Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazol-3-one Intermediates for Scalable Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and safer synthetic routes to bioactive scaffolds. A significant breakthrough in this domain is detailed in patent CN112538054B, which discloses a robust preparation method for 1,2,4-triazole-3-ketone compounds. These nitrogen-containing five-membered heterocycles are pivotal structural motifs found in a vast array of biologically active molecules, exhibiting potent antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the biological context below, these cores serve as critical pharmacophores in diverse therapeutic agents ranging from PPARα agonists to antifungal drugs like Itraconazole.
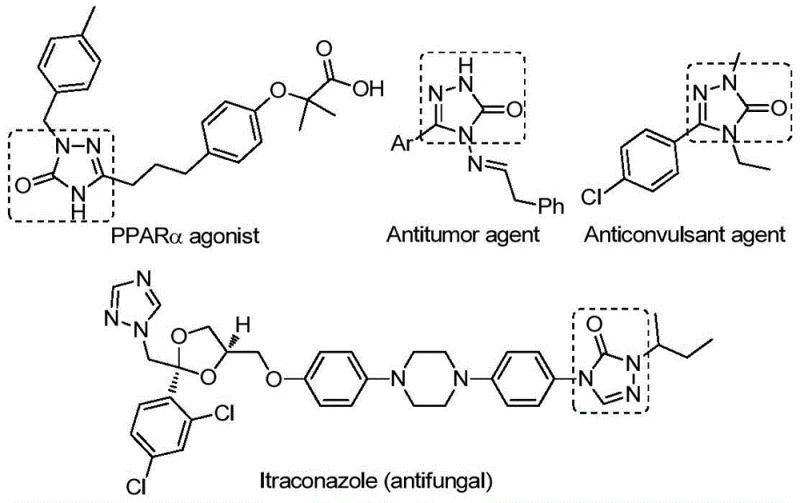
The patented methodology represents a paradigm shift from conventional synthesis strategies by employing a transition metal palladium-catalyzed carbonylation tandem cyclization reaction. By utilizing inexpensive and readily available chlorinated hydrazones and sodium azide as starting materials, coupled with a solid carbon monoxide substitute, this process achieves high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology offers a compelling value proposition through its operational simplicity and the ability to synthesize derivatives with varied substituents, thereby widening the practical applicability for drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazol-3-one scaffold has been fraught with synthetic challenges that hinder efficient large-scale production. Traditional literature reports describe several common pathways, such as the cyclization of benzoyl hydrazide with urea under potassium hydroxide catalysis, or the tandem cyclization of hydrazides with isocyanates. Other methods involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines. These legacy processes are severely constrained by苛刻 reaction conditions that often require extreme temperatures or strong bases, leading to safety concerns and energy inefficiencies. Furthermore, many of these routes necessitate the pre-activation of substrates, adding extra synthetic steps that reduce overall atom economy and increase waste generation. Perhaps most critically for process chemists, these conventional methods frequently suffer from low yields and a narrow substrate scope, failing to tolerate sensitive functional groups that are often required in modern medicinal chemistry campaigns.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach outlined in the patent leverages a sophisticated palladium-catalyzed system to streamline the synthesis into a single, efficient operation. The core innovation lies in the use of chlorohydrazones as electrophilic partners and sodium azide as a nitrogen source, mediated by a catalyst system comprising Pd2(dba)3 and Xantphos. Crucially, the method employs TFBen (1,3,5-tricarboxylic acid phenol ester) as a safe, solid surrogate for carbon monoxide gas, eliminating the need for high-pressure CO cylinders and specialized gas-handling infrastructure. This transformation proceeds smoothly in aprotic solvents like 1,4-dioxane at a moderate temperature of 100°C. The result is a highly versatile protocol that not only delivers superior yields but also demonstrates remarkable tolerance for diverse functional groups, including halogens, alkyl chains, and aryl substituents. This flexibility allows for the rapid generation of compound libraries, addressing the critical need for cost reduction in pharmaceutical intermediate manufacturing by minimizing step counts and maximizing throughput.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
To fully appreciate the elegance of this transformation, one must delve into the intricate catalytic cycle that drives the formation of the triazole ring. The reaction is postulated to initiate with the oxidative addition of the zero-valent palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate (Structure II), generating a reactive divalent palladium intermediate. Simultaneously, the TFBen additive undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the newly formed carbon-palladium bond, creating an acyl-palladium species. This acyl intermediate subsequently reacts with sodium azide to form an acyl azide compound, which is the precursor for the key ring-closing event. The acyl azide then undergoes a Curtius rearrangement, a thermally driven process that expels nitrogen gas and generates a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs where the adjacent nitrogen atom attacks the electrophilic carbon of the isocyanate group, closing the five-membered ring to yield the final 1,2,4-triazol-3-one product (Structure I). This mechanistic pathway highlights the precision of modern organometallic chemistry in constructing complex heterocycles from simple precursors.

Beyond the primary catalytic cycle, the control of impurities is a paramount concern for any process intended for high-purity pharmaceutical intermediate production. The choice of ligands, specifically Xantphos in this system, plays a critical role in stabilizing the palladium center and preventing the formation of palladium black or other inactive species that could lead to side reactions. Furthermore, the use of a solid CO source like TFBen ensures a steady, controlled release of carbon monoxide, preventing the accumulation of free CO that could lead to over-carbonylation or other off-cycle pathways. The reaction conditions, particularly the temperature range of 100°C to 120°C and the reaction time of 16 to 30 hours, are optimized to ensure complete conversion of the starting material while minimizing thermal degradation of the sensitive azide and isocyanate intermediates. Post-reaction processing involves straightforward filtration and silica gel treatment followed by column chromatography, a standard purification technique that effectively removes residual palladium species and organic byproducts, ensuring the final product meets stringent quality specifications required for downstream drug synthesis.
How to Synthesize 1,2,4-Triazol-3-one Efficiently
The synthesis of these valuable heterocycles is designed to be operationally simple, making it accessible for both laboratory-scale discovery and pilot-scale production. The general procedure involves charging a reaction vessel, such as a Schlenk tube, with the palladium catalyst, the bidentate phosphine ligand, the CO source (TFBen), the specific chlorohydrazone substrate, and sodium azide. An appropriate organic solvent, preferably 1,4-dioxane due to its superior solubilizing properties and reaction promotion capabilities, is added to the mixture. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the guide below to ensure reproducibility and safety.
- Charge a reaction vessel with Pd2(dba)3 catalyst, Xantphos ligand, TFBen (CO source), chlorohydrazone substrate, and sodium azide in an aprotic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to allow for oxidative addition, CO insertion, and cyclization.
- Upon completion, filter the mixture, perform silica gel treatment, and purify the crude product via column chromatography to isolate the target 1,2,4-triazol-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional synthesis methods to this novel palladium-catalyzed protocol offers substantial strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Chlorohydrazones and sodium azide are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of supply disruption compared to specialized reagents like isocyanates or activated hydrazides which may have limited vendors. Additionally, the elimination of gaseous carbon monoxide from the process equation removes a significant logistical and safety burden, as there is no longer a need for specialized gas cylinders, leak detection systems, or high-pressure reactors. This shift inherently lowers the capital expenditure required for setting up production lines and reduces the regulatory compliance overhead associated with handling toxic gases.
- Cost Reduction in Manufacturing: The economic impact of this new method is profound, primarily driven by the use of inexpensive starting materials and the high atom efficiency of the tandem reaction. By avoiding multi-step pre-activation sequences and utilizing a catalytic amount of palladium rather than stoichiometric reagents, the overall cost of goods sold (COGS) is significantly reduced. The high yields reported across a broad range of substrates mean that less raw material is wasted, and the downstream purification burden is lighter, further driving down processing costs. Moreover, the use of a solid CO surrogate eliminates the need for expensive high-pressure autoclaves, allowing the reaction to be run in standard glass-lined or stainless steel reactors, which represents a massive saving in equipment investment and maintenance.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is non-negotiable for continuous pharmaceutical manufacturing. This process enhances reliability by relying on robust, shelf-stable reagents that do not require cryogenic storage or special transport conditions. Sodium azide and the chlorohydrazone precursors are stable solids that can be stocked in bulk without significant degradation, ensuring that production schedules are not impacted by just-in-time delivery failures. The broad substrate scope also means that if a specific derivative is needed, the same platform technology can be applied immediately without developing a completely new synthetic route, allowing for agile responses to changing market demands or clinical trial requirements.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but this methodology is inherently scalable due to its homogeneous nature and moderate operating conditions. The reaction can be easily transferred from milligram scale in the lab to kilogram or tonnage scale in the plant without fundamental changes to the chemistry. From an environmental perspective, the process aligns well with green chemistry principles by reducing the number of synthetic steps and avoiding the use of hazardous gaseous reagents. The waste stream is primarily composed of organic solvents which can be recovered and recycled, and the absence of heavy metal waste (beyond the trace palladium which is removed during purification) simplifies wastewater treatment and disposal, ensuring compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of adopting this route for your specific manufacturing needs.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require harsh conditions, pre-activated substrates, or toxic gases, this novel approach utilizes cheap chlorohydrazones and solid CO sources (TFBen) under mild conditions (100°C), offering broader substrate compatibility and higher yields.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol uses commercially available reagents and standard organic solvents like dioxane. The avoidance of high-pressure carbon monoxide gas significantly simplifies safety protocols and equipment requirements, facilitating easier scale-up from gram to tonnage levels.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a safe and efficient solid surrogate for carbon monoxide. Under heating conditions, it releases CO in situ, which inserts into the palladium-carbon bond to form the necessary acyl-palladium intermediate for cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazol-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our infrastructure is designed to meet the demanding timelines and quality standards of the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data for our existing inventory and conduct comprehensive route feasibility assessments to determine the best path forward for your synthesis needs. Let us be your partner in bringing life-saving medicines to market faster and more efficiently.
