Advanced Electrochemical Trifluoromethylation for High-Purity Pharmaceutical Intermediates
Introduction to Green Electrochemical Trifluoromethylation
The introduction of trifluoromethyl groups into organic molecules is a critical transformation in modern medicinal chemistry, significantly enhancing the lipophilicity, metabolic stability, and membrane permeability of drug candidates. As highlighted in patent CN111690947B, filed in October 2021, there is a pressing industrial need for efficient methods to access these motifs without relying on hazardous reagents. Traditional approaches often suffer from the use of toxic oxidants and expensive transition metals, creating substantial environmental and purification burdens for manufacturers. This patent discloses a novel electrochemical synthesis method that utilizes electricity as a clean reagent to drive the direct C-H trifluoromethylation of arylamide derivatives. By operating under mild conditions ranging from 25-75°C and employing a constant current of 5-20mA, this technology offers a sustainable pathway for producing high-value pharmaceutical intermediates. The method is particularly notable for its atom economy and the elimination of external chemical oxidants, aligning perfectly with the principles of green chemistry that are increasingly demanded by global regulatory bodies.
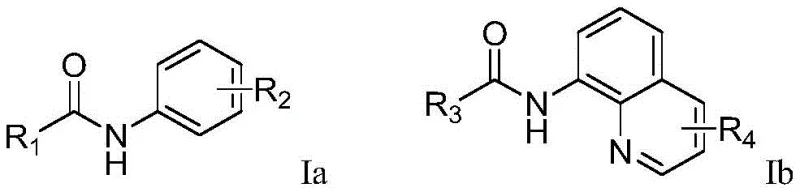
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct trifluoromethylation of aromatic systems has relied heavily on transition metal catalysis, utilizing metals such as copper, nickel, or iron in conjunction with strong chemical oxidants like potassium persulfate or ammonium persulfate. While effective in laboratory settings, these conventional methods present significant challenges for commercial scale-up. The use of stoichiometric amounts of heavy metal catalysts necessitates rigorous and costly downstream purification steps to ensure that residual metal levels meet the stringent limits imposed by pharmacopeial standards for active pharmaceutical ingredients. Furthermore, the reliance on strong chemical oxidants introduces safety hazards related to thermal runaway and the generation of large volumes of sulfate-containing waste streams. These factors collectively increase the cost of goods sold and complicate the environmental compliance profile of the manufacturing process, making them less attractive for long-term supply chain sustainability.
The Novel Approach
In stark contrast, the electrochemical methodology presented in the patent data offers a paradigm shift by replacing chemical oxidants with electrons. The process involves the anodic oxidation of sodium trifluoromethanesulfonate to generate trifluoromethyl radicals in situ, which then react with the arylamide substrate directed by a pyridine or quinoline moiety. This approach completely obviates the need for transition metal catalysts, thereby simplifying the reaction mixture and drastically reducing the complexity of the workup procedure. The reaction conditions are remarkably mild, typically proceeding at 50°C with a constant current of 15mA, which minimizes energy consumption and thermal stress on sensitive functional groups. By utilizing simple electrode materials such as a carbon anode and a platinum cathode, the method ensures that the process is not only chemically efficient but also economically viable for large-scale operations, providing a reliable route for the synthesis of complex fluorinated building blocks.
Mechanistic Insights into Electrochemical C-H Functionalization
The core mechanism of this transformation relies on the unique ability of the amide directing group to coordinate with the substrate and facilitate site-selective C-H activation at the ortho-position. Under the applied electrical potential, the trifluoromethanesulfonate anion undergoes single-electron oxidation at the anode surface to release sulfur dioxide and generate a highly reactive trifluoromethyl radical. This radical species subsequently attacks the electron-rich aromatic ring of the arylamide, which has been activated by the coordination of the nitrogen atom in the pyridine or quinoline ring. The resulting cyclohexadienyl radical intermediate is then oxidized, either at the anode or via a mediated process, to restore aromaticity and yield the final trifluoromethylated product. This electrochemical cycle ensures that the redox equivalents are supplied continuously and precisely by the power source, avoiding the accumulation of reactive intermediates that could lead to side reactions or decomposition.
From an impurity control perspective, the absence of metal catalysts significantly reduces the risk of metal-induced side reactions such as homocoupling or over-oxidation, which are common pitfalls in traditional cross-coupling chemistries. The selectivity of the reaction is further enhanced by the specific interaction between the directing group and the electrode interface, ensuring that trifluoromethylation occurs predominantly at the desired ortho-position relative to the amide nitrogen. This high regioselectivity is crucial for pharmaceutical applications where isomeric impurities can be difficult to separate and may possess different toxicological profiles. The use of tetrabutylammonium salts as supporting electrolytes further stabilizes the reaction environment, facilitating ion transport without participating in unwanted side reactions, thus ensuring a clean reaction profile that simplifies the isolation of the target compound.
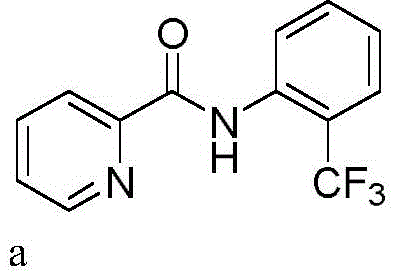
How to Synthesize Trifluoromethylated Arylamides Efficiently
The synthesis protocol outlined in the patent provides a robust framework for executing this transformation with high reproducibility. The procedure begins with the assembly of an undivided electrochemical cell equipped with a carbon rod anode and a platinum plate cathode, which are readily available and cost-effective materials. The reaction mixture is prepared by dissolving the arylamide substrate and sodium trifluoromethanesulfonate in acetonitrile, along with a quaternary ammonium electrolyte such as tetrabutylammonium tetrafluoroborate to ensure sufficient conductivity. Once the components are mixed, a constant current is applied while maintaining the temperature between 25°C and 75°C, with 50°C being the optimal setpoint for balancing reaction rate and selectivity. Detailed standardized synthetic steps for specific derivatives are provided in the technical guide below to assist process chemists in replicating these results.
- Prepare the electrochemical cell by installing a carbon anode and a platinum cathode in a reaction vessel equipped with stirring capabilities.
- Charge the vessel with the arylamide substrate, sodium trifluoromethanesulfonate as the CF3 source, a quaternary ammonium electrolyte, and acetonitrile solvent.
- Apply a constant current of 5-20mA at a temperature between 25-75°C for 30-180 minutes, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits regarding cost structure and supply reliability. The most significant advantage lies in the elimination of expensive transition metal catalysts and the associated ligands, which often constitute a major portion of the raw material costs in fine chemical synthesis. By removing these components from the bill of materials, manufacturers can achieve substantial cost savings while simultaneously reducing their exposure to the price volatility of precious metals like palladium or iridium. Furthermore, the simplified purification process means that fewer processing units and less solvent are required for workup, leading to a reduction in overall manufacturing cycle time and utility consumption. This efficiency gain allows for faster throughput and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts eliminates the need for specialized scavenging resins or complex extraction protocols designed to lower metal residues to ppm levels. This simplification of the downstream processing directly lowers the operational expenditure per kilogram of product. Additionally, the use of electricity as the primary oxidant is inherently cheaper and more controllable than purchasing and handling bulk quantities of chemical oxidants, which often require special storage and handling procedures due to their hazardous nature. The overall process mass intensity is improved, meaning less waste is generated per unit of product, which further reduces waste disposal costs and environmental levies.
- Enhanced Supply Chain Reliability: The reagents required for this electrochemical process, such as sodium trifluoromethanesulfonate and common quaternary ammonium salts, are commodity chemicals with stable and robust global supply chains. Unlike specialized catalysts that may have single-source suppliers or long lead times, these materials are readily available from multiple vendors, mitigating the risk of supply disruptions. The mild reaction conditions also reduce the wear and tear on reactor equipment, extending the lifespan of capital assets and minimizing unplanned maintenance downtime. This reliability ensures consistent delivery schedules for customers who depend on a steady flow of high-quality intermediates for their own drug development pipelines.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, moving from laboratory benchtop cells to industrial flow reactors with relative ease. The continuous nature of electrochemical processes allows for better heat and mass transfer control compared to large batch reactors, reducing the risk of hot spots and improving safety profiles. From an environmental standpoint, the process generates minimal hazardous waste, as the primary byproduct is sulfur dioxide which can be easily scrubbed, and there are no heavy metal sludge disposal issues. This aligns with increasingly strict environmental regulations in major manufacturing hubs, ensuring long-term operational continuity without the threat of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical trifluoromethylation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific projects. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed trifluoromethylation?
A: The electrochemical method described in CN111690947B eliminates the need for toxic transition metal catalysts such as copper or nickel, and avoids the use of stoichiometric chemical oxidants like persulfates. This results in a greener process with simplified downstream purification, as there are no heavy metal residues to remove from the final pharmaceutical intermediate.
Q: What types of substrates are compatible with this trifluoromethylation protocol?
A: The protocol demonstrates broad substrate scope, successfully functionalizing N-phenylpicolinamides and N-(quinolin-8-yl)benzamides. It tolerates various substituents including halogens, alkyl groups, cyano, and nitro groups on the aromatic rings, making it versatile for synthesizing diverse drug-like scaffolds.
Q: Is this electrochemical process suitable for large-scale commercial production?
A: Yes, the reaction operates under mild conditions (25-75°C) and uses simple electrode materials like carbon and platinum. The absence of hazardous oxidants and the use of electricity as a traceless reagent make the process inherently safer and more scalable for industrial manufacturing compared to batch reactions requiring dangerous chemical oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Arylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of advanced pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111690947B can be successfully translated into robust industrial processes. We are committed to delivering high-purity API intermediates that meet stringent purity specifications, utilizing our state-of-the-art rigorous QC labs to verify every batch. Our infrastructure is designed to handle complex fluorination chemistries safely and efficiently, providing our partners with a secure and compliant source for their critical building blocks.
We invite you to collaborate with us to leverage this green chemistry advantage for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our electrochemical capabilities can optimize your supply chain and reduce your overall manufacturing costs. Let us be your partner in navigating the complexities of modern fluorine chemistry.
