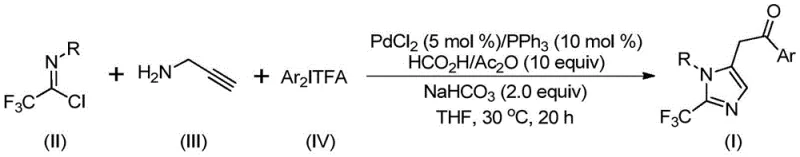
Scalable Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Pharmaceutical Applications
Scalable Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Pharmaceutical Applications
The landscape of modern pharmaceutical synthesis is constantly evolving to meet the demands for higher purity, greater structural diversity, and more sustainable manufacturing processes. A significant breakthrough in this domain is documented in Chinese Patent CN111423381B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. These heterocyclic scaffolds are pivotal in medicinal chemistry, known for enhancing the bioavailability, metabolic stability, and lipophilicity of drug candidates. The introduction of the trifluoromethyl group into the imidazole core is particularly challenging yet rewarding, as it imparts unique electronic properties that are crucial for binding affinity in biological targets. This novel methodology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. By operating under mild conditions at 30°C, this process represents a paradigm shift from energy-intensive traditional routes, offering a robust platform for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant technical and economic hurdles. Conventional methods often rely on the direct reaction of synthons with trifluoromethyl groups, such as trifluorodiazoethane or specialized trifluoroethylimidoyl halides, which can be unstable, hazardous, or prohibitively expensive to source on a large scale. Many existing protocols require harsh reaction conditions, including elevated temperatures and pressures, which not only increase energy consumption but also pose safety risks in a commercial manufacturing environment. Furthermore, traditional approaches frequently suffer from poor substrate compatibility, limiting the ability to introduce diverse functional groups necessary for structure-activity relationship (SAR) studies in drug discovery. The reliance on high-pressure carbon monoxide gas in some carbonylation reactions adds another layer of complexity regarding equipment requirements and safety protocols, making scale-up difficult and costly for many fine chemical manufacturers.
The Novel Approach
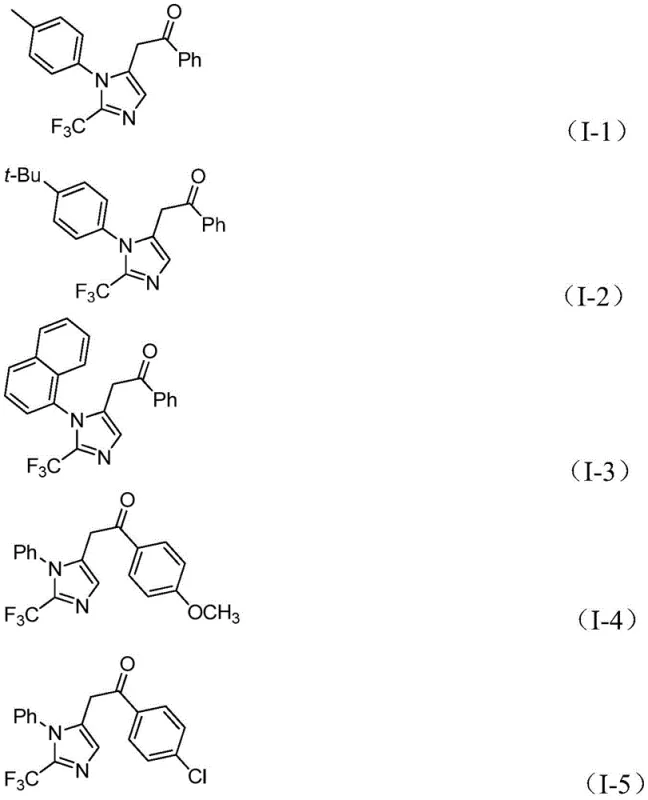
In stark contrast to these legacy techniques, the method described in patent CN111423381B offers a streamlined and versatile solution. By employing a palladium-catalyzed system with diaryl iodonium salts and a formic acid/acetic anhydride mixture as a CO source, the reaction proceeds efficiently at a remarkably low temperature of 30°C. This mild thermal profile drastically reduces energy costs and simplifies reactor requirements, facilitating easier heat management during exothermic phases. The use of propargylamine and trifluoroethylimidoyl chloride as building blocks allows for the construction of the imidazole ring with high atom economy. The reaction demonstrates exceptional functional group tolerance, accommodating substituents such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, and nitro groups on the aryl rings without significant loss in yield. This versatility enables the rapid generation of diverse libraries of 2-trifluoromethyl imidazoles, accelerating the lead optimization phase for R&D teams.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The success of this synthetic route lies in its intricate yet efficient catalytic cycle, which orchestrates the formation of multiple bonds in a single pot. The mechanism initiates with the formation of a trifluoroacetamidine compound through an intermolecular carbon-nitrogen bond promoted by the alkali additive, sodium bicarbonate. Following isomerization, the palladium catalyst engages with the alkyne moiety of the propargylamine derivative, undergoing palladation to generate a key alkenyl palladium intermediate. This species then isomerizes to an alkyl palladium intermediate, positioning the metal center for the subsequent carbonylation step. Crucially, the carbon monoxide required for this insertion is generated in situ from the decomposition of formic acid activated by acetic anhydride, eliminating the need for external CO gas cylinders. The resulting acyl palladium intermediate then undergoes oxidative addition with the diaryl iodonium salt, forming a high-valent tetravalent palladium species. Finally, reductive elimination releases the desired 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst, closing the loop.
From an impurity control perspective, this mechanism offers distinct advantages. The use of diaryl iodonium salts as arylating agents is known for their high reactivity and clean byproduct profiles, typically yielding iodobenzene derivatives that are easily separated during workup. The mild reaction temperature of 30°C minimizes thermal degradation of sensitive functional groups and suppresses side reactions such as polymerization of the alkyne or hydrolysis of the imidoyl chloride. Furthermore, the specific stoichiometry of the catalyst system—using palladium chloride and triphenylphosphine in a optimized ratio—ensures high turnover numbers while preventing the formation of palladium black, which can sequester the catalyst and reduce yield. The post-treatment process involving filtration and silica gel mixing followed by column chromatography ensures that the final high-purity pharmaceutical intermediate meets stringent quality specifications, with residual metal levels kept to a minimum.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized protocol to maximize yield and purity. The process begins with the preparation of the catalyst system in an anhydrous organic solvent, preferably tetrahydrofuran (THF), which has been shown to provide superior solubility and reaction kinetics compared to other solvents. The careful addition of reagents in the specified molar ratios is critical; specifically, maintaining a slight excess of the trifluoroethylimidoyl chloride relative to the propargylamine helps drive the reaction to completion. The reaction time is typically between 16 to 24 hours, a window that balances complete conversion with operational efficiency. Detailed standardized synthetic steps, including specific quenching procedures and purification parameters, are outlined below to ensure reproducibility across different batches.
- Prepare the reaction mixture by adding palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate (2.0 equiv), acetic anhydride, and formic acid (10 equiv) into an organic solvent such as THF.
- Introduce the substrates: trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt into the Schlenk tube containing the catalyst system.
- Stir the mixture at 30°C for 16 to 24 hours. Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material supply chain. The key starting materials—aromatic amines, alkynylamines, formic acid, acetic anhydride, and palladium chloride—are commodity chemicals available from multiple global vendors, reducing the risk of supply disruption associated with proprietary or exotic reagents. This abundance ensures price stability and allows for competitive sourcing strategies. Moreover, the elimination of high-pressure gas handling equipment reduces capital expenditure (CAPEX) for new production lines and lowers maintenance costs for existing facilities. The mild reaction conditions also imply lower utility costs, as there is no need for extensive heating or cooling infrastructure, contributing to a leaner manufacturing overhead.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of several factors. First, the use of inexpensive palladium chloride instead of more exotic ligand-heavy catalyst systems significantly lowers the catalyst cost per kilogram of product. Second, the in situ generation of carbon monoxide removes the logistical and safety costs associated with storing and transporting high-pressure CO gas. Third, the high reaction efficiency and substrate compatibility mean that fewer purification steps are required, reducing solvent consumption and waste disposal costs. By streamlining the synthesis into a one-pot procedure, labor hours are minimized, and overall throughput is increased, leading to substantial cost savings in the final API manufacturing budget.
- Enhanced Supply Chain Reliability: In the volatile landscape of global chemical trade, reliability is paramount. This synthesis route enhances supply chain resilience by relying on widely available bulk chemicals rather than niche intermediates that might be subject to export controls or single-source bottlenecks. The robustness of the reaction against variations in substrate electronics (e.g., electron-withdrawing vs. electron-donating groups) means that suppliers can switch between different aryl amine sources without needing to re-optimize the entire process. This flexibility allows for rapid response to market demands and ensures continuous production even if a specific raw material grade fluctuates in availability. The scalability from gram to multi-ton levels further guarantees that the supply can grow in lockstep with the clinical or commercial success of the downstream drug product.
- Scalability and Environmental Compliance: As regulatory scrutiny on chemical manufacturing intensifies, the environmental profile of a process becomes a key differentiator. This method scores highly on green chemistry metrics due to its atom economy and the avoidance of toxic reagents. The use of sodium bicarbonate as a mild base generates benign byproducts, and the solvent system (THF) is recyclable. The absence of heavy metal waste streams, beyond the recoverable palladium catalyst, simplifies wastewater treatment and reduces the environmental footprint. From a scalability standpoint, the exothermic nature of the reaction is manageable at 30°C, allowing for safe operation in large-scale reactors without the risk of thermal runaway. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, ensuring compliance with increasingly strict environmental, health, and safety (EHS) regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method operates under exceptionally mild conditions (30°C) compared to harsh traditional methods. It utilizes cheap and readily available starting materials like propargylamine and trifluoroacetimidoyl chloride, offering superior substrate compatibility and functional group tolerance.
Q: What is the role of formic acid and acetic anhydride in this reaction?
A: Formic acid and acetic anhydride serve as the carbon monoxide (CO) surrogate. They generate CO in situ, which is essential for the carbonylation step that constructs the imidazole ring backbone without requiring high-pressure CO gas cylinders.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the patent explicitly states the method is suitable for gram-level expansion and provides possibilities for industrial large-scale production due to its simple operation, high reaction efficiency, and use of stable, commercially available reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
The technological potential of this palladium-catalyzed carbonylation route is immense, offering a pathway to high-value heterocyclic building blocks that are essential for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs and analytical instrumentation to guarantee stringent purity specifications for every batch of 2-trifluoromethyl imidazole delivered. We understand the critical nature of timeline and quality in the pharmaceutical industry, and our dedicated team is committed to maintaining the highest standards of operational excellence.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your bill of materials. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Together, we can accelerate the delivery of life-saving medicines to the market through superior chemical innovation.
