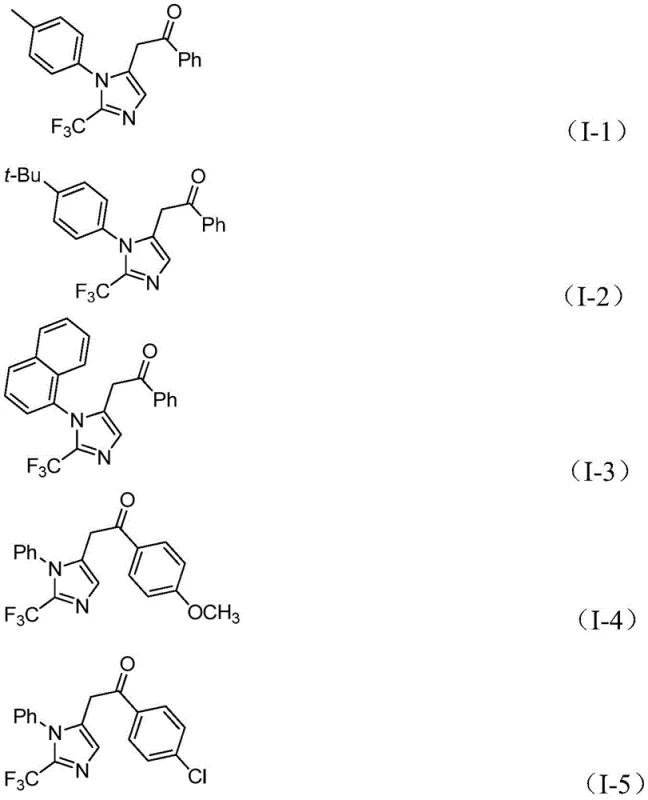
Furthermore, the versatility of this catalytic system is evidenced by its broad substrate scope, accommodating various substituents on both the aryl groups of the imidoyl chloride and the diaryl iodonium salt. Whether the substituents are electron-donating groups like methyl or methoxy, or electron-withdrawing groups such as chlorine, bromine, trifluoromethyl, or nitro, the reaction maintains high efficiency. This flexibility allows chemists to tailor the electronic and steric properties of the final imidazole scaffold to meet specific biological activity requirements without needing to redesign the entire synthetic route. The ability to synthesize compounds with different substitutions at the 1 and 5 positions of the imidazole ring further underscores the utility of this method in generating diverse chemical libraries for drug discovery programs.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and purity. The process is designed to be straightforward, minimizing the need for specialized equipment while ensuring reproducible results. Operators should focus on the precise stoichiometric ratios of the catalyst system and the careful control of the reaction temperature to maintain the integrity of the sensitive intermediates. The following section outlines the standardized operational protocol derived from the patent data, serving as a foundational guide for process chemists.
- Prepare the reaction mixture by adding palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid into an organic solvent such as tetrahydrofuran (THF).
- Introduce the key substrates: trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt into the reaction vessel under stirring.
- Maintain the reaction at 30°C for 18 to 20 hours, followed by filtration and purification via silica gel column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The shift towards this methodology directly addresses the perennial challenges of cost volatility and supply continuity in the fine chemical sector. By fundamentally altering the input materials and process conditions, manufacturers can achieve a more resilient and cost-efficient production model.
- Cost Reduction in Manufacturing: The economic implications of switching to this protocol are substantial, primarily driven by the replacement of expensive and hazardous reagents with commodity chemicals. Trifluoroethylimidoyl chloride and propargylamine are significantly cheaper and more abundant than traditional trifluoromethyl synthons like trifluorodiazoethane. Additionally, the use of palladium chloride, a relatively inexpensive palladium source compared to other exotic catalysts, combined with common ligands like triphenylphosphine, lowers the catalyst loading cost. The mild reaction temperature of 30°C eliminates the need for energy-intensive heating or cooling systems, resulting in lower utility costs per batch. Furthermore, the high conversion rates and selectivity reduce the volume of waste generated, lowering disposal costs and improving the overall atom economy of the process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the commercial availability of all key starting materials. Since trifluoroethylimidoyl chloride can be rapidly synthesized from widely available aromatic amines, and diaryl iodonium salts can be prepared from aryl phenylboronic acids and aryl iodides, the risk of raw material shortages is minimized. This reliance on established chemical supply chains ensures consistent access to inputs, reducing the likelihood of production delays caused by sourcing bottlenecks. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the supply of the final intermediate. This reliability is critical for maintaining continuous manufacturing schedules and meeting the stringent delivery timelines required by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles and regulatory standards. The operation at ambient pressure and low temperature inherently reduces safety risks, simplifying the engineering controls required for scale-up from gram to multi-ton levels. The post-treatment procedure involves standard filtration and column chromatography, techniques that are easily adaptable to industrial-scale purification units. Moreover, the avoidance of toxic diazo compounds and the use of benign solvents like tetrahydrofuran contribute to a safer working environment and a reduced environmental footprint. This compliance with safety and environmental regulations facilitates smoother regulatory approvals and reduces the liability associated with handling hazardous materials, making it an ideal choice for sustainable long-term production.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries based on the detailed experimental data provided in the patent documentation. These insights address practical concerns regarding reaction parameters, product quality, and adaptability.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride and operates under exceptionally mild conditions (30°C), avoiding the hazards and costs associated with traditional trifluoromethyl synthons like trifluorodiazoethane.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is simple to operate, has high reaction efficiency, and can be expanded to the gram level, providing a strong foundation for industrial large-scale production and application.
Q: What is the typical yield and purity profile for these compounds?
A: Experimental data indicates high reaction efficiency with yields reaching up to 97% for certain substrates. The post-treatment involves standard column chromatography, ensuring high-purity specifications suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
As the global demand for fluorinated heterocycles continues to surge, partnering with a technically proficient manufacturer is essential for securing a competitive edge in the marketplace. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced catalytic technologies like the one described in CN111423381B to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from R&D to full-scale manufacturing. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl imidazole meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your specific supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your next project.
