Advanced Electrochemical Synthesis of Trifluoromethylated Arylamides for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to introduce trifluoromethyl groups into molecular scaffolds, given their profound impact on lipophilicity and metabolic stability. Patent CN111690947B discloses a groundbreaking electrochemical synthesis method for trifluoromethylated arylamide derivatives that addresses critical limitations in current manufacturing technologies. This innovation utilizes a constant current electrolysis approach to achieve direct C-H trifluoromethylation, bypassing the need for stoichiometric chemical oxidants and transition metal catalysts. By leveraging electricity as a clean reagent, this process aligns perfectly with the principles of green chemistry while maintaining high atom economy. For R&D directors and procurement managers, this technology represents a significant opportunity to streamline the supply chain for complex heterocyclic intermediates used in drugs like Efavirenz and Celebrex analogues.
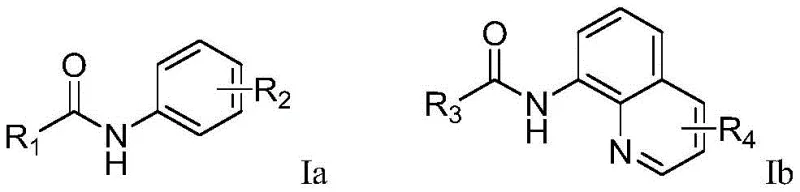
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for introducing trifluoromethyl groups often rely heavily on transition metal catalysis involving nickel, copper, or iron complexes paired with strong chemical oxidants such as potassium persulfate. These conventional routes present substantial challenges for large-scale manufacturing, primarily due to the difficulty in removing trace metal residues to meet stringent pharmaceutical purity standards. Furthermore, the use of stoichiometric oxidants generates significant amounts of inorganic waste, complicating wastewater treatment and increasing the environmental footprint of the synthesis. The requirement for pre-functionalized substrates or harsh reaction conditions also limits the scope of compatible functional groups, often leading to lower overall yields and higher production costs for high-purity pharmaceutical intermediates.
The Novel Approach
The electrochemical method described in the patent offers a transformative alternative by replacing chemical oxidants with electrons generated at the anode surface. This approach operates under mild conditions, typically between 25°C and 75°C, using simple electrolytes like tetrabutylammonium tetrafluoroborate in acetonitrile. The absence of external metal catalysts means the final product is free from heavy metal contamination, drastically simplifying the purification workflow. As illustrated in the reaction scope, this method accommodates a wide variety of substrates including pyridyl amides and quinolinyl amides with diverse substituents. This versatility allows for the rapid synthesis of library compounds for drug discovery while providing a robust pathway for commercial manufacturing of key intermediates.
Mechanistic Insights into Electrochemical C-H Activation
The core mechanism involves the anodic oxidation of the trifluoromethylation reagent, likely sodium trifluoromethanesulfonate, to generate reactive trifluoromethyl radical species. These radicals are then intercepted by the arylamide substrate, which is activated through coordination with the electrode surface or via a mediated electron transfer process. The directing group effect of the amide moiety ensures high regioselectivity, favoring ortho-trifluoromethylation on the aromatic ring. This precise control over reactivity minimizes the formation of regioisomers, which are often difficult to separate and can compromise the quality of the final active pharmaceutical ingredient. The cathodic reaction simultaneously balances the charge, typically involving proton reduction, ensuring the overall electrochemical cell remains stable throughout the 30 to 180-minute reaction window.
Impurity control in this electrochemical system is inherently superior because the reaction does not produce metal salts or reduced oxidant byproducts that typically plague thermal radical reactions. The primary side reactions are limited to over-oxidation of the substrate or solvent decomposition, both of which can be mitigated by optimizing the current density and electrode material. The patent specifies the use of carbon anodes and platinum cathodes, a combination that provides a stable potential window for generating the desired radicals without degrading the sensitive amide backbone. This mechanistic clarity gives process chemists confidence in scaling the reaction, as the parameters are easily monitored and controlled via potentiostatic or galvanostatic regulation.
How to Synthesize Trifluoromethylated Arylamides Efficiently
To implement this synthesis effectively, one must carefully select the electrode materials and electrolyte concentration to ensure consistent current flow and radical generation. The patent outlines a straightforward procedure where the substrate, reagent, and electrolyte are dissolved in acetonitrile and subjected to constant current electrolysis. Detailed operational parameters such as temperature control and reaction time are critical to maximizing yield while minimizing energy consumption. The standardized synthetic steps below provide a reliable framework for reproducing the high-quality results reported in the patent examples, ensuring that the process is robust enough for technology transfer.
- Prepare the reaction vessel with a carbon anode and platinum cathode, adding substrate arylamide, sodium trifluoromethanesulfonate, and electrolyte.
- Add acetonitrile solvent and stir the mixture at 25-75°C while applying a constant current of 5-20mA for 30-180 minutes.
- Concentrate the reaction mixture under reduced pressure and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to electrochemical synthesis offers tangible benefits in terms of cost structure and logistical simplicity. By eliminating the need for expensive and often volatile transition metal catalysts, the raw material costs are significantly reduced, and the supply chain becomes less vulnerable to fluctuations in the price of precious metals. Additionally, the removal of metal catalysts obviates the need for specialized scavenging resins or complex extraction protocols, leading to faster batch turnover times and reduced solvent usage. This streamlined workflow translates directly into lower operating expenses and a more sustainable manufacturing profile that aligns with modern corporate environmental goals.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and stoichiometric chemical oxidants removes two major cost drivers from the bill of materials. Without the need for expensive metal scavengers or extensive washing steps to meet residual metal specifications, the downstream processing costs are drastically simplified. This efficiency gain allows for a more competitive pricing structure for the final trifluoromethylated intermediates, making them attractive for high-volume generic drug production.
- Enhanced Supply Chain Reliability: The reagents required for this electrochemical process, such as sodium trifluoromethanesulfonate and tetrabutylammonium salts, are commercially available and stable, reducing the risk of supply disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, the inputs for this method are commodity chemicals. This reliability ensures consistent production schedules and reduces the inventory burden on manufacturing sites, facilitating a just-in-time supply model for critical API intermediates.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, and the mild reaction conditions reduce the energy load associated with heating and cooling large vessels. The absence of hazardous oxidants like persulfates minimizes safety risks during storage and handling, lowering insurance and compliance costs. Furthermore, the reduced waste stream simplifies effluent treatment, helping facilities meet increasingly strict environmental regulations without capital-intensive upgrades to wastewater infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical trifluoromethylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is essential for evaluating the feasibility of adopting this method for your specific product pipeline.
Q: What are the advantages of this electrochemical method over traditional metal-catalyzed trifluoromethylation?
A: This method eliminates the need for toxic transition metal catalysts like Nickel or Copper and avoids hazardous chemical oxidants, resulting in a cleaner impurity profile and reduced downstream purification costs.
Q: What is the typical yield and purity achievable with this process?
A: According to patent data, yields range from 30% to 80% depending on the substrate, with high purity achievable through standard silica gel chromatography due to the absence of metal residues.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the mild reaction conditions (25-75°C) and use of stable electrolytes make it highly suitable for commercial scale-up, avoiding the safety risks associated with strong chemical oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Arylamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting innovative synthetic technologies to deliver high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods like this electrochemical process are successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoromethylated arylamide meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this green synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can optimize your supply chain for these critical fluorinated building blocks.
