Revolutionizing Triazole Synthesis: Scalable Iodine-Catalyzed Process for High-Purity Pharmaceutical Intermediates
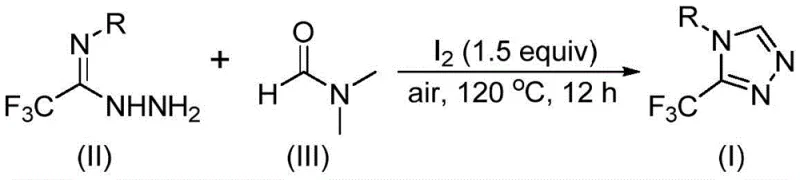
The patent CN114920707B introduces a groundbreaking synthetic methodology for 3-trifluoromethyl-substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical manufacturing. This innovative process leverages dimethylformamide (DMF) as both solvent and carbon source under molecular iodine catalysis, operating effectively in ambient air without requiring specialized anhydrous or anaerobic conditions that typically complicate traditional triazole syntheses. The methodology demonstrates exceptional practicality through its use of commercially available starting materials and straightforward reaction parameters (120°C for 12 hours), addressing critical pain points in the production of fluorinated heterocyclic intermediates essential for modern drug development. By eliminating the need for expensive transition metal catalysts and complex purification protocols, this approach offers substantial operational advantages while maintaining high functional group tolerance across diverse aryl substrates. The patent's emphasis on DMF's dual role as solvent and reactant establishes a new paradigm for sustainable heterocycle synthesis that directly supports the growing demand for trifluoromethylated building blocks in the pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted 1,2,4-triazoles typically require stringent anhydrous and anaerobic conditions that significantly increase operational complexity and cost while limiting scalability in commercial manufacturing environments. These methods often depend on expensive transition metal catalysts such as palladium or copper complexes that necessitate additional purification steps to remove trace metal contaminants, creating substantial quality control challenges for pharmaceutical applications where stringent purity specifications are mandatory. The narrow substrate scope of conventional approaches restricts structural diversity, making it difficult to access the broad range of functionalized triazole derivatives required for modern drug discovery programs targeting complex biological pathways. Furthermore, the multi-step sequences commonly employed generate significant waste streams that conflict with increasingly stringent environmental regulations governing pharmaceutical manufacturing processes. These combined limitations create substantial barriers to efficient production of critical intermediates like GlyT1 inhibitors and factor IXa inhibitors that rely on trifluoromethylated triazole scaffolds.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-catalyzed tandem cyclization that operates effectively under ambient air conditions using DMF as both solvent and carbon source, eliminating the need for specialized equipment or hazardous reagents. This single-step process demonstrates remarkable functional group tolerance across diverse aryl substitutions including halogen, alkyl, alkoxy, and trifluoromethyl groups, enabling the synthesis of complex pharmaceutical intermediates such as Sitagliptin precursors and anticonvulsant drug building blocks with unprecedented flexibility. The reaction's operational simplicity—requiring only standard glassware and common laboratory equipment—dramatically reduces capital investment while enhancing process robustness for commercial scale-up from laboratory to industrial production volumes. By leveraging DMF's dual functionality as both reaction medium and carbon donor, the process avoids expensive metal catalysts entirely, thereby eliminating costly metal removal steps and associated quality control challenges inherent in traditional approaches. This innovative strategy represents a paradigm shift toward more sustainable and economically viable production of fluorinated heterocyclic compounds essential for next-generation pharmaceutical development.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism involves two distinct pathways where DMF's methyl and formyl groups serve as carbon sources under iodine catalysis. When the formyl group participates, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate, followed by intramolecular cyclization with elimination of dimethylamine to yield the triazole product. Alternatively, when the N-methyl group acts as carbon source, DMF first reacts with iodine to form an amine salt that eliminates hydrogen iodide before nucleophilic addition with the hydrazide component. This generates an azadiene intermediate that undergoes intramolecular nucleophilic addition followed by oxidative aromatization to produce the final triazole structure. Both pathways operate efficiently under identical reaction conditions (I₂ at 1.5 equivalents, 120°C for 12 hours in air), demonstrating the remarkable versatility of this catalytic system. The molecular iodine catalyst facilitates multiple bond-forming events through sequential oxidation steps without requiring additional oxidants or co-catalysts.
Impurity control is achieved through the reaction's inherent selectivity and mild conditions that minimize side reactions commonly observed in traditional triazole syntheses. The absence of transition metals eliminates potential metal contamination pathways while the air-stable reaction environment prevents oxidation byproducts that often complicate anaerobic processes. The well-defined reaction pathway ensures consistent product formation across diverse substrates as demonstrated by the patent's comprehensive examples covering electron-donating and electron-withdrawing substituents on the aryl ring. Post-reaction purification via standard column chromatography effectively removes minor impurities without requiring specialized techniques, ensuring stringent purity specifications are met for pharmaceutical applications. This robust impurity profile directly supports regulatory compliance requirements for API intermediates while maintaining high process efficiency across multiple production scales.
How to Synthesize 3-Trifluoromethyl-1,2,4-triazole Efficiently
This innovative iodine-catalyzed process represents a significant advancement in the synthesis of fluorinated triazole intermediates critical for pharmaceutical development. The methodology leverages readily available starting materials and standard laboratory equipment while operating under ambient atmospheric conditions, making it exceptionally suitable for both research-scale synthesis and commercial manufacturing environments. By utilizing DMF as both solvent and carbon source, the process eliminates multiple synthetic steps required by conventional approaches while maintaining excellent functional group tolerance across diverse aryl substrates. The following standardized procedure details the precise implementation of this patented technology for consistent production of high-purity triazole compounds essential for pharmaceutical applications.
- Combine molecular iodine (1.5 equivalents) with trifluoroethyliminohydrazide in DMF solvent under ambient air conditions.
- Heat the reaction mixture at 120°C for 12 hours in a standard Schlenk tube without requiring anhydrous or anaerobic environments.
- Purify the crude product through silica gel column chromatography after standard filtration and washing procedures.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial commercial advantages by addressing critical pain points in pharmaceutical intermediate procurement and supply chain management. The elimination of specialized equipment requirements and transition metal catalysts creates immediate cost savings while enhancing supply chain resilience through simplified logistics and reduced dependency on scarce specialty chemicals. The process's compatibility with standard manufacturing infrastructure enables seamless integration into existing production facilities without requiring capital-intensive modifications or specialized operator training. These operational efficiencies translate directly into improved supply chain reliability and reduced time-to-market for critical pharmaceutical intermediates while maintaining the stringent quality standards required by global regulatory authorities.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated purification steps significantly reduces raw material costs while avoiding complex waste treatment procedures required for metal-contaminated streams. The use of commercially available DMF as dual solvent/reactant streamlines material sourcing and inventory management without requiring specialized handling protocols or safety measures associated with hazardous reagents commonly used in traditional syntheses.
- Enhanced Supply Chain Reliability: The process operates effectively with standard laboratory equipment under ambient air conditions using readily available starting materials that can be sourced from multiple global suppliers. This flexibility eliminates single-point failure risks associated with specialized reagents or equipment while enabling rapid scale-up from laboratory to commercial production volumes without revalidation requirements.
- Scalability and Environmental Compliance: The straightforward reaction protocol demonstrates excellent scalability from milligram to multi-kilogram quantities while generating minimal waste streams compared to conventional multi-step syntheses. The absence of heavy metals and hazardous byproducts simplifies environmental compliance procedures and reduces disposal costs while supporting corporate sustainability initiatives through inherently greener chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics. These insights derive directly from the patent's experimental data and mechanistic studies, providing evidence-based responses to common inquiries from procurement specialists and technical decision-makers evaluating this synthetic approach for pharmaceutical intermediate production.
Q: Why is this method superior to conventional triazole synthesis?
A: This process eliminates the need for stringent anhydrous and anaerobic conditions required by traditional methods, significantly reducing operational complexity while maintaining high substrate versatility across diverse aryl substitutions.
Q: How does DMF dual functionality enhance commercial viability?
A: DMF serves as both reaction solvent and carbon source, eliminating expensive metal catalysts and specialized equipment while enabling direct scalability from laboratory to industrial production volumes.
Q: What substrate scope supports diverse pharmaceutical applications?
A: The methodology accommodates broad functional group tolerance including halogen, alkyl, alkoxy, and trifluoromethyl substitutions, enabling customized synthesis of API intermediates like GlyT1 inhibitors and anticonvulsant drug precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-triazole Supplier
Our patented iodine-catalyzed synthesis represents a transformative approach to producing critical fluorinated heterocyclic intermediates with exceptional efficiency and scalability. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities. Our dedicated technical teams specialize in optimizing complex synthetic routes like this DMF-based triazole methodology to ensure consistent quality and reliable supply for global pharmaceutical partners requiring high-purity intermediates meeting exacting regulatory standards.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments demonstrating how this innovative methodology can enhance your supply chain resilience while delivering substantial operational efficiencies.
