Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Manufacturing
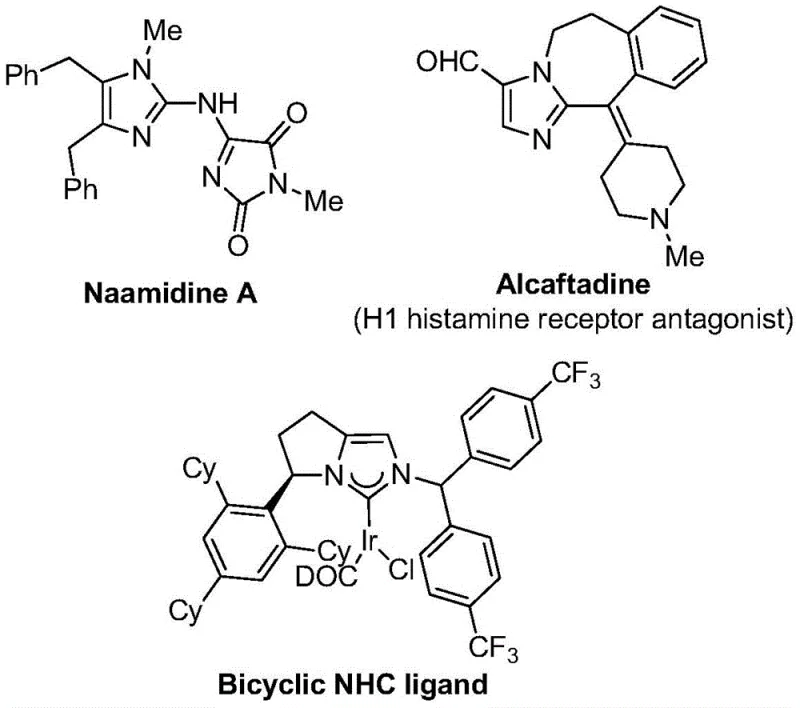
The introduction of trifluoromethyl groups into heterocyclic scaffolds is a cornerstone strategy in modern medicinal chemistry, profoundly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As detailed in the groundbreaking patent CN111423381B, a novel and highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds has been developed, addressing critical bottlenecks in the synthesis of these valuable pharmacophores. Imidazole derivatives are ubiquitous in biologically active molecules, serving as key structural motifs in drugs like Naamidine A and receptor antagonists such as Alcaftadine, as well as in advanced ligand systems for coordination catalysis. The ability to install a trifluoromethyl group at the 2-position of the imidazole ring with high precision and under mild conditions represents a significant technological leap for the pharmaceutical industry.
This patented methodology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtainable starting materials including trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. For R&D directors and process chemists seeking robust routes for high-purity pharmaceutical intermediates, this technology offers a compelling alternative to legacy methods that often suffer from harsh conditions or limited substrate scope. The reaction operates at a remarkably low temperature of 30°C, ensuring energy efficiency and safety, while delivering high yields across a diverse range of substrates. This report analyzes the technical merits and commercial implications of this synthesis, positioning it as a vital tool for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the direct reaction of synthons containing the trifluoromethyl group with suitable substrates. Common trifluoromethylating agents such as trifluorodiazoethane, while effective, present significant challenges in terms of safety, stability, and cost. These reagents can be hazardous to handle on a large scale due to their potential explosivity and toxicity, necessitating specialized equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, many conventional methods exhibit poor functional group tolerance, limiting the structural diversity of the resulting imidazole libraries. The inability to efficiently incorporate electron-withdrawing or sterically hindered substituents often forces medicinal chemists to resort to lengthy multi-step syntheses, thereby increasing lead times and reducing the overall agility of the drug discovery process.
The Novel Approach
In stark contrast to these traditional limitations, the method disclosed in patent CN111423381B introduces a streamlined, one-pot strategy that fundamentally reshapes the synthetic landscape for 2-trifluoromethyl imidazoles. By employing trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the core building blocks, the process achieves a complex molecular assembly through a sophisticated palladium-catalyzed cascade. The general reaction scheme illustrates how these three distinct components converge to form the target imidazole scaffold with high atom economy.
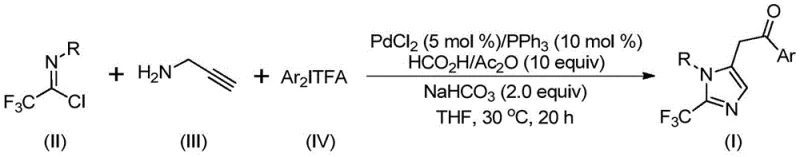
This novel approach not only simplifies the operational workflow but also dramatically expands the chemical space accessible to researchers. The compatibility with a wide array of substituents on both the imidoyl chloride and the diaryl iodonium salt allows for the rapid generation of diverse analogues, facilitating structure-activity relationship (SAR) studies. For procurement managers focused on cost reduction in API manufacturing, the use of commercially available and inexpensive reagents like palladium chloride and triphenylphosphine, rather than exotic ligands or precious metal complexes, translates directly into lower raw material costs. The mild reaction temperature of 30°C further underscores the economic and environmental advantages, reducing energy consumption and minimizing the formation of thermal degradation byproducts.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a sequence of well-orchestrated steps that construct the imidazole ring and install the ketone functionality simultaneously. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the base, likely generating a trifluoroacetamidine intermediate in situ. This species then undergoes isomerization, setting the stage for the palladium cycle. The palladium catalyst, activated by triphenylphosphine, coordinates with the alkyne moiety of the propargylamine derivative, leading to palladation and the formation of an alkenyl palladium intermediate. Subsequent isomerization converts this into a more stable alkyl palladium species, which is primed for the crucial carbonylation step.
Under the influence of carbon monoxide generated in situ from the formic acid and acetic anhydride mixture, the alkyl palladium intermediate undergoes insertion to form an acyl palladium complex. This is followed by the oxidative addition of the diaryl iodonium salt, generating a high-valent tetravalent palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst. This intricate mechanism ensures high regioselectivity and minimizes side reactions, which is critical for maintaining a clean impurity profile. For quality control teams, understanding this mechanism highlights the importance of precise stoichiometry, particularly the ratio of formic acid to acetic anhydride, to ensure a steady supply of the carbonyl source without overwhelming the system with acidic byproducts.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol is designed to be user-friendly, utilizing standard Schlenk techniques or sealed vessels to maintain an inert atmosphere if necessary, although the robustness of the catalyst system often tolerates minor variations. The key to success lies in the proper activation of the palladium catalyst and the controlled release of carbon monoxide. Operators should ensure that the mixture of palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid is thoroughly homogenized before the addition of the organic substrates. Detailed standardized synthetic steps for this process are provided in the guide below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodide in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted imidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented methodology offers transformative benefits that extend far beyond the laboratory bench. The reliance on commodity chemicals such as propargylamine and simple aromatic amines (precursors to the imidoyl chlorides) ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or regulated reagents. This stability is paramount for commercial scale-up of complex pharmaceutical intermediates, where continuity of supply is often the difference between meeting market demand and facing costly delays. The simplicity of the workup procedure, involving basic filtration and standard column chromatography, further streamlines the downstream processing, reducing the burden on purification teams and accelerating the time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and hazardous trifluoromethylating agents with cost-effective alternatives like trifluoroethylimidoyl chloride. By eliminating the need for cryogenic conditions or high-pressure reactors, the method significantly lowers capital expenditure (CAPEX) and operational expenditure (OPEX). The use of a simple palladium chloride catalyst system, which is cheaper than many specialized palladium complexes, coupled with the ability to recycle solvents like THF, contributes to substantial cost savings. Additionally, the high reaction efficiency and yields reported (often exceeding 90% for optimized substrates) minimize waste generation and maximize the throughput of the manufacturing facility, directly improving the bottom line.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply chain reliability by reducing the sensitivity to minor fluctuations in temperature or reagent quality. Since the starting materials are widely available from multiple global suppliers, the risk of single-source dependency is minimized. This diversification of the supply base is a critical strategic advantage for procurement managers tasked with securing long-term contracts for API production. Furthermore, the mild reaction conditions reduce the likelihood of equipment failure or safety incidents, ensuring uninterrupted production schedules and consistent delivery timelines to downstream customers.
- Scalability and Environmental Compliance: Scalability is inherently built into this process, as evidenced by its successful demonstration at the gram level with clear pathways to kilogram and tonne scales. The absence of toxic gases like carbon monoxide (which is generated in situ and consumed immediately) and the use of relatively benign solvents align with green chemistry principles. This facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing process. For companies striving to meet stringent ESG (Environmental, Social, and Governance) goals, this method offers a sustainable route to high-value intermediates, reducing the burden on waste treatment facilities and lowering the overall E-factor of the synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when integrating this method into their workflows. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this palladium-catalyzed method over traditional trifluoromethylation techniques?
A: Unlike traditional methods that often rely on hazardous or expensive reagents like trifluorodiazoethane, this patented approach utilizes cheap and readily available starting materials such as trifluoroethylimidoyl chloride and propargylamine. Furthermore, the reaction proceeds under exceptionally mild conditions (30°C) with high efficiency and excellent substrate compatibility, significantly reducing operational risks and costs.
Q: What is the substrate scope for the aryl groups in this synthesis?
A: The method demonstrates broad substrate tolerance, accommodating various substituted aryl groups for both the R and Ar positions. Substituents such as methyl, tert-butyl, chlorine, bromine, trifluoromethyl, nitro, and methoxy groups are well-tolerated at ortho, meta, or para positions, allowing for the diverse design of imidazole derivatives tailored for specific biological activities.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is scalable to the gram level and provides a strong possibility for industrial large-scale production. The use of inexpensive catalysts like palladium chloride, combined with simple post-processing steps like filtration and column chromatography, makes it highly viable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced synthetic methodologies play in accelerating drug development and optimizing production costs. Our team of expert chemists has extensively evaluated the potential of the palladium-catalyzed route described in CN111423381B and is fully equipped to translate this academic innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are designed to handle sensitive organometallic reactions with the utmost precision, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee the highest quality standards for every batch.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of specific 2-trifluoromethyl imidazole derivatives or comprehensive process development services, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this innovative method can enhance your supply chain and reduce your overall manufacturing costs.
