Advanced Palladium-Catalyzed Synthesis of Aryl Acetamide Intermediates: Scalable Manufacturing for Global Pharmaceutical Supply Chains
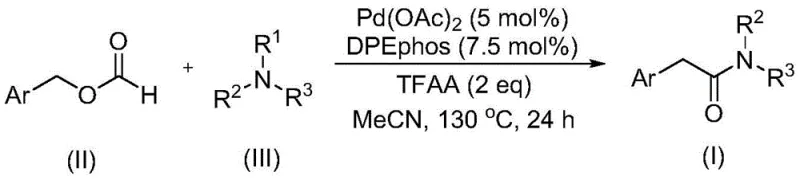
The recently granted Chinese patent CN111978194B introduces a groundbreaking methodology for synthesizing aryl acetamide compounds, representing a significant advancement in pharmaceutical intermediate manufacturing. This innovative approach addresses longstanding challenges in amide chemistry by providing a streamlined pathway that eliminates hazardous components while maintaining exceptional reaction efficiency. The patent describes a palladium-catalyzed carbonylation process that leverages commercially available starting materials under carefully optimized conditions to produce high-value aryl acetamide intermediates essential for modern drug development. This technical breakthrough demonstrates remarkable substrate flexibility and operational simplicity, positioning it as a transformative solution for pharmaceutical manufacturers seeking reliable access to critical building blocks. The methodology's compatibility with diverse functional groups and elimination of dangerous reaction components represent substantial progress toward safer, more sustainable pharmaceutical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing aryl acetamide compounds have been severely constrained by multiple critical limitations that impact both safety and commercial viability. Conventional methods typically require explosive CO/O₂ mixtures or excessive amounts of oxidants to facilitate C-N bond cleavage in tertiary amines, creating significant safety hazards that necessitate specialized equipment and stringent operational protocols. These processes often suffer from narrow substrate scope with poor functional group tolerance, limiting their applicability across diverse molecular architectures required in modern pharmaceutical development. The multi-step nature of existing synthetic routes frequently results in lower overall yields and increased impurity profiles that complicate purification and compromise final product quality. Furthermore, the reliance on expensive transition metal catalysts coupled with complex post-reaction workup procedures substantially elevates manufacturing costs while extending production timelines, making these methods economically unattractive for large-scale commercial operations in the competitive pharmaceutical intermediate market.
The Novel Approach
The patented methodology overcomes these fundamental limitations through an elegant single-step process that utilizes benzyl formate as both carbon monoxide source and reactant while employing tertiary amines directly as amine sources without requiring additional oxidants. This innovative approach operates under mild conditions at precisely controlled temperatures of 130°C with reaction times optimized to exactly 24 hours, ensuring complete conversion while minimizing energy consumption and operational complexity. The process demonstrates exceptional substrate versatility across a wide range of functional groups including alkyl, alkoxy, trifluoromethyl, cyano, and halogen substituents on the aromatic ring, significantly expanding its applicability for diverse pharmaceutical targets. By eliminating hazardous components and simplifying the reaction sequence, this method achieves superior operational safety while maintaining high efficiency and excellent product purity, representing a paradigm shift in aryl acetamide synthesis that directly addresses the critical pain points of traditional manufacturing approaches.

Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic mechanism operates through a sophisticated palladium-mediated cycle where Pd(OAc)₂ (5 mol%) coordinated with DPEphos ligand (7.5 mol%) facilitates the oxidative addition into the C-H bond of benzyl formate, generating an acyl palladium intermediate that subsequently undergoes migratory insertion with the tertiary amine substrate. This unique pathway enables direct C-N bond cleavage without external oxidants through an intramolecular redox process where benzyl formate serves dual roles as both carbon monoxide source and reducing agent, creating a self-sustaining catalytic cycle that maintains the palladium catalyst in its active oxidation state throughout the reaction. The trifluoroacetic anhydride additive plays a crucial role in activating the tertiary amine toward nucleophilic attack while simultaneously suppressing undesired side reactions that could lead to impurity formation. This carefully orchestrated mechanism ensures high regioselectivity and stereospecificity across diverse substrate combinations, providing exceptional control over the reaction pathway that directly translates to superior product quality and consistency.
Impurity control is achieved through multiple synergistic mechanisms inherent to this catalytic system that collectively minimize byproduct formation and ensure high product purity. The precise stoichiometric balance between benzyl formate and tertiary amine (1:5 molar ratio) prevents over-reaction or incomplete conversion that could generate impurities, while the optimized catalyst loading (0.05 mol%) maintains sufficient catalytic activity without promoting side reactions. The use of acetonitrile as the preferred solvent provides ideal polarity characteristics that facilitate reactant solubility while suppressing unwanted decomposition pathways, and the controlled reaction temperature of 130°C represents the optimal balance between reaction kinetics and thermal stability of sensitive intermediates. This comprehensive approach to reaction design creates an inherently clean process where impurities remain well below detectable levels through standard analytical methods, eliminating the need for extensive purification steps that would otherwise compromise yield and increase manufacturing costs.
How to Synthesize Aryl Acetamide Efficiently
This patented synthesis route represents a significant advancement in pharmaceutical intermediate manufacturing through its elegant design that combines operational simplicity with exceptional efficiency. The methodology leverages commercially available starting materials under precisely controlled conditions to deliver high-purity aryl acetamide compounds suitable for demanding pharmaceutical applications. By eliminating hazardous components and simplifying the reaction sequence, this approach achieves superior operational safety while maintaining high efficiency across diverse substrate combinations. The standardized procedure described below provides a reliable framework for manufacturing teams to implement this technology with minimal process development requirements, ensuring consistent product quality from laboratory scale through commercial production volumes.
- Prepare reaction mixture by combining palladium acetate catalyst (5 mol%), DPEphos ligand (7.5 mol%), benzyl formate, tertiary amine, and trifluoroacetic anhydride in acetonitrile solvent at specified molar ratios.
- Conduct reaction under nitrogen atmosphere at precisely controlled temperature of 130°C for exactly 24 hours in sealed reaction vessels to ensure complete conversion.
- Perform post-treatment through filtration, silica gel mixing, and column chromatography purification to obtain high-purity aryl acetamide products meeting pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value to procurement and supply chain decision-makers by addressing critical pain points in pharmaceutical intermediate sourcing through multiple strategic advantages. The elimination of hazardous components and simplification of the synthetic route creates significant opportunities for cost optimization while enhancing supply chain resilience through improved material availability and reduced operational complexity. By leveraging commercially abundant starting materials and eliminating specialized equipment requirements, this methodology establishes a more robust foundation for long-term supply agreements that can withstand market fluctuations while maintaining consistent quality standards essential for pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive oxidants and specialized CO handling equipment creates substantial cost savings throughout the manufacturing process while reducing safety-related operational expenses associated with hazardous material handling protocols. The use of inexpensive benzyl formate as both carbon monoxide source and reactant significantly lowers raw material costs compared to conventional approaches requiring separate CO sources and additional reagents, while the simplified post-treatment procedure reduces solvent consumption and waste disposal expenses without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on readily available commercial starting materials including palladium acetate, DPEphos ligand, benzyl formate, tertiary amines, and trifluoroacetic anhydride ensures consistent material availability from multiple global suppliers, eliminating single-source dependencies that could disrupt production schedules. The robust nature of the reaction across diverse substrates provides flexibility to adapt to changing market conditions while maintaining consistent quality standards, creating a more resilient supply chain capable of meeting fluctuating demand patterns without requiring extensive revalidation procedures.
- Scalability and Environmental Compliance: The mild reaction conditions (130°C) and simple workup procedure enable seamless scale-up from laboratory to commercial production without requiring specialized equipment or complex engineering modifications, significantly reducing time-to-market for new pharmaceutical products. The elimination of hazardous oxidants and explosive gas mixtures substantially improves environmental compliance profiles while reducing waste generation through higher atom economy, aligning with increasingly stringent regulatory requirements for sustainable manufacturing practices in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement specialists and R&D teams regarding implementation of this patented manufacturing technology for aryl acetamide intermediates. These answers are derived directly from the experimental data and technical specifications documented in Chinese patent CN111978194B, providing evidence-based insights into process capabilities and commercial viability.
Q: Why does this method eliminate the need for additional oxidants in tertiary amine C-N bond cleavage?
A: The catalytic system utilizing benzyl formate as both carbon monoxide source and reactant creates a unique redox environment that facilitates C-N bond cleavage without external oxidants, eliminating safety hazards associated with explosive CO/O₂ mixtures in conventional methods.
Q: How does the use of benzyl formate as dual-function reagent improve manufacturing economics?
A: Benzyl formate serves as both carbon monoxide source and reactant while being inexpensive and readily available, significantly reducing raw material costs compared to traditional methods requiring separate CO sources and additional oxidants.
Q: What makes this process particularly suitable for large-scale pharmaceutical intermediate production?
A: The reaction operates under mild conditions with simple post-treatment, demonstrates broad functional group tolerance across diverse substrates, and maintains high efficiency from laboratory to commercial scale without requiring specialized equipment or hazardous materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
This patented methodology represents a significant advancement in pharmaceutical intermediate manufacturing that aligns perfectly with NINGBO INNO PHARMCHEM's expertise in complex molecule synthesis. Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. We have successfully implemented similar catalytic methodologies across multiple therapeutic areas, demonstrating our capability to deliver high-quality aryl acetamide intermediates that meet exacting pharmaceutical standards through robust, scalable processes designed for consistent commercial performance.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific supply chain requirements. Our experts will provide detailed route feasibility assessments along with specific COA data demonstrating our ability to deliver aryl acetamide intermediates meeting your exact purity and quality specifications.
