Advanced Manufacturing of 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane for Antiviral Drug Production
Advanced Manufacturing of 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane for Antiviral Drug Production
The global pharmaceutical landscape has been profoundly reshaped by the urgent demand for effective antiviral therapeutics, particularly those targeting SARS-CoV-2. At the heart of this medical breakthrough lies Nirmatrelvir, the active protease inhibitor component of Paxlovid, which relies heavily on the availability of high-quality bicyclic amine intermediates. Patent CN114605311B introduces a transformative preparation method for 6,6-dimethyl-3-azabicyclo[3.1.0]hexane, a critical scaffold not only for Nirmatrelvir but also for Hepatitis C treatments like Boceprevir. This technical disclosure represents a significant leap forward in process chemistry, shifting away from scarce and expensive precursors toward a streamlined, cost-efficient synthesis starting from 3,3-dimethyl-4-pentenoic acid. By optimizing the construction of the strained cyclopropane ring and the piperidine nitrogen heterocycle, this innovation addresses key bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
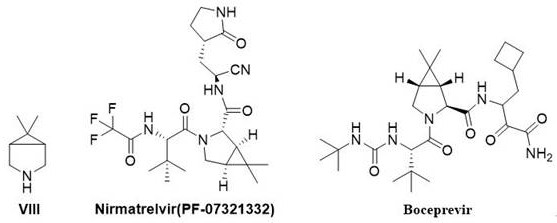
The structural complexity of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane presents unique challenges in organic synthesis, primarily due to the strain energy of the fused three-membered ring and the stereochemical requirements for biological activity. As illustrated in the structural overview, this bicyclic core serves as the rigid backbone that positions the warhead groups in Nirmatrelvir for optimal binding to the viral 3CL protease. The ability to manufacture this scaffold with high purity and consistent stereochemistry is paramount for regulatory compliance and drug efficacy. The patented methodology described herein offers a robust solution that aligns with the rigorous quality standards expected by R&D directors and procurement teams in the top-tier pharmaceutical industry, ensuring a reliable supply of this essential building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital bicyclic amine has relied heavily on routes originating from caronic anhydride, a pathway that suffers from significant economic and operational drawbacks. As depicted in the conventional synthesis diagram, the reliance on caronic anhydride introduces a major supply chain vulnerability due to the limited number of manufacturers capable of producing this specialized starting material at scale, leading to inflated costs and potential shortages. Furthermore, the chemical transformation required to convert the anhydride into the target amine involves a demanding carbonyl reduction step that consumes large quantities of potent reducing agents, thereby increasing both the raw material costs and the safety hazards associated with handling reactive hydrides on a large scale.
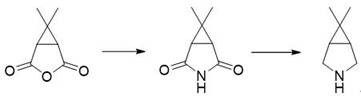
Beyond the economic inefficiencies, the traditional caronic anhydride route poses severe purification challenges that hinder industrial scalability. The final product obtained through these legacy methods exhibits high mutual solubility with water, making the isolation of the free amine exceptionally difficult and often requiring energy-intensive distillation or complex chromatographic techniques that result in substantial yield losses. Additionally, alternative historical approaches, such as those utilizing dichlorochrysanthemic acid, involve excessively long synthetic sequences with harsh reaction conditions that are environmentally burdensome and difficult to control in a multi-ton reactor setting. These cumulative factors render conventional methods suboptimal for meeting the surging global demand for antiviral medications.
The Novel Approach
In stark contrast to these legacy processes, the novel approach detailed in patent CN114605311B leverages 3,3-dimethyl-4-pentenoic acid, a commodity chemical that is inexpensive, widely available, and structurally poised for efficient functionalization. This innovative route fundamentally reimagines the construction of the bicyclic system by employing a strategic halolactonization followed by amination and intramolecular cyclization, effectively bypassing the need for the problematic caronic anhydride precursor. The process is characterized by its operational simplicity, utilizing standard organic transformations that are well-understood and easily controlled in a manufacturing environment, thus significantly lowering the barrier to entry for large-scale production.
A critical advantage of this new methodology lies in its streamlined reduction strategy, which targets a lactam intermediate rather than a ketone or anhydride. This subtle yet powerful modification drastically reduces the stoichiometric requirement for reducing agents, translating directly into substantial cost savings and a reduced environmental footprint. Moreover, the incorporation of a benzyl protecting group throughout the synthesis facilitates the management of polarity and solubility, allowing for the final target product to be isolated via simple filtration and concentration after deprotection. This elegant solution to the purification bottleneck ensures high recovery rates and exceptional purity, making it ideally suited for the rigorous demands of commercial API manufacturing.
Mechanistic Insights into Halolactonization and Cyclization Cascade
The core of this synthetic innovation is a sophisticated cascade of reactions that efficiently builds molecular complexity from simple linear precursors. The process initiates with a halolactonization reaction, where 3,3-dimethyl-4-pentenoic acid reacts with a halogenating agent in the presence of an oxidant to form a halogenated lactone. This step is crucial as it sets the stereochemistry and installs the necessary leaving group for subsequent nucleophilic attacks. Following this, an amination reaction introduces the nitrogen atom, which then participates in an intramolecular aminolysis to close the six-membered lactam ring. This sequence effectively constructs the piperidinone core with high regioselectivity, minimizing the formation of unwanted isomeric byproducts that could complicate downstream purification.
The formation of the strained cyclopropane ring, a defining feature of the target molecule, is achieved through a precise intramolecular nucleophilic substitution. After converting the hydroxyl group of the lactam intermediate into a superior leaving group (such as a mesylate or halide), treatment with a strong base induces an intramolecular SN2 reaction that closes the three-membered ring. This step requires careful control of reaction conditions to prevent elimination side reactions, but the patented protocol optimizes the base and solvent system to favor cyclization. The final stages involve the reduction of the lactam carbonyl to a methylene group using a borohydride-Lewis acid system, followed by catalytic hydrogenolysis to remove the benzyl protecting group. This mechanistic pathway ensures that the final amine is produced with minimal impurities, addressing the critical quality attributes required for pharmaceutical intermediates.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
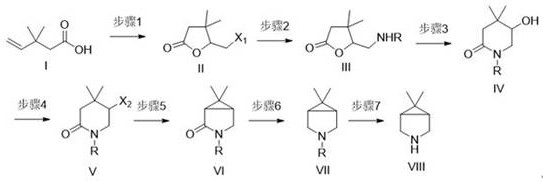
The execution of this synthesis requires a disciplined approach to reaction parameters to maximize yield and safety. The process begins with the activation of the alkene moiety in the starting acid, followed by the sequential introduction of nitrogen and the closure of the rings. Each step has been optimized to use common solvents like acetonitrile, THF, and dichloromethane, ensuring compatibility with standard stainless steel reactors. The detailed standardized synthesis steps below outline the specific reagents and conditions necessary to replicate this high-efficiency route in a pilot or production plant setting.
- Perform halolactonization on 3,3-dimethyl-4-pentenoic acid to form a halogenated lactone intermediate.
- Conduct amination followed by intramolecular transesterification to generate the piperidinone core.
- Execute halogenation or sulfonylation, followed by base-mediated cyclization to close the cyclopropane ring, and finally reduce and deprotect.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers compelling strategic benefits that extend beyond mere technical feasibility. The shift from specialty starting materials to commodity chemicals fundamentally alters the cost structure of the intermediate, insulating the supply chain from the volatility associated with niche reagent markets. By eliminating the dependency on caronic anhydride, manufacturers can secure a more stable and predictable supply of raw materials, which is critical for maintaining continuous production schedules in the face of fluctuating global demand for antiviral therapeutics.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic reduction in reagent consumption and the simplification of unit operations. By replacing the expensive carbonyl reduction of the traditional route with a more efficient lactam reduction, the process significantly lowers the usage of costly reducing agents. Furthermore, the ability to isolate the final product through simple filtration and concentration eliminates the need for expensive and time-consuming chromatographic purification steps, resulting in substantial overall cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: The reliance on 3,3-dimethyl-4-pentenoic acid as the starting material ensures a robust supply chain foundation, as this chemical is produced by multiple vendors globally. This diversification of supply sources mitigates the risk of single-source bottlenecks that have plagued previous manufacturing campaigns. Additionally, the operational simplicity of the reaction steps reduces the likelihood of batch failures due to complex process upsets, thereby enhancing the overall reliability and consistency of the manufacturing output.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is markedly superior to existing alternatives. The reduction in hazardous waste generation, particularly from excess reducing agents and complex solvent systems used in purification, aligns with modern green chemistry principles. The process is designed to be scalable from kilogram to metric-ton quantities without requiring specialized equipment, facilitating rapid technology transfer and commercial scale-up of complex pharmaceutical intermediates while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and advantageous effects reported in the patent documentation, providing clarity on the practical aspects of adopting this technology for industrial applications.
Q: What is the primary advantage of this new synthesis route over the traditional caronic anhydride method?
A: The new route utilizes 3,3-dimethyl-4-pentenoic acid, which is significantly cheaper and more readily available than caronic anhydride. Furthermore, it simplifies the reduction step by targeting a lactam rather than a carbonyl group, drastically reducing the consumption of expensive reducing agents and simplifying purification.
Q: How does this process address purification challenges associated with the final amine product?
A: Traditional methods often struggle with the water solubility and purification of the final bicyclic amine. This patented method employs a benzyl protecting group strategy that allows for robust intermediate purification. The final deprotection step yields the target product through simple filtration and concentration, avoiding complex chromatographic separations.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It relies on commodity chemicals, avoids harsh conditions found in alternative routes (such as those using dichlorochrysanthemic acid), and features straightforward work-up procedures like filtration and extraction, making it highly viable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
As the pharmaceutical industry continues to evolve, the need for agile and technically proficient manufacturing partners has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in process development to bring complex synthetic routes like the one described in CN114605311B to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global health initiatives. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for API synthesis.
We invite potential partners to engage with our technical procurement team to explore how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and security in your antiviral drug manufacturing programs.
