Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Applications
Advanced Molybdenum-Copper Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Applications
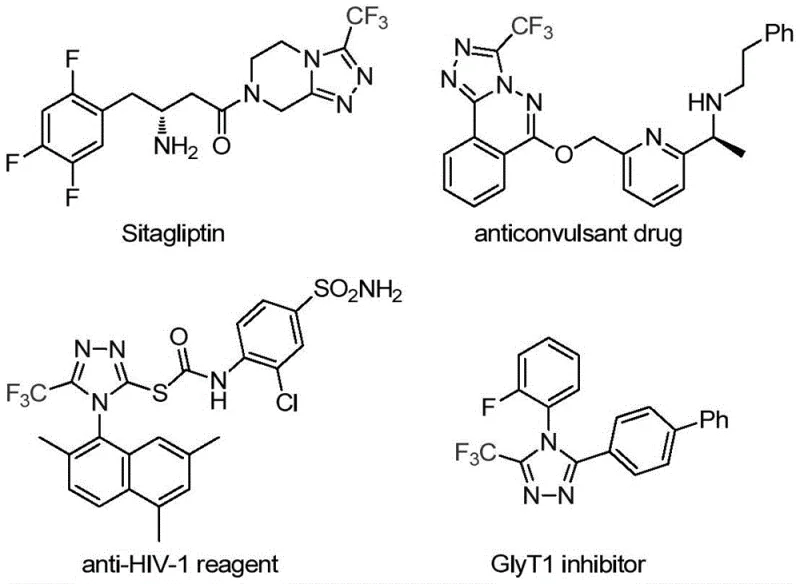
The rapid evolution of medicinal chemistry has placed a premium on heterocyclic scaffolds that offer enhanced metabolic stability and bioavailability, with the 1,2,4-triazole ring standing out as a cornerstone structure in modern drug design. As detailed in the groundbreaking patent CN113307778A, a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds has been developed, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a sophisticated molybdenum-copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride and functionalized isonitriles under remarkably mild conditions. The significance of this advancement cannot be overstated, as trifluoromethyl-substituted triazoles are pivotal motifs found in blockbuster medications such as Sitagliptin and various anticonvulsant agents, where the introduction of the trifluoromethyl group drastically improves lipophilicity and electronegativity profiles. For R&D directors and procurement strategists alike, this patent represents a paradigm shift towards more efficient, scalable, and economically viable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazole frameworks has been plagued by significant synthetic challenges that hinder large-scale production and increase overall manufacturing costs. Traditional literature methods often rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently require harsh reaction conditions, expensive reagents, and generate substantial hazardous waste. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose serious safety risks due to the explosive nature of diazo compounds, making them unsuitable for kilogram-scale operations in standard GMP facilities. These legacy methods also suffer from limited substrate scope and poor atom economy, often necessitating complex protection-deprotection sequences that elongate the synthetic timeline and reduce the overall yield of the desired active pharmaceutical ingredient intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology disclosed in CN113307778A introduces a streamlined, one-pot cycloaddition strategy that utilizes readily available trifluoroethylimidoyl chloride and functionalized isonitriles as starting materials. This innovative route employs a dual-catalyst system comprising molybdenum hexacarbonyl and cuprous acetate, which synergistically activate the reactants to form the five-membered triazole ring with high efficiency and selectivity. The reaction proceeds smoothly in common organic solvents like THF at moderate temperatures ranging from 70°C to 90°C, eliminating the need for cryogenic conditions or high-pressure equipment that typically drive up capital expenditure. By avoiding unstable diazo intermediates and utilizing stable, commercially accessible precursors, this novel approach not only enhances operational safety but also significantly simplifies the downstream purification process, thereby offering a robust solution for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Mo-Cu Co-Catalyzed Cycloaddition
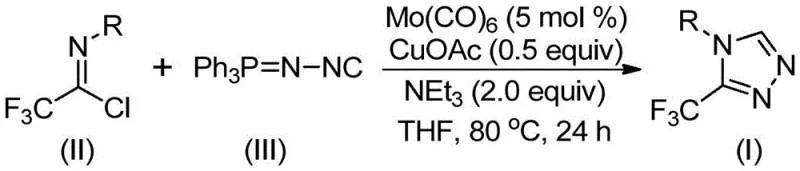
The mechanistic elegance of this transformation lies in the cooperative interaction between the molybdenum and copper species, which orchestrates the assembly of the triazole ring through a carefully balanced sequence of coordination and bond-forming events. Initially, the molybdenum hexacarbonyl acts as a potent metal activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that increases the nucleophilicity of the carbon center. Concurrently, the cuprous acetate promotes a [3+2] cycloaddition reaction between this activated isonitrile species and the trifluoroethylimidoyl chloride, facilitating the formation of a key five-membered ring intermediate. This intermediate subsequently undergoes a dehydration or elimination step, potentially assisted by trace water or the basic environment provided by triethylamine, to expel triphenylphosphine oxide and aromatize the system into the final 3-trifluoromethyl-substituted 1,2,4-triazole product. Understanding this catalytic cycle is crucial for process chemists aiming to optimize reaction parameters, as the precise stoichiometry of the catalysts—typically 5 mol% for Mo(CO)6 and 0.5 equivalents for CuOAc—is critical for maximizing turnover frequency while minimizing metal contamination in the final API.
From an impurity control perspective, the mildness of this catalytic system plays a pivotal role in ensuring a clean reaction profile, which is a primary concern for regulatory compliance in drug substance manufacturing. Unlike harsh acidic or basic hydrolysis methods that can lead to the degradation of sensitive functional groups or the formation of polymeric by-products, this Mo-Cu co-catalyzed pathway operates under neutral to slightly basic conditions that preserve the integrity of diverse substituents on the aromatic rings. The use of molecular sieves in the reaction mixture further aids in scavenging moisture that could otherwise hydrolyze the sensitive imidoyl chloride starting material, thereby suppressing the formation of trifluoroacetamide side products. This high level of chemoselectivity allows for the successful incorporation of electron-withdrawing groups like nitro and halogens, as well as electron-donating groups like methoxy and alkyl chains, without significant loss of yield, providing R&D teams with a versatile platform for generating diverse libraries of triazole analogs for structure-activity relationship studies.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis protocol is designed to be straightforward and adaptable to various scales of production, ranging from milligram-level discovery chemistry to multi-kilogram pilot plant campaigns. The procedure begins with the precise weighing of molybdenum hexacarbonyl, cuprous acetate, and triethylamine, which are combined with the substrate trifluoroethylimidoyl chloride and the functionalized isonitrile in a dry Schlenk tube or reactor charged with tetrahydrofuran (THF) as the solvent. The reaction mixture is then heated to a controlled temperature of 80°C and stirred for approximately 24 hours, a duration that has been empirically determined to balance complete conversion with minimal thermal degradation of the product. Following the reaction period, the workup involves a simple filtration step to remove insoluble metal salts and molecular sieves, after which the filtrate is concentrated and subjected to standard silica gel column chromatography to afford the pure triazole derivative. For a comprehensive, step-by-step breakdown of the standardized operating procedures and safety precautions, please refer to the detailed guide below.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the high-purity 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling strategic advantages that directly impact the bottom line and operational resilience of the manufacturing organization. The primary driver of value creation is the substantial cost reduction in manufacturing achieved through the utilization of inexpensive, commodity-grade catalysts and reagents that are abundantly available in the global chemical market. By replacing exotic or hazardous reagents with stable alternatives like cuprous acetate and triethylamine, the process eliminates the need for specialized storage infrastructure and expensive hazard mitigation protocols, thereby lowering the total cost of ownership for the production facility. Furthermore, the high reaction efficiency and broad substrate tolerance mean that a single production line can be utilized to manufacture a wide array of different triazole derivatives simply by swapping the starting amine or isonitrile, maximizing asset utilization and reducing the need for dedicated campaign lines for each specific intermediate.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of earth-abundant transition metals rather than precious metals like palladium or rhodium, which are subject to volatile market pricing and supply constraints. The elimination of expensive ligands and the ability to run the reaction at relatively low catalyst loadings significantly decrease the raw material cost per kilogram of the final product. Additionally, the simplified workup procedure, which avoids complex aqueous extractions or distillation steps, reduces solvent consumption and energy usage, contributing to a leaner and more sustainable manufacturing footprint that aligns with modern green chemistry initiatives.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on starting materials that are either commercially off-the-shelf or easily synthesized from bulk commodities in just one or two steps. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or out-of-specification results that often plague more sensitive synthetic methodologies. This reliability allows supply chain planners to forecast demand with greater accuracy and maintain lower safety stock levels, freeing up working capital and reducing the risk of inventory obsolescence in a fast-moving pharmaceutical landscape.
- Scalability and Environmental Compliance: The scalability of this method from gram to kilogram scales has been explicitly demonstrated, proving its readiness for commercial scale-up of complex pharmaceutical intermediates without the need for extensive re-engineering of the process parameters. The use of THF, a solvent with well-established recovery and recycling protocols, coupled with the generation of minimal heavy metal waste due to the low catalyst loading, simplifies environmental compliance and wastewater treatment requirements. This ease of scale-up ensures that the transition from clinical trial material to commercial launch supply can be executed rapidly, reducing lead time for high-purity intermediates and accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled a set of frequently asked questions based on the specific technical details and experimental data provided in the patent documentation. These inquiries address common concerns regarding catalyst optimization, substrate compatibility, and purification strategies, offering clear insights into the practical application of this Mo-Cu co-catalyzed system. By understanding the nuances of reaction stoichiometry and post-processing requirements, teams can better assess the resource allocation needed for successful implementation and anticipate potential challenges during the technology transfer phase.
Q: What are the optimal reaction conditions for the Mo-Cu co-catalyzed triazole synthesis?
A: According to patent CN113307778A, the optimal conditions involve using THF as the solvent at temperatures between 70°C and 90°C for 18 to 30 hours, utilizing a molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile of approximately 1:1.5.
Q: Does this method support a wide range of substrate functional groups?
A: Yes, the method demonstrates excellent tolerance for various substituents on the aryl ring, including methyl, methoxy, fluoro, chloro, and nitro groups, allowing for the synthesis of diverse derivatives suitable for drug discovery.
Q: How does the purification process impact the final yield and purity?
A: The post-treatment involves simple filtration followed by silica gel mixing and column chromatography, which effectively removes metal residues and by-products, ensuring high-purity intermediates required for pharmaceutical applications without complex extraction protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics, and we are uniquely positioned to support your projects with our advanced synthetic capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early stages of drug discovery or preparing for global market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole we deliver meets the highest standards of quality and consistency required by regulatory authorities worldwide.
We invite you to engage with our technical procurement team to discuss how this innovative Mo-Cu co-catalyzed synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this superior manufacturing route. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a dialogue regarding route feasibility assessments for your proprietary molecules, ensuring a seamless and efficient supply chain partnership.
