Revolutionizing Aryl Acetamide Production: A Safe, Scalable Pd-Catalyzed Carbonylation Strategy for Global Markets
Revolutionizing Aryl Acetamide Production: A Safe, Scalable Pd-Catalyzed Carbonylation Strategy for Global Markets
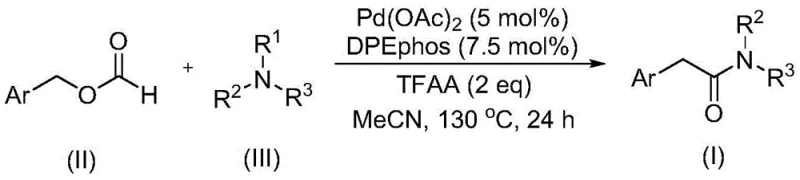
The landscape of organic synthesis is constantly evolving, driven by the need for safer, more efficient, and cost-effective methodologies for producing high-value intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN111978194B, which discloses a novel preparation method for aryl acetamide compounds. This technology addresses long-standing challenges in amide synthesis, particularly the activation of tertiary amine C-N bonds, by utilizing a palladium-catalyzed carbonylation strategy. Unlike traditional methods that often rely on hazardous carbon monoxide gas or expensive activated acids, this innovative approach employs benzyl formate as a dual-purpose reagent, serving as both the carbon monoxide source and a reactant. For R&D directors and procurement managers seeking a reliable aryl acetamide supplier, this patent represents a pivotal shift towards greener and more economically viable manufacturing processes that do not compromise on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamide compounds has predominantly relied on the direct amidation of phenylacetic acid and its derivatives. While straightforward in concept, these conventional routes often suffer from significant drawbacks when applied to complex molecular architectures or when specific substitution patterns are required. Furthermore, transition metal-catalyzed carbonylation reactions have emerged as a powerful alternative; however, the majority of existing literature focuses on primary and secondary amines as nitrogen sources. The activation of tertiary amines remains a formidable challenge due to the strength and stability of the C-N bond, which typically resists cleavage under standard conditions. Existing methods that do attempt this transformation frequently necessitate the use of excessive oxidants or explosive mixtures of carbon monoxide and oxygen, posing severe safety hazards and complicating the regulatory compliance for industrial scale-up. These limitations create bottlenecks in the supply chain for high-purity aryl acetamides, driving up costs and extending lead times for pharmaceutical and agrochemical manufacturers.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in patent CN111978194B introduces a transformative solution by leveraging benzyl formate and tertiary amines in a palladium-catalyzed system. This approach elegantly bypasses the need for external oxidants for C-N bond breakage, a feature that significantly simplifies the reaction profile and enhances safety. By using benzyl formate, the process avoids the logistical nightmares and safety risks associated with handling high-pressure carbon monoxide gas, making it far more accessible for standard laboratory and production facilities. The reaction operates under relatively mild thermal conditions compared to high-pressure carbonylations, yet it maintains high efficiency and broad substrate tolerance. This novel pathway not only expands the chemical space accessible to synthetic chemists but also offers a robust platform for the cost reduction in pharmaceutical intermediate manufacturing by streamlining raw material sourcing and reaction engineering.
Mechanistic Insights into Pd-Catalyzed Carbonylation with Benzyl Formate
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst, the specialized ligand, and the unique reactivity of benzyl formate. The reaction mechanism likely involves the oxidative addition of the palladium species into the C-O bond of the benzyl formate, facilitated by the presence of trifluoroacetic anhydride (TFAA) which acts as an activator. The bidentate ligand, specifically bis(2-diphenylphosphinophenyl) ether (DPEphos), plays a critical role in stabilizing the palladium center and promoting the subsequent migratory insertion of the carbonyl moiety. This is followed by the nucleophilic attack of the tertiary amine and the crucial C-N bond cleavage step, which releases the aryl acetamide product and regenerates the catalytic cycle. The absence of additional oxidants suggests a redox-neutral or internally balanced cycle, which minimizes the formation of unwanted byproducts and simplifies the impurity profile. For R&D teams, understanding this mechanism is vital for optimizing reaction parameters and predicting the behavior of novel substrates, ensuring that the commercial scale-up of complex amide intermediates proceeds with minimal deviation.
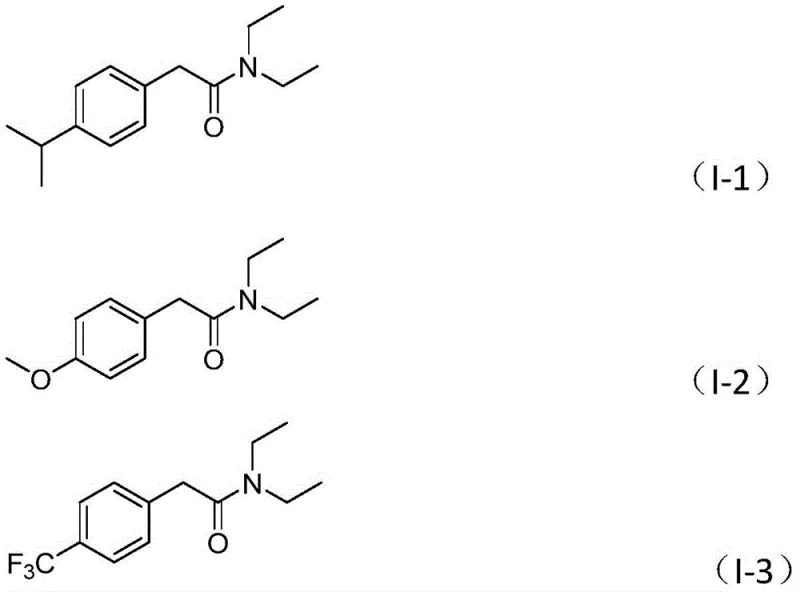
Furthermore, the impurity control mechanism inherent in this design is superior to traditional acid-chloride or coupling reagent methods. Since the carbonyl source is generated in situ from a stable liquid precursor, the risk of over-carbonylation or polymerization is significantly reduced. The specific choice of acetonitrile as the solvent further enhances the solubility of polar intermediates and facilitates the turnover of the catalyst. The patent data indicates that this system tolerates a wide range of functional groups on the aryl ring, including electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as trifluoromethyl and cyano groups. This broad compatibility ensures that the final product meets stringent purity specifications required for downstream applications in drug discovery and development, thereby reducing the burden on purification teams and lowering the overall cost of goods sold.
How to Synthesize Aryl Acetamide Efficiently
To implement this cutting-edge synthesis in a practical setting, operators must adhere to precise stoichiometric ratios and thermal profiles as outlined in the patent documentation. The process begins with the careful weighing of palladium acetate and the DPEphos ligand, which are then combined with trifluoroacetic anhydride, benzyl formate, and the chosen tertiary amine in a sealed reaction vessel. The use of a sealed tube is essential to maintain the integrity of the volatile components and ensure the reaction proceeds to completion without loss of material. While the general procedure is robust, attention to detail regarding the order of addition and the quality of the solvent can influence the final yield. For those seeking to replicate these results or adapt them for larger batches, the detailed standardized synthesis steps见下方的指南 provide a comprehensive roadmap for execution.
- Combine palladium acetate, DPEphos ligand, trifluoroacetic anhydride, benzyl formate, and tertiary amine in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C in a sealed tube and maintain this temperature for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target aryl acetamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that resonate deeply with procurement managers and supply chain heads who are tasked with optimizing margins and ensuring continuity. The primary driver of value is the substitution of hazardous and logistically complex reagents with benign, commodity chemicals. Benzyl formate and common tertiary amines like triethylamine are widely available in the global chemical market, ensuring a stable supply chain that is less susceptible to geopolitical disruptions or sudden price spikes. This accessibility translates directly into enhanced supply chain reliability, allowing manufacturers to plan production schedules with greater confidence and reducing the lead time for high-purity aryl acetamides. Moreover, the elimination of high-pressure carbon monoxide equipment removes a significant capital expenditure barrier, enabling smaller and mid-sized facilities to produce these valuable intermediates without massive infrastructure investments.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the use of inexpensive starting materials and the simplification of the reaction setup. By avoiding the need for specialized high-pressure reactors and the associated safety protocols required for carbon monoxide gas, the operational overhead is drastically simplified. Additionally, the high yields reported in the patent examples, ranging consistently above 70% and reaching up to 87% for optimized substrates, mean that less raw material is wasted per unit of product. This efficiency, combined with the straightforward post-treatment involving simple filtration and chromatography, results in substantial cost savings across the entire production lifecycle, making it a highly attractive option for cost-sensitive projects.
- Enhanced Supply Chain Reliability: In the volatile world of fine chemicals, the availability of raw materials is often the weakest link. This synthesis route relies on benzyl formate and tertiary amines, which are produced on a massive scale for other industries, ensuring a redundant and robust supply network. Unlike specialized coupling reagents that may have single-source suppliers, these commodities can be sourced from multiple vendors globally. This diversification mitigates the risk of supply interruptions and allows procurement teams to negotiate better pricing terms. Furthermore, the stability of these reagents allows for longer storage times without degradation, facilitating bulk purchasing strategies that further lock in lower costs and secure inventory levels against market fluctuations.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and emissions intensify, the environmental profile of a chemical process becomes a critical decision factor. This method generates fewer hazardous byproducts compared to traditional activation methods that produce stoichiometric amounts of salt waste. The absence of external oxidants and the use of a closed system for the carbonylation step minimize the release of volatile organic compounds and toxic gases. This cleaner profile simplifies the permitting process for new production lines and reduces the cost associated with waste treatment and disposal. Consequently, the process is inherently more scalable, allowing for a seamless transition from gram-scale laboratory optimization to multi-ton commercial production without encountering insurmountable environmental or safety hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the experimental data and claims within the patent specification. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific manufacturing needs. Understanding these nuances is essential for making informed decisions about process integration and vendor selection.
Q: Why is benzyl formate preferred over carbon monoxide gas in this synthesis?
A: Benzyl formate serves as a safe, liquid surrogate for carbon monoxide, eliminating the need for high-pressure gas equipment and reducing safety risks associated with toxic CO handling while acting as both the carbonyl source and a reactant.
Q: Can this method accommodate sterically hindered tertiary amines?
A: Yes, the patented process demonstrates excellent tolerance for various tertiary amines, including triethylamine and triisopropylamine, achieving high yields without requiring additional oxidants for C-N bond cleavage.
Q: What are the scalability advantages of this Pd-catalyzed route?
A: The use of commercially available reagents, mild reaction conditions relative to high-pressure carbonylation, and simple post-treatment procedures make this method highly suitable for commercial scale-up of complex amide intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN111978194B and have integrated similar advanced catalytic strategies into our own manufacturing portfolio. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale market supply. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical and fine chemical sectors, consistency is key, and our robust quality management systems are designed to deliver high-purity aryl acetamides that meet the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage these technical advancements for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to deep technical expertise and a flexible supply chain capable of adapting to your evolving needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can drive efficiency and value for your organization.
