Revolutionizing Antiviral Drug Intermediate Production: A Cost-Effective Cobalt-Catalyzed Green Synthesis Approach for Commercial Manufacturing
The Chinese patent CN114671798B presents a significant advancement in the manufacturing of critical antiviral drug intermediates through an innovative cobalt-catalyzed green synthesis methodology. This patented process specifically addresses the production of (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate derivatives or salts, which serve as essential building blocks for life-saving antiviral medications including nirmatrelvir (Paxlovid), boceprevir, and narlaprevir. The technology represents a substantial improvement over conventional synthetic routes by eliminating hazardous reagents, reducing processing time from days to hours, and achieving consistently high yields above ninety percent without requiring column chromatography purification steps. This breakthrough directly responds to industry demands for more sustainable pharmaceutical manufacturing processes while maintaining stringent quality requirements essential for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for these critical antiviral intermediates have been plagued by multiple significant limitations that hinder both economic viability and manufacturing scalability. The WO2021250648A1 method requires five days of room temperature stirring followed by multiple purification steps including filtration through celite and extensive extraction procedures, resulting in only seventy-five percent yield. Similarly, CN114057627A employs hazardous reagents such as n-butyllithium which presents serious safety concerns during manufacturing operations while requiring column chromatography purification that substantially increases production costs and processing time. These conventional methods also incorporate expensive transition metal catalysts that necessitate additional removal steps to meet pharmaceutical purity standards, creating significant bottlenecks in commercial scale-up efforts. The lengthy processing times and complex purification requirements inherent in these older methodologies create substantial barriers to reliable supply chain management for pharmaceutical manufacturers facing urgent demand for antiviral therapeutics.
The Novel Approach
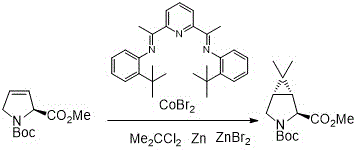
The patented methodology described in CN114671798B overcomes these critical limitations through an elegant one-step cyclization process that operates efficiently at room temperature without requiring hazardous reagents or complex purification procedures. By utilizing zinc powder and dihaloalkane in combination with an innovative cobalt-based catalyst system, this approach achieves high yields exceeding ninety percent within one to three hours of reaction time while eliminating the need for column chromatography. The catalyst system employs readily available ligands derived from simple condensation reactions between substituted anilines and diacetylpyridine compounds, creating an economically viable solution that maintains exceptional selectivity and efficiency. Crucially, this method avoids using zinc halide additives that were previously required in similar transformations, further simplifying both the reaction process and subsequent workup procedures while maintaining excellent stereochemical control essential for pharmaceutical applications.
Mechanistic Insights into Cobalt-Catalyzed Cyclization
The catalytic cycle begins with the formation of an active cobalt complex through reduction by zinc powder, generating a low-valent cobalt species that facilitates oxidative addition into the dihaloalkane substrate. This key step creates an organocobalt intermediate that subsequently coordinates with the pyrrolidine starting material through nitrogen coordination, positioning it for intramolecular cyclization. The cobalt catalyst enables selective C-C bond formation through a well-defined oxidative addition/reductive elimination pathway that maintains stereochemical integrity throughout the transformation process. The use of specifically designed tridentate ligands featuring pyridine-based frameworks with aryl substituents creates an optimal steric environment that promotes high regioselectivity while preventing undesired side reactions that would compromise product purity. This mechanistic understanding explains why alternative ligand systems containing bulky tert-butyl groups demonstrate significantly reduced catalytic efficiency compared to methyl-substituted variants.
The impurity profile generated by this methodology demonstrates exceptional control over byproduct formation due to the precise steric environment created by the catalyst system. The absence of zinc halide additives eliminates potential sources of halogen-containing impurities that would require additional purification steps in conventional approaches. The one-step nature of this transformation prevents accumulation of intermediate impurities that commonly occur in multi-step syntheses, resulting in consistently high purity levels exceeding ninety-nine percent without requiring chromatographic purification. The carefully optimized reaction conditions maintain excellent control over stereochemistry throughout the cyclization process, preserving critical chiral centers essential for subsequent conversion to active pharmaceutical ingredients. This level of impurity control directly addresses regulatory requirements while significantly reducing quality control testing burdens during commercial manufacturing operations.
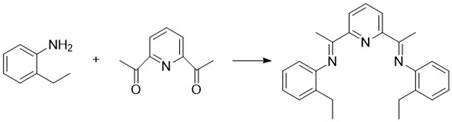
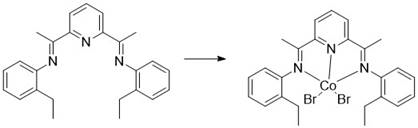
How to Synthesize Nirmatrelvir Intermediate Efficiently
This innovative cobalt-catalyzed methodology represents a significant advancement in intermediate synthesis for antiviral pharmaceuticals, offering substantial improvements over conventional approaches through its streamlined process design and exceptional efficiency metrics. The following standardized procedure details the precise implementation of this patented technology for reliable commercial-scale manufacturing operations.
- Prepare catalyst by combining methyl-substituted pyridine-based ligand with cobalt halide under nitrogen atmosphere in anhydrous THF
- Add zinc powder to the catalyst solution at room temperature and stir for approximately fifteen minutes until deep purple color develops
- Introduce dihaloalkane reagent and maintain room temperature stirring for one to three hours before workup
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial commercial benefits that directly address critical pain points experienced by procurement and supply chain management teams within pharmaceutical manufacturing organizations. By eliminating hazardous reagents and complex purification requirements while maintaining exceptional yield and purity metrics, this approach creates significant value across multiple dimensions of pharmaceutical production operations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous reagents such as n-butyllithium significantly reduces raw material costs while avoiding specialized handling requirements that increase operational expenses. The one-step nature of this transformation eliminates multiple intermediate processing steps required in conventional approaches, substantially reducing overall manufacturing costs through simplified process flow and reduced equipment utilization requirements.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials and eliminating specialized equipment requirements, this methodology provides greater flexibility in sourcing raw materials while maintaining consistent quality output. The simplified process design reduces vulnerability to supply chain disruptions that commonly affect multi-step syntheses requiring specialized reagents or equipment, creating more resilient manufacturing operations capable of meeting fluctuating demand patterns for critical antiviral therapeutics.
- Scalability and Environmental Compliance: The room temperature operation without specialized equipment requirements enables straightforward scale-up from laboratory to commercial production volumes while maintaining consistent quality metrics. The elimination of hazardous reagents and reduction in solvent usage through simplified purification procedures significantly improves environmental sustainability metrics while reducing waste treatment costs associated with traditional manufacturing approaches.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this innovative synthesis methodology based on extensive analysis of patent data and industry requirements.
Q: How does this cobalt-catalyzed method improve upon previous synthesis approaches for nirmatrelvir intermediates?
A: This method eliminates hazardous reagents like n-butyllithium used in prior art, reduces reaction time from days to hours, achieves higher yields (90%+), and avoids column chromatography purification steps that increased production costs.
Q: What specific cost advantages does this green synthesis method provide for pharmaceutical manufacturers?
A: The process eliminates expensive metal catalysts and hazardous reagents while significantly reducing processing time and eliminating column chromatography requirements, resulting in substantial cost savings throughout manufacturing.
Q: How does this synthesis method address supply chain reliability concerns for antiviral drug production?
A: By using readily available starting materials and eliminating specialized equipment requirements, this method provides greater supply chain flexibility while maintaining high purity standards essential for pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nirmatrelvir Intermediate Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through our state-of-the-art QC labs. As your strategic partner in antiviral drug development, we combine deep technical expertise in organometallic catalysis with comprehensive manufacturing capabilities to deliver consistent quality intermediates that meet exacting pharmaceutical standards while optimizing cost structures through innovative process design.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments through our Customized Cost-Saving Analysis service, which will demonstrate how our patented technology can enhance your supply chain resilience while delivering significant economic benefits.
