Advanced Pd-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Introduction to Next-Generation Imidazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN111423381B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses many limitations of prior art. This technology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. The significance of this development cannot be overstated for manufacturers of high-purity pharmaceutical intermediates, as it offers a streamlined pathway to complex molecular scaffolds found in bioactive molecules like histamine receptor antagonists and coordination ligands.
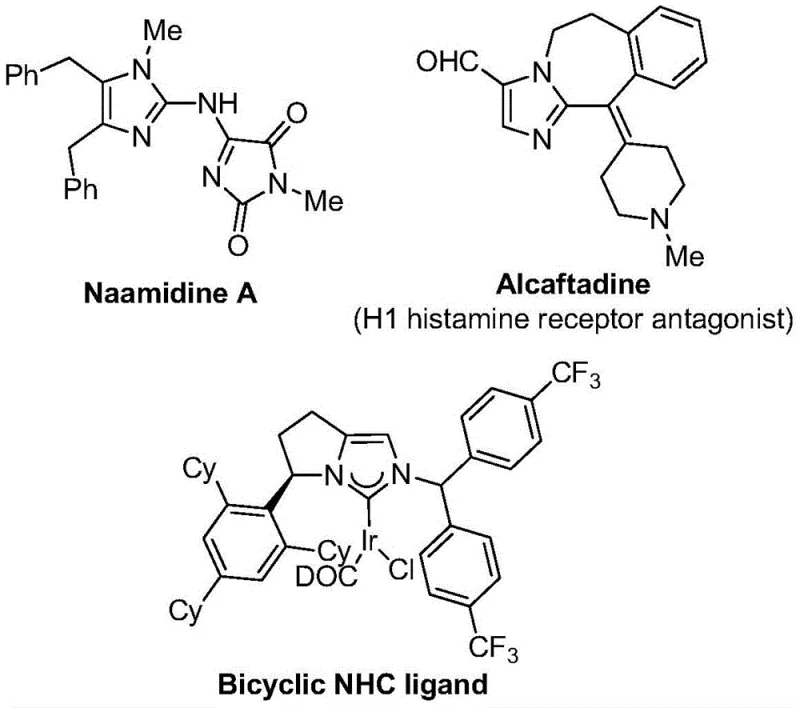
Furthermore, the introduction of the trifluoromethyl group into these heterocyclic systems is critical for optimizing the electronegativity and bioavailability of the parent molecule, making this synthetic route highly valuable for drug discovery pipelines. The method described allows for the design and synthesis of diversified substituted imidazole compounds, thereby broadening the utility of this chemical space for R&D teams aiming to create novel therapeutic agents. By operating under mild conditions and utilizing accessible reagents, this patent represents a significant step forward in cost reduction in pharmaceutical intermediate manufacturing, enabling more efficient supply chains for global healthcare providers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrogen-containing heterocycles with trifluoromethyl functional groups has relied on reacting synthons bearing the trifluoromethyl group with suitable substrates, often involving harsh conditions or unstable reagents. Common trifluoromethyl synthons like trifluorodiazoethane can be hazardous and difficult to handle on a large scale, posing significant safety risks and logistical challenges for supply chain heads. Additionally, many conventional methods suffer from limited substrate compatibility, requiring extensive optimization for different functional groups, which increases development time and cost. The reliance on direct carbon monoxide gas in some carbonylation reactions further complicates the process due to the toxicity and handling requirements of CO, necessitating specialized equipment that increases capital expenditure.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a sophisticated yet operationally simple palladium-catalyzed system that generates carbon monoxide in situ from formic acid and acetic anhydride. This eliminates the need for handling toxic CO gas directly, significantly enhancing workplace safety and reducing regulatory burdens associated with hazardous gas storage. The reaction proceeds efficiently at a mild temperature of 30°C, which is a substantial improvement over high-temperature protocols that can lead to decomposition of sensitive functional groups. As illustrated in the general reaction scheme below, the convergence of trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts creates a versatile platform for constructing the imidazole core with high precision.
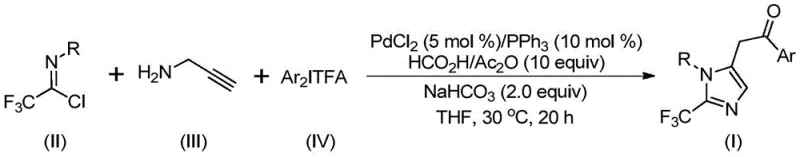
This methodology not only simplifies the operational workflow but also ensures high reaction efficiency and excellent substrate tolerance, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates. The ability to use commercially available catalysts like palladium chloride and triphenylphosphine further lowers the barrier to entry for adoption, allowing manufacturers to implement this technology without significant retooling of existing infrastructure.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation involves a intricate sequence of organometallic steps initiated by the formation of an intermolecular carbon-nitrogen bond promoted by alkali to yield a trifluoroacetamidine compound. Following isomerization, the palladium-catalyzed alkyne amine undergoes palladation to generate a key alkenyl palladium intermediate, which subsequently isomerizes to an alkyl palladium species. This intermediate then participates in a carbonylation reaction driven by the carbon monoxide released from the formic acid and acetic anhydride mixture, forming a reactive acyl palladium intermediate. The cycle concludes with the oxidative addition of the diaryl iodonium salt to form a tetravalent palladium intermediate, followed by reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst.
Understanding this mechanism is crucial for R&D directors focused on impurity control, as the specific sequence of bond formations minimizes side reactions typically associated with free radical pathways. The use of sodium bicarbonate as a base helps neutralize acidic byproducts, maintaining a stable pH environment that preserves the integrity of the sensitive imidazole ring during formation. Moreover, the choice of tetrahydrofuran (THF) as the preferred organic solvent ensures sufficient dissolution of all reactants, facilitating effective collisions between the catalytic species and substrates. This precise control over the reaction environment results in high conversion rates and simplifies the downstream purification process, ultimately delivering high-purity OLED material precursors or drug candidates with minimal impurity profiles.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, manufacturers should adhere to the optimized molar ratios and conditions specified in the patent data. The process begins by combining the palladium catalyst, ligand, and additives in the chosen solvent before introducing the core building blocks. Maintaining the reaction temperature at 30°C for a duration of 16 to 24 hours is critical to ensuring complete conversion while preventing thermal degradation of the product. The detailed standardized synthesis steps, including specific workup procedures like filtration and silica gel mixing, are outlined in the guide below to ensure reproducibility and quality consistency across batches.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture.
- Stir the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The reliance on cheap and easily obtainable starting materials, such as aromatic amines and alkynylamines, mitigates the risk of raw material shortages and price volatility often seen with exotic reagents. Furthermore, the elimination of hazardous gases and the use of mild reaction conditions translate directly into lower operational costs and reduced insurance premiums, contributing to a more resilient and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The use of inexpensive palladium chloride instead of more exotic noble metal catalysts, combined with the avoidance of high-pressure equipment for carbon monoxide, leads to significant capital and operational expenditure savings. The high atom economy of the multicomponent reaction ensures that a larger proportion of raw materials end up in the final product, minimizing waste disposal costs and maximizing resource utilization efficiency.
- Enhanced Supply Chain Reliability: Since the key reagents like trifluoroethylimidoyl chloride and diaryl iodonium salts can be synthesized from widely available precursors, the supply chain is less vulnerable to disruptions caused by single-source dependencies. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demands without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The mild temperature profile and the use of common organic solvents like THF facilitate straightforward scale-up from gram to kilogram and ton scales without the need for complex engineering controls. Additionally, the simplified post-treatment process involving standard column chromatography reduces the generation of hazardous waste streams, aligning with increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a reliable basis for decision-making.
Q: What are the key advantages of this Pd-catalyzed method over traditional imidazole synthesis?
A: This method operates under mild conditions (30°C) using inexpensive catalysts and avoids hazardous carbon monoxide gas by using formic acid/acetic anhydride as a CO source, significantly improving safety and operational simplicity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial large-scale production due to the use of cheap, readily available starting materials and simple post-processing.
Q: What is the substrate scope for the aryl groups in this reaction?
A: The reaction demonstrates excellent compatibility with various functional groups, including methyl, tert-butyl, chlorine, bromine, trifluoromethyl, and nitro substituents on both the imidoyl chloride and the iodonium salt components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-catalyzed synthesis for producing high-value heterocyclic intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole delivered meets the highest industry standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
