Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking robust, safe, and scalable routes to fluorinated heterocycles, particularly 1,2,3-triazoles, due to their profound impact on metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a simpler, base-promoted protocol that eliminates the need for hazardous azide reagents. For R&D directors and procurement specialists, this innovation offers a compelling value proposition: enhanced safety profiles, reduced reliance on precious metal catalysts, and a streamlined workflow that facilitates the rapid generation of diverse chemical libraries for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloadditions (CuAAC) or organocatalytic reactions involving organic azides and trifluoromethyl ketones. While effective, these conventional pathways suffer from critical drawbacks that hinder large-scale manufacturing. The primary concern is the inherent instability and toxicity of organic azides, which pose severe explosion hazards and require specialized handling infrastructure, thereby inflating operational costs. Furthermore, copper-catalyzed routes necessitate rigorous purification steps to remove trace heavy metals to meet stringent regulatory limits for Active Pharmaceutical Ingredients (APIs). These additional purification stages not only extend lead times but also result in substantial yield losses, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
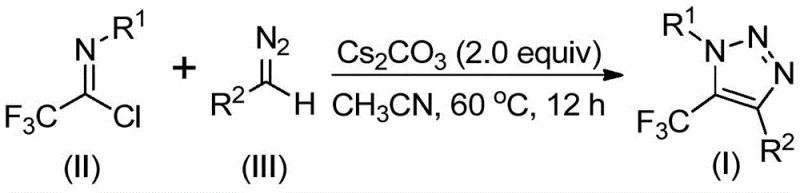
In stark contrast, the methodology described in patent CN113121462B utilizes readily available trifluoroethylimidoyl chlorides and diazo compounds as starting materials, completely bypassing the use of explosive azides and toxic transition metals. As illustrated in the reaction scheme below, this base-promoted cyclization operates under mild conditions, typically employing cesium carbonate in acetonitrile. This approach not only mitigates safety risks associated with azide handling but also simplifies the downstream processing by eliminating the need for complex metal scavenging protocols. The versatility of this method allows for the introduction of various functional groups at the N-1 and C-4 positions, providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies without the logistical burdens of traditional synthesis.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation is elegantly simple yet highly efficient, driven by the nucleophilic character of the diazo species. The reaction is believed to initiate with a base-promoted intermolecular nucleophilic addition of the diazo compound to the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion. Following this addition-elimination sequence, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is crucial as it closes the triazole ring, establishing the stable 1,2,3-triazole core substituted with the trifluoromethyl group at the 5-position. The use of cesium carbonate is particularly advantageous here, as the cesium cation likely stabilizes the developing negative charge on the nitrogen or oxygen atoms during the transition state, lowering the activation energy and driving the reaction to completion at moderate temperatures.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Since no transition metals are involved, the risk of metal-induced side reactions, such as Glaser coupling of alkynes or oxidative degradation, is entirely removed. The primary byproducts are typically inorganic salts derived from the base and the leaving group, which are easily removed during the aqueous workup or filtration steps. This clean reaction profile ensures that the crude product possesses high purity, reducing the burden on final purification stages. For quality control teams, this translates to a more predictable impurity profile and easier validation of the manufacturing process, which is essential for maintaining compliance with Good Manufacturing Practices (GMP) in the production of clinical trial materials.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves combining the trifluoroethylimidoyl chloride and the diazo compound in an aprotic solvent, with cesium carbonate serving as the promoter. The reaction mixture is then heated to a temperature range of 50°C to 70°C, typically around 60°C, and stirred for a duration of 8 to 16 hours. This thermal window is broad enough to accommodate various substrate electronic properties while remaining energy-efficient. Upon completion, the reaction mixture is filtered to remove inorganic salts, and the filtrate is concentrated. The resulting crude material can be purified via standard silica gel column chromatography to afford the target triazole derivatives in good to excellent yields, as demonstrated across multiple examples in the patent data.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free, azide-free technology presents a strategic opportunity to optimize cost structures and enhance supply reliability. By shifting away from hazardous azide chemistry, companies can significantly reduce the costs associated with specialized safety equipment, hazardous waste disposal, and insurance premiums. The elimination of copper catalysts further drives cost reduction in API manufacturing by removing the expense of precious metals and the complex ligands often required to maintain catalytic activity. Moreover, the simplified workup procedure, which avoids tedious metal scavenging steps, shortens the overall production cycle time, allowing for faster turnover of batches and improved responsiveness to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and hazardous azide reagents fundamentally alters the cost equation of triazole production. Without the need for expensive copper salts or phosphine ligands, raw material costs are inherently lower. Additionally, the simplified purification process reduces solvent consumption and labor hours dedicated to metal removal, leading to substantial operational savings. This economic efficiency makes the process highly attractive for the commercial scale-up of complex pharmaceutical intermediates where margin pressure is often intense.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically trifluoroethylimidoyl chlorides and diazo compounds, are generally more stable and easier to source in bulk quantities compared to sensitive organic azides. This stability ensures a more robust supply chain with fewer disruptions caused by the degradation of reactive intermediates during storage or transport. Consequently, manufacturers can maintain higher inventory levels of key precursors with confidence, ensuring continuous production schedules and reducing the lead time for high-purity intermediates required for urgent drug development projects.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of heavy metals align perfectly with modern green chemistry principles and environmental regulations. Scaling this process from gram to kilogram or ton scales does not introduce the exponential safety risks associated with azide accumulation. Furthermore, the waste stream is less toxic and easier to treat, facilitating compliance with increasingly strict environmental discharge standards. This environmental compatibility not only future-proofs the manufacturing process but also enhances the corporate sustainability profile of the producing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patent utilizes stable diazo compounds and imidoyl chlorides, significantly reducing safety risks and handling costs.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds in acetonitrile at moderate temperatures between 50°C and 70°C, using cesium carbonate as a promoter, avoiding the need for expensive transition metal catalysts.
Q: Can this process be scaled for industrial API intermediate production?
A: Yes, the patent explicitly demonstrates scalability to gram levels with simple workup procedures like filtration and column chromatography, making it suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the next generation of fluorinated drugs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate meets the highest industry standards for potency and impurity control.
We invite you to collaborate with us to leverage this advanced chemistry for your pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for high-performance pharmaceutical intermediates.
