Advanced Palladium-Catalyzed Synthesis of High-Purity Aryl Acetamides for Pharmaceutical Manufacturing Scalability
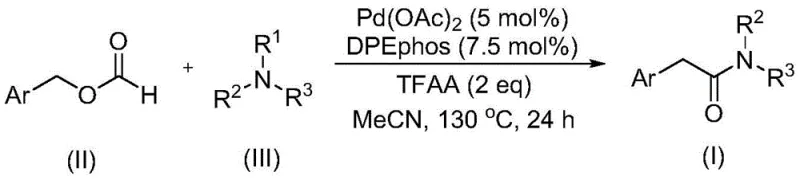
The recently granted Chinese patent CN111978194B introduces a groundbreaking methodology for synthesizing aryl acetamide compounds, which represent critical building blocks in pharmaceutical development due to their prevalence in bioactive molecules and drug candidates across multiple therapeutic areas. This innovative process employs a palladium-catalyzed carbonylation strategy that uniquely utilizes benzyl formate as both a carbon monoxide source and reactant while leveraging tertiary amines as amine precursors, thereby eliminating the need for hazardous oxidants that have plagued conventional approaches for decades. The methodology demonstrates exceptional substrate flexibility with tolerance for diverse functional groups including alkyl, alkoxy, trifluoromethyl, cyano, and halogen substituents across various aromatic systems from phenyl to naphthyl frameworks. Notably, the reaction proceeds efficiently at precisely controlled 130°C for exactly 24 hours in acetonitrile solvent using commercially available palladium acetate and DPEphos ligand at optimized stoichiometric ratios of 5 mol% and 7.5 mol% respectively. This advancement addresses long-standing challenges in amide synthesis by providing a safer, more sustainable pathway that maintains high reaction efficiency while significantly simplifying downstream processing requirements through elimination of explosive CO/O2 mixtures required in traditional methods. The practical implications for pharmaceutical manufacturers are substantial, as this method enables access to complex aryl acetamide intermediates with improved process economics and enhanced safety profiles compared to conventional methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to aryl acetamide synthesis predominantly rely on amidation reactions of phenylacetic acid derivatives or transition metal-catalyzed carbonylation processes that require hazardous oxidants and specialized handling procedures due to explosive CO/O2 mixtures under elevated temperatures. These conventional methodologies suffer from significant limitations including narrow substrate scope where sensitive functional groups necessitate extensive protection/deprotection strategies that dramatically increase both cost and processing time while reducing overall yield. The requirement for stoichiometric amounts of strong oxidants creates substantial safety hazards requiring specialized infrastructure and trained personnel, thereby increasing capital expenditure and operational complexity while introducing potential regulatory compliance challenges during scale-up. Furthermore, these processes often generate complex impurity profiles that necessitate multiple purification steps to achieve pharmaceutical-grade purity specifications, resulting in significant material loss and extended production timelines that directly impact supply chain reliability for time-sensitive drug development programs. The inherent limitations of conventional approaches have constrained access to structurally diverse aryl acetamide compounds despite their demonstrated importance in pharmaceutical applications ranging from CNS agents to anti-infectives.
The Novel Approach
The patented methodology overcomes these critical limitations through an elegant design where benzyl formate serves dual functionality as both carbon monoxide source and reactant while tertiary amines function as direct amine precursors without requiring additional oxidation steps. This innovative approach eliminates hazardous oxidants entirely while maintaining high reaction efficiency across a broad spectrum of aromatic substrates including those bearing electron-donating and electron-withdrawing substituents at various positions on the aromatic ring. The process operates under mild conditions using commercially available palladium acetate catalyst with DPEphos ligand at precisely controlled stoichiometric ratios that optimize both conversion and selectivity while minimizing metal contamination in the final product. Crucially, the methodology demonstrates exceptional functional group tolerance that accommodates halogens, cyano groups, trifluoromethyl moieties, and various alkyl/alkoxy substituents without requiring protective groups or specialized handling procedures. This streamlined approach significantly reduces both capital and operational costs while enhancing process safety profiles through elimination of explosive gas mixtures and hazardous reagents typically required in conventional carbonylation methodologies.
Mechanistic Insights into Palladium-Catalyzed Aryl Acetamide Synthesis
The catalytic cycle begins with oxidative addition of benzyl formate to the palladium(0) species generated in situ from palladium acetate reduction, followed by decarbonylation to form a palladium-formyl intermediate that subsequently undergoes migratory insertion with the tertiary amine substrate. This key step facilitates C-N bond cleavage through a concerted mechanism where trifluoroacetic anhydride activates the amine group without requiring external oxidants, enabling direct formation of the iminium ion intermediate that subsequently reacts with the acyl-palladium species to form the final amide product. The precise coordination geometry provided by the DPEphos ligand stabilizes critical intermediates throughout the catalytic cycle while preventing undesired β-hydride elimination pathways that would lead to reduced yields or side products. This mechanistic pathway represents a significant advancement over prior art by eliminating the need for stoichiometric oxidants typically required for C-N bond activation in tertiary amines, thereby creating a more atom-economical process with inherently cleaner reaction profiles. The absence of additional oxidation steps also prevents over-oxidation side reactions that commonly generate difficult-to-remove impurities in conventional methodologies.
The reaction design inherently minimizes impurity formation through several key mechanisms: first, the controlled decarbonylation of benzyl formate provides precise CO delivery without excess gas generation; second, the use of trifluoroacetic anhydride as both activator and solvent component creates a homogeneous reaction environment that prevents localized concentration gradients; third, the optimized temperature profile of 130°C maintains sufficient energy for reaction progression while avoiding thermal decomposition pathways that would generate byproducts. This impurity control strategy results in cleaner crude reaction mixtures that require minimal purification steps compared to conventional methods where multiple chromatographic separations are typically necessary to achieve pharmaceutical-grade purity specifications. The methodology consistently delivers products meeting stringent quality requirements across diverse substrate classes as demonstrated by comprehensive analytical data including NMR characterization of multiple representative compounds.

How to Synthesize Aryl Acetamides Efficiently
This patented methodology represents a significant advancement in aryl acetamide synthesis through its innovative use of commercially available starting materials under optimized catalytic conditions that eliminate hazardous reagents while maintaining high efficiency across diverse substrate classes. The process demonstrates exceptional practicality for pharmaceutical manufacturing due to its straightforward implementation using standard laboratory equipment without requiring specialized infrastructure typically associated with carbonylation chemistry involving pressurized carbon monoxide gas. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in the patent literature, ensuring consistent results across different production scales while maintaining strict quality control parameters essential for pharmaceutical applications. The following section provides step-by-step guidance for implementing this efficient synthesis route.
- Combine palladium acetate catalyst (5 mol%), DPEphos ligand (7.5 mol%), trifluoroacetic anhydride (2 equivalents), benzyl formate, and tertiary amine in acetonitrile solvent under inert atmosphere with precise stoichiometric ratios.
- Heat the reaction mixture to 130°C and maintain at this temperature for 24 hours with continuous stirring to ensure complete conversion while monitoring reaction progress through standard analytical techniques.
- Perform post-treatment by filtration, silica gel mixing, and column chromatography purification to isolate high-purity aryl acetamide product with stringent quality control verification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits specifically addressing critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing organizations seeking reliable access to high-quality intermediates. The elimination of hazardous oxidants not only reduces material costs but also significantly lowers associated safety infrastructure requirements and regulatory compliance burdens that typically increase both capital expenditure and operational complexity during scale-up activities. By utilizing readily available starting materials with established global supply chains, this approach minimizes procurement risks while providing greater flexibility in sourcing strategies compared to conventional methods dependent on specialized or restricted reagents that may face supply constraints during market volatility.
- Cost Reduction in Manufacturing: The elimination of additional oxidants represents a significant cost-saving opportunity by removing both the material expense of these hazardous reagents and the associated safety infrastructure required for their handling and disposal procedures. Furthermore, the use of inexpensive and readily available starting materials such as benzyl formate and tertiary amines creates substantial economic advantages over alternative synthetic routes that depend on more expensive or specialized reagents requiring complex handling protocols. The simplified reaction profile also reduces operational complexity by eliminating multiple purification steps typically needed to remove oxidant-derived impurities, thereby lowering labor costs and minimizing potential production delays caused by extended processing requirements.
- Enhanced Supply Chain Reliability: The reliance on globally available commodity chemicals with established supply networks significantly reduces procurement risks compared to methodologies requiring specialized or restricted materials that may face availability constraints during market disruptions. Benzyl formate and tertiary amines are produced by multiple global suppliers with redundant manufacturing capabilities across different geographic regions, providing procurement teams with greater flexibility in sourcing strategies while maintaining consistent quality standards through well-established quality control protocols. This robust supply chain foundation enables reliable delivery schedules even during periods of market volatility while supporting just-in-time manufacturing approaches preferred by modern pharmaceutical operations.
- Scalability and Environmental Compliance: The straightforward reaction protocol operating under standard pressure conditions without hazardous gas handling requirements enables seamless scale-up from laboratory development directly to commercial production without requiring specialized equipment modifications typically needed for pressurized CO systems. This simplified engineering profile significantly reduces capital expenditure during plant implementation while accelerating time-to-market for new pharmaceutical products requiring these critical intermediates. Additionally, the elimination of hazardous oxidants substantially reduces environmental impact by minimizing waste streams containing toxic metal residues or explosive byproducts that require specialized treatment procedures under current environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address specific technical concerns raised by procurement specialists and R&D professionals regarding implementation of this patented methodology within pharmaceutical manufacturing environments. These answers are derived directly from experimental data documented in patent CN111978194B and reflect practical considerations observed during extensive laboratory validation studies across multiple substrate classes.
Q: Why does this method eliminate the need for additional oxidants in tertiary amine C-N bond cleavage?
A: The patent demonstrates that benzyl formate serves as both carbon monoxide source and reactant while trifluoroacetic anhydride facilitates the C-N bond cleavage through an innovative mechanistic pathway that avoids hazardous oxidants required in conventional methods.
Q: How does the broad functional group tolerance benefit pharmaceutical manufacturers?
A: The methodology accommodates diverse substituents including alkyl, alkoxy, trifluoromethyl, cyano, and halogen groups across various aromatic systems without requiring specialized protection/deprotection steps, significantly expanding accessible compound space for drug discovery.
Q: What supply chain advantages does using commercially available starting materials provide?
A: Benzyl formate and tertiary amines are inexpensive, widely accessible reagents that eliminate dependency on specialized or hazardous materials, thereby enhancing supply chain resilience and reducing procurement complexities for global pharmaceutical operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at parts-per-million levels. As a trusted partner to leading global pharmaceutical companies, we have successfully implemented this patented methodology across multiple production scales while ensuring consistent quality through comprehensive process validation protocols that meet all regulatory requirements across major markets worldwide. Our technical team works closely with client R&D departments to optimize synthesis routes while maintaining full intellectual property confidentiality throughout all development phases.
We invite you to request our Customized Cost-Saving Analysis which demonstrates specific economic benefits achievable through implementation of this innovative methodology for your particular compound requirements. Contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your manufacturing needs.
