Advanced Synthesis Of 6 6-Dimethyl-3-Azabicyclo Hexane For Commercial Scale-Up
The global pharmaceutical landscape has witnessed an unprecedented surge in demand for high-purity antiviral intermediates, particularly those serving as the structural backbone for next-generation protease inhibitors. Patent CN114751853A introduces a transformative methodology for preparing (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane compounds, which are critical building blocks for drugs like Nirmatrelvir. This specific chemical scaffold is not merely a commodity but a strategic asset in the fight against viral pathogens, necessitating a supply chain that prioritizes both safety and efficiency. The structural complexity of the target molecule, as seen in the final drug assembly, requires precise stereochemical control to ensure biological efficacy. 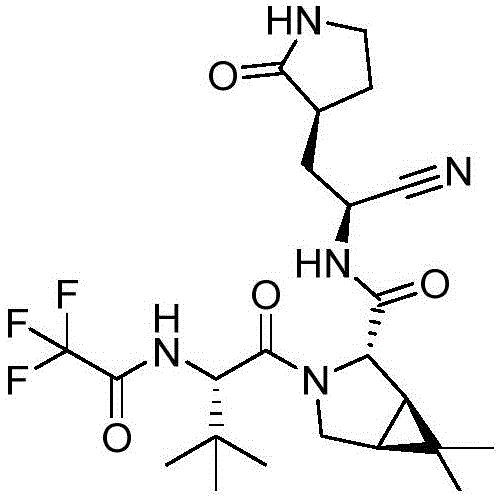 . Traditional manufacturing approaches have often struggled with the inherent risks associated with cyanation steps, creating bottlenecks for reliable pharmaceutical intermediates supplier networks. This new patent addresses these challenges by re-engineering the cyanation source, moving away from hazardous solids to a more manageable liquid reagent system. For R&D Directors and Procurement Managers, this shift represents a significant opportunity to optimize cost reduction in pharmaceutical intermediates manufacturing while adhering to stricter environmental and safety regulations. The ability to produce high-purity OLED material or pharmaceutical grades relies on such foundational process improvements, ensuring that the final API meets the rigorous standards required by regulatory bodies worldwide.
. Traditional manufacturing approaches have often struggled with the inherent risks associated with cyanation steps, creating bottlenecks for reliable pharmaceutical intermediates supplier networks. This new patent addresses these challenges by re-engineering the cyanation source, moving away from hazardous solids to a more manageable liquid reagent system. For R&D Directors and Procurement Managers, this shift represents a significant opportunity to optimize cost reduction in pharmaceutical intermediates manufacturing while adhering to stricter environmental and safety regulations. The ability to produce high-purity OLED material or pharmaceutical grades relies on such foundational process improvements, ensuring that the final API meets the rigorous standards required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane derivatives has been plagued by inefficient multi-step routes that rely heavily on dangerous reagents. Prior art, such as the methods disclosed in WO2004113295, utilizes solid potassium cyanide or sodium cyanide, which pose severe occupational health hazards and require extensive safety infrastructure. 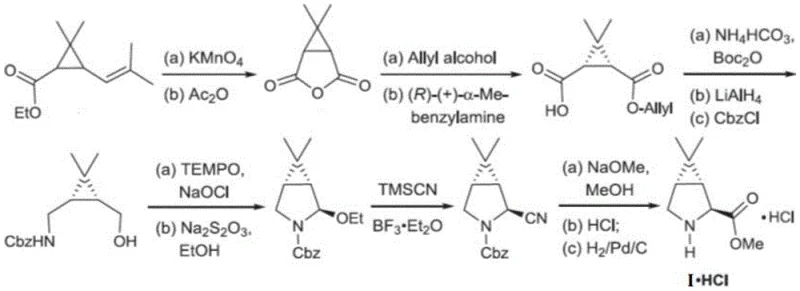 . These conventional pathways are not only lengthy, involving numerous protection and deprotection steps, but they also suffer from suboptimal enantiomeric excess (ee) in the final product. The handling of solid cyanide salts necessitates specialized containment facilities, rigorous personal protective equipment protocols, and complex waste treatment procedures, all of which drive up operational expenditures. Furthermore, the variability in reaction conditions often leads to inconsistent batch quality, complicating the commercial scale-up of complex polymer additives or pharmaceutical intermediates. The economic burden of managing toxic waste streams and the potential for production shutdowns due to safety incidents make these legacy routes unsustainable for modern high-volume manufacturing. Consequently, supply chain heads face significant challenges in reducing lead time for high-purity pharmaceutical intermediates when relying on these outdated technologies.
. These conventional pathways are not only lengthy, involving numerous protection and deprotection steps, but they also suffer from suboptimal enantiomeric excess (ee) in the final product. The handling of solid cyanide salts necessitates specialized containment facilities, rigorous personal protective equipment protocols, and complex waste treatment procedures, all of which drive up operational expenditures. Furthermore, the variability in reaction conditions often leads to inconsistent batch quality, complicating the commercial scale-up of complex polymer additives or pharmaceutical intermediates. The economic burden of managing toxic waste streams and the potential for production shutdowns due to safety incidents make these legacy routes unsustainable for modern high-volume manufacturing. Consequently, supply chain heads face significant challenges in reducing lead time for high-purity pharmaceutical intermediates when relying on these outdated technologies.
The Novel Approach
In stark contrast, the novel approach detailed in CN114751853A leverages acetone cyanohydrin as a liquid cyanating agent, fundamentally altering the safety and efficiency profile of the synthesis. This liquid reagent can be transported via sealed pipelines and metered automatically, eliminating the need for manual weighing and direct human contact with toxic substances. The new route significantly shortens the reaction sequence, bypassing the need for intermediate sulfonate formation required in older enzymatic or chemical methods. By streamlining the process, manufacturers can achieve a total yield of over 80%, a substantial improvement over the fragmented yields of previous generations. This method also demonstrates superior stereoselectivity, ensuring that the (1R,2S,5S) configuration is maintained with high fidelity throughout the transformation. The use of common alcohol solvents like methanol and isopropanol further simplifies the downstream processing, allowing for easier solvent recovery and recycling. For a reliable agrochemical intermediate supplier or pharma partner, this translates to a more robust and predictable production schedule. The elimination of solid hazardous waste not only lowers disposal costs but also aligns with green chemistry principles, enhancing the overall sustainability of the manufacturing operation.
Mechanistic Insights into Liquid Phase Cyanation and Cyclization
The core of this technological breakthrough lies in the nucleophilic addition of the cyanide equivalent to the activated double bond of the azabicyclo precursor. In the first step, the compound of formula III reacts with acetone cyanohydrin under mild alkaline catalysis, typically using sodium carbonate or potassium carbonate in an aqueous alcohol mixture. 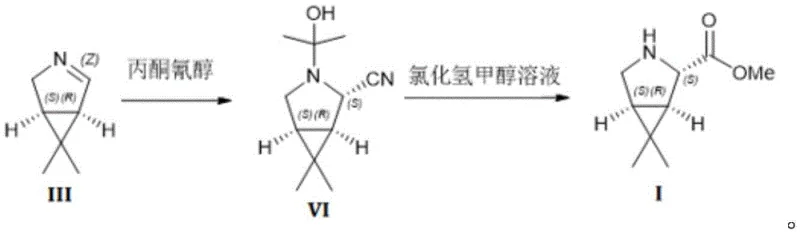 . The mechanism involves the in situ generation of cyanide ions from the liquid cyanohydrin, which then attack the electrophilic center with high regioselectivity. This liquid-phase reaction environment ensures homogeneous mixing and precise temperature control, typically maintained between 20 to 40°C, which is crucial for preventing side reactions and decomposition. The resulting intermediate, compound VI, is formed with high purity, setting the stage for the subsequent alcoholysis step. The choice of base and solvent ratio is critical; a molar ratio of base to substrate between 1:0.1 to 0.3 optimizes the reaction kinetics without promoting excessive hydrolysis. This precise control over reaction parameters is what allows for the consistent production of high-purity pharmaceutical intermediates, meeting the exacting standards of R&D Directors who prioritize impurity profile management. The mechanistic elegance of this route lies in its simplicity, avoiding the complex catalytic cycles or expensive chiral auxiliaries often required in asymmetric synthesis.
. The mechanism involves the in situ generation of cyanide ions from the liquid cyanohydrin, which then attack the electrophilic center with high regioselectivity. This liquid-phase reaction environment ensures homogeneous mixing and precise temperature control, typically maintained between 20 to 40°C, which is crucial for preventing side reactions and decomposition. The resulting intermediate, compound VI, is formed with high purity, setting the stage for the subsequent alcoholysis step. The choice of base and solvent ratio is critical; a molar ratio of base to substrate between 1:0.1 to 0.3 optimizes the reaction kinetics without promoting excessive hydrolysis. This precise control over reaction parameters is what allows for the consistent production of high-purity pharmaceutical intermediates, meeting the exacting standards of R&D Directors who prioritize impurity profile management. The mechanistic elegance of this route lies in its simplicity, avoiding the complex catalytic cycles or expensive chiral auxiliaries often required in asymmetric synthesis.
Following the cyanation, the process moves to a sophisticated alcoholysis and hydrolysis sequence that converts the nitrile group into the desired methyl ester. The compound of formula VI is treated with a methanolic hydrogen chloride solution, which facilitates the formation of an imido ester intermediate. This intermediate is then hydrolyzed upon contact with water, yielding the free amine ester which is subsequently captured as the hydrochloride salt. The pH adjustment to greater than 8.5 during the workup is a critical control point, ensuring that the amine is fully liberated into the organic phase for extraction while leaving acidic impurities behind. The use of ether solvents like methyl tert-butyl ether or isopropyl ether for extraction provides excellent phase separation and product recovery. Finally, crystallization from an alcohol-ether mixture yields the hydrochloride salt with a chemical purity exceeding 99.7% and no detectable enantiomers. This level of purity is essential for the commercial scale-up of complex pharmaceutical additives, where even trace impurities can affect the stability and efficacy of the final drug product. The robustness of this mechanism ensures that the process is scalable from kilogram to multi-ton production without loss of quality.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
To implement this synthesis effectively, operators must adhere to strict parameter controls regarding temperature, molar ratios, and solvent quality. The process begins with the preparation of the reaction mixture, ensuring that the acetone cyanohydrin is added dropwise to maintain thermal stability. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety across different production scales. . Adhering to these protocols minimizes the risk of exothermic runaways and ensures consistent product quality. The subsequent crystallization step requires careful control of the cooling rate and anti-solvent addition to maximize crystal purity and filterability. By following this optimized workflow, manufacturers can achieve the high yields and safety standards necessary for modern pharmaceutical supply chains.
- React compound of formula III with acetone cyanohydrin in an alcohol solvent under alkali catalysis to obtain compound of formula VI.
- Perform alcoholysis of compound VI with methanolic hydrogen chloride, followed by hydrolysis and pH adjustment to greater than 8.5.
- Crystallize the resulting compound I using hydrogen chloride in an alcohol solvent with an ether anti-solvent to obtain the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
The transition to this novel synthesis route offers profound commercial benefits that extend beyond mere technical feasibility, directly impacting the bottom line and supply chain resilience. For procurement managers, the elimination of solid cyanide salts removes a significant regulatory and logistical burden, simplifying the sourcing of raw materials and reducing the cost of compliance. Cost Reduction in Manufacturing: The streamlined two-step process significantly reduces the consumption of solvents and reagents compared to multi-step legacy routes. By avoiding expensive chiral resolution steps and hazardous waste disposal fees associated with solid cyanides, the overall cost of goods sold is drastically lowered. The higher yield directly translates to more product per batch, optimizing asset utilization and reducing the cost per kilogram of the active intermediate. This efficiency allows for more competitive pricing strategies without compromising on quality margins. Enhanced Supply Chain Reliability: The use of liquid reagents that can be pipeline-transferred ensures a continuous and stable supply of key inputs, reducing the risk of production stoppages due to material handling issues. The simplified process flow shortens the manufacturing cycle time, enabling faster response to market demand fluctuations and reducing lead time for high-purity pharmaceutical intermediates. Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, generating less hazardous waste and utilizing common, recoverable solvents. This aligns with global sustainability goals and reduces the environmental footprint of the manufacturing site, ensuring long-term operational viability and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and process descriptions within the patent documentation to provide accurate guidance. . Understanding these details is crucial for technical teams evaluating the feasibility of adopting this technology for their own production lines.
Q: Why is acetone cyanohydrin preferred over solid cyanide salts in this synthesis?
A: Acetone cyanohydrin is a liquid at room temperature, allowing for pipeline conveyance and sealed operations. This eliminates the need for manual weighing of highly toxic solid sodium or potassium cyanide, significantly improving production safety and reducing occupational health risks.
Q: What is the expected yield and purity of the final hydrochloride salt?
A: The patented process achieves a total yield of over 80% from the starting material. The final hydrochloride salt demonstrates high chemical purity, typically exceeding 99.7%, with no detectable enantiomers, ensuring suitability for stringent pharmaceutical applications.
Q: How does this route impact the scalability of antiviral intermediate manufacturing?
A: By shortening the synthetic route and removing hazardous solid handling steps, the process facilitates easier commercial scale-up. The use of common solvents like methanol and isopropanol further simplifies solvent recovery and waste management, enhancing overall supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of this intermediate in the global supply chain for antiviral therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest industry standards. Our infrastructure is designed to handle complex chemistries safely, leveraging the advantages of liquid-phase reagents to maximize efficiency and safety. We understand that time-to-market is crucial, and our optimized processes are ready for immediate technology transfer and scale-up.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us be your partner in delivering high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
