Scalable Electrochemical Synthesis of Trifluoromethylated Arylamides for Commercial API Production
Scalable Electrochemical Synthesis of Trifluoromethylated Arylamides for Commercial API Production
The introduction of trifluoromethyl groups into organic molecules is a critical transformation in modern medicinal chemistry, profoundly influencing the lipophilicity, metabolic stability, and membrane permeability of drug candidates. As highlighted in the groundbreaking patent CN111690947B, published on October 15, 2021, a novel electrochemical synthesis method has been developed to produce trifluoromethylated arylamide derivatives with exceptional efficiency and environmental compatibility. This technology represents a paradigm shift from traditional stoichiometric oxidant-dependent methods to a sustainable, electricity-driven process. By leveraging the power of anodic oxidation, this method facilitates direct C-H functionalization under mild conditions, avoiding the use of hazardous reagents often associated with conventional trifluoromethylation protocols. For pharmaceutical manufacturers seeking reliable pharmaceutical intermediate suppliers, this electrochemical route offers a robust pathway to access high-value scaffolds found in blockbuster drugs such as Efavirenz and Celecoxib.
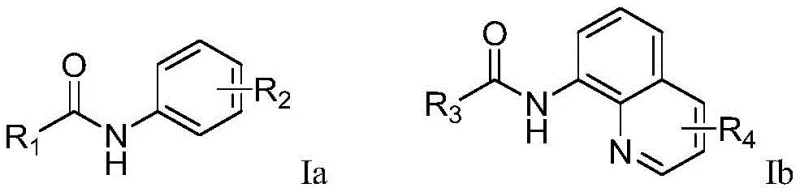
The core innovation lies in the utilization of simple arylamide substrates, specifically those containing picolinamide or 8-aminoquinoline directing groups, which undergo selective ortho-trifluoromethylation. The reaction system employs sodium trifluoromethanesulfonate as the trifluoromethyl source and operates under constant current electrolysis. This approach not only streamlines the synthetic workflow but also aligns perfectly with the principles of green chemistry by minimizing waste generation. The versatility of the method is demonstrated across a broad range of substrates, accommodating various electronic and steric environments, which is essential for the diverse needs of API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the installation of trifluoromethyl groups onto aromatic rings has relied heavily on transition metal catalysis, often involving nickel, copper, or iron complexes coupled with strong chemical oxidants such as persulfates. While effective in some contexts, these traditional methods suffer from significant drawbacks that hinder their application in large-scale commercial production. The primary concern is the presence of toxic heavy metal residues in the final product, which necessitates rigorous and costly purification steps to meet stringent pharmaceutical regulatory standards. Furthermore, the use of stoichiometric amounts of strong oxidants generates substantial quantities of inorganic salt waste, creating environmental disposal challenges and increasing the overall carbon footprint of the synthesis. Additionally, many of these reactions require harsh conditions or pre-functionalized substrates, limiting their atom economy and increasing raw material costs.
The Novel Approach
In stark contrast, the electrochemical method disclosed in patent CN111690947B circumvents these issues by using electrons as the traceless oxidant. This metal-free protocol operates at mild temperatures ranging from 25°C to 75°C and utilizes a constant current of 5-20mA to drive the reaction. The absence of external chemical oxidants and transition metal catalysts fundamentally simplifies the reaction mixture, leading to cleaner crude profiles and easier isolation of the target compounds. The method demonstrates excellent functional group tolerance, successfully transforming substrates with halogens, cyano groups, and alkyl substituents without compromising yield or selectivity. This technological advancement provides a compelling solution for cost reduction in pharmaceutical intermediate manufacturing by eliminating expensive catalysts and reducing waste treatment overheads.
Mechanistic Insights into Electrochemical C-H Functionalization
The mechanistic pathway of this electrochemical transformation involves the anodic oxidation of the trifluoromethanesulfonate anion to generate a trifluoromethyl radical species. In the presence of the arylamide substrate, the picolinamide or quinoline amide moiety acts as a bidentate directing group, coordinating to the reactive intermediate or facilitating electron transfer at the electrode surface. This coordination creates a transient cyclic structure that lowers the activation energy for C-H bond cleavage at the ortho-position. The resulting aryl radical then couples with the trifluoromethyl radical to form the new C-C bond, followed by rearomatization to yield the final trifluoromethylated product. This radical-polar crossover mechanism ensures high regioselectivity, exclusively targeting the position adjacent to the amide nitrogen.
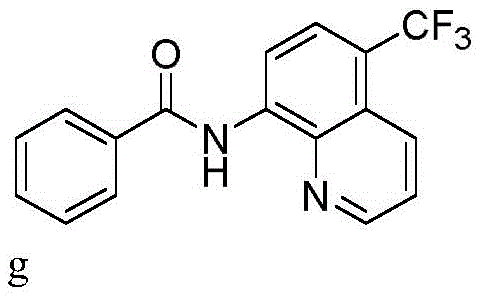
Impurity control is inherently superior in this electrochemical system due to the precise control over the oxidation potential via current regulation. Unlike chemical oxidants which may over-oxidize sensitive functional groups, the constant current mode allows for a steady generation of reactive species, minimizing side reactions such as polymerization or non-selective ring oxidation. The use of tetrabutylammonium salts as electrolytes further stabilizes the reaction medium and enhances conductivity without introducing nucleophilic impurities. For R&D teams focused on high-purity pharmaceutical intermediates, this level of control translates to simplified crystallization processes and higher overall purity specifications, reducing the burden on analytical quality control laboratories during scale-up campaigns.
How to Synthesize Trifluoromethylated Arylamides Efficiently
The practical implementation of this synthesis is straightforward and adaptable to standard laboratory equipment, making it highly accessible for process development teams. The procedure involves charging an undivided cell with a carbon anode and a platinum cathode, followed by the addition of the substrate, electrolyte, and solvent. The reaction proceeds under galvanostatic conditions, requiring minimal supervision once the parameters are set. Detailed standardized synthetic steps for specific derivatives are outlined in the technical guide below, providing a clear roadmap for replication and optimization.
- Prepare an electrochemical cell equipped with a carbon anode and a platinum cathode, ensuring the system is ready for constant current operation.
- Charge the reactor with the arylamide substrate, sodium trifluoromethanesulfonate as the CF3 source, a quaternary ammonium electrolyte, and acetonitrile solvent.
- Apply a constant current of 5-20mA at 25-75°C for 30-180 minutes, followed by concentration and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical technology offers transformative benefits for procurement and supply chain management within the fine chemical sector. The elimination of precious metal catalysts removes a major variable cost driver and mitigates supply risks associated with fluctuating metal prices. Furthermore, the simplified workup procedure reduces solvent consumption and processing time, directly contributing to operational efficiency. For supply chain heads, the robustness of the method ensures consistent quality and reliable delivery schedules, which are critical for maintaining continuous API production lines.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the complete removal of transition metal catalysts and stoichiometric oxidants. In traditional processes, the cost of noble metals and the subsequent scavenging resins required to remove metal traces can account for a substantial portion of the COGS. By replacing these with electricity and inexpensive electrolytes, the process achieves drastic cost savings. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The reagents utilized in this electrochemical method, such as sodium trifluoromethanesulfonate and tetrabutylammonium salts, are commodity chemicals with stable global supply chains. Unlike specialized ligands or air-sensitive catalysts that may face availability bottlenecks, these materials are readily sourced from multiple vendors. This diversification of the supply base reduces the risk of production delays and ensures that commercial scale-up of complex pharmaceutical intermediates can proceed without interruption, safeguarding the continuity of downstream drug manufacturing.
- Scalability and Environmental Compliance: Electrochemical synthesis is uniquely suited for scale-up through the use of flow reactors, which offer superior mass transfer and heat dissipation compared to batch processes. This scalability allows for the seamless transition from gram-scale discovery to multi-ton commercial production. Moreover, the green nature of the process, characterized by the absence of heavy metal waste and reduced solvent usage, simplifies regulatory compliance and environmental permitting. This aligns with the increasing industry demand for sustainable manufacturing practices and helps companies meet their corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical trifluoromethylation process. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed trifluoromethylation?
A: The electrochemical approach eliminates the need for toxic transition metal catalysts (such as Nickel, Copper, or Iron) and stoichiometric chemical oxidants. This significantly simplifies downstream processing by removing the requirement for expensive heavy metal scavenging steps, thereby reducing production costs and environmental impact.
Q: How does the reaction achieve regioselectivity for the ortho-position?
A: The reaction utilizes a directing group strategy where the picolinamide or 8-aminoquinoline amide moiety coordinates with the reactive species generated at the anode. This coordination directs the trifluoromethyl radical specifically to the ortho-C-H bond, ensuring high regioselectivity without the need for pre-functionalized substrates.
Q: Is this electrochemical process suitable for large-scale commercial manufacturing?
A: Yes, electrochemical synthesis is inherently scalable. By utilizing flow electrochemical reactors or larger batch cells with optimized electrode surface areas, the process can be transitioned from laboratory scale to multi-ton production while maintaining the green chemistry benefits of using electrons as the primary reagent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Arylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies in securing a competitive edge in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the electrochemical trifluoromethylation route described in CN111690947B and confirmed its potential for delivering high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the absence of metal impurities and confirming structural integrity through advanced spectroscopic analysis.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge technology for your next-generation drug candidates. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our electrochemical capabilities can optimize your supply chain and accelerate your time to market.
