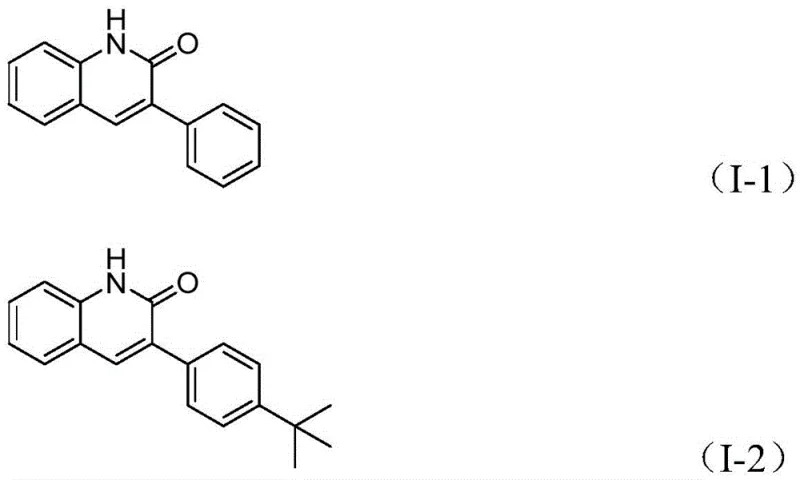
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
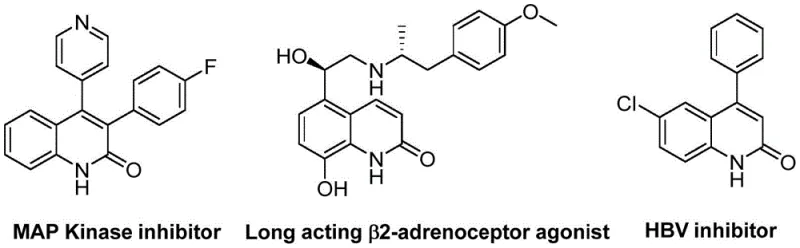
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable pathways to access bioactive scaffolds. A significant breakthrough in this domain is documented in patent CN113045489B, which discloses a robust preparation method for 3-arylquinolin-2(1H)one derivatives. These compounds are not merely academic curiosities; they represent a critical class of pharmacophores found in numerous natural products and therapeutic agents, ranging from antibiotics and antiplatelet drugs to potent antitumor candidates and endothelin receptor antagonists. The strategic importance of these molecules cannot be overstated, as they serve as pivotal building blocks in the development of next-generation medicines. The disclosed methodology leverages a sophisticated palladium-catalyzed aminocarbonylation strategy that fundamentally alters the synthetic logic traditionally employed for constructing the quinolinone core.
What sets this invention apart is its ingenious utilization of benzisoxazole as a dual-purpose reagent. In conventional synthetic planning, introducing both the nitrogen atom and the carbonyl functionality often requires multiple steps or distinct reagents, each adding to the cumulative cost and waste profile of the process. However, this novel approach exploits the latent reactivity of benzisoxazole, allowing it to act simultaneously as the nitrogen source and the formyl donor. This atom-economical design drastically simplifies the reaction setup and reduces the reliance on hazardous gaseous carbon monoxide sources, which are typically required for carbonylation reactions. By integrating these features, the process offers a streamlined route that is particularly attractive for industrial applications where safety and operational simplicity are paramount concerns for a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one skeleton has relied heavily on classical condensation reactions such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods have served the chemical community well for decades, they are increasingly viewed through the lens of modern green chemistry as suboptimal for large-scale manufacturing. The Friedlander reaction, for instance, often necessitates the use of strong acids or bases and high temperatures, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles that are difficult to separate. Furthermore, many traditional routes require pre-functionalized starting materials that are themselves expensive or difficult to synthesize, creating a bottleneck in the supply chain. The reliance on gaseous carbon monoxide for carbonylation variants introduces significant safety hazards, requiring specialized high-pressure equipment and rigorous safety protocols that inflate capital expenditure and operational overheads.
The Novel Approach
In stark contrast, the methodology described in CN113045489B offers a paradigm shift by employing a transition metal-catalyzed cascade that proceeds under relatively mild conditions. The reaction utilizes readily available benzisoxazoles and benzyl chlorides as the primary feedstocks, both of which are commodity chemicals with stable global supply chains. The use of molybdenum hexacarbonyl as a solid carbon monoxide surrogate eliminates the need for handling toxic CO gas, thereby enhancing workplace safety and reducing regulatory burdens. The reaction demonstrates exceptional functional group tolerance, accommodating electron-donating and electron-withdrawing substituents on both the benzisoxazole and the benzyl chloride moieties without compromising yield. This versatility allows for the rapid generation of diverse libraries of 3-arylquinolin-2(1H)one derivatives, facilitating the optimization of structure-activity relationships in drug discovery programs.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
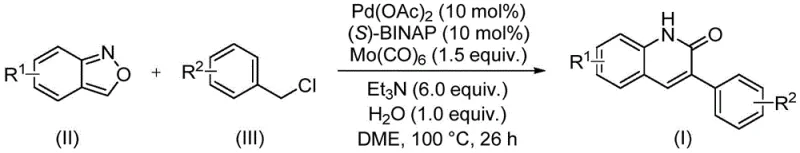
The core of this transformation lies in a sophisticated palladium catalytic cycle that orchestrates the cleavage of the N-O bond in benzisoxazole and the subsequent insertion of a carbonyl unit. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step forms an organopalladium intermediate that is poised for carbonyl insertion. Uniquely, the carbonyl source is derived from the decomposition of molybdenum hexacarbonyl under the reaction conditions, releasing CO in a controlled manner that matches the kinetics of the catalytic cycle. The coordination of the benzisoxazole nitrogen to the palladium center facilitates the ring-opening of the isoxazole moiety, effectively delivering the nitrogen atom required for the lactam formation while simultaneously providing the necessary carbon framework.
Following the migratory insertion of the carbonyl group into the palladium-carbon bond, the cycle proceeds through a reductive elimination step that constructs the final C-C bond of the quinolinone ring system. This sequence regenerates the active palladium catalyst, allowing the turnover to continue. The presence of water and triethylamine plays a crucial role in proton management and neutralizing the hydrochloric acid byproduct generated from the benzyl chloride, ensuring the reaction medium remains conducive to catalysis. The high regioselectivity observed in this process minimizes the formation of isomeric byproducts, which is a critical factor for achieving high-purity pharmaceutical intermediates. The robust nature of this catalytic system ensures that even with complex substrates, the reaction proceeds with high efficiency, yielding products that often require minimal purification beyond standard column chromatography.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
The practical execution of this synthesis is designed to be straightforward, minimizing the need for specialized equipment or exotic reagents. The protocol involves charging a sealed reaction vessel with the palladium catalyst system, the solid CO source, the base, and the two primary organic substrates in a polar aprotic solvent such as dimethoxyethane (DME). The mixture is then heated to a moderate temperature, typically around 100°C, and maintained for a duration sufficient to drive the reaction to completion, usually approximately 26 hours. This extended reaction time ensures full conversion of the starting materials, maximizing the overall yield and reducing the burden on downstream purification processes. For detailed procedural specifics regarding reagent ratios and workup techniques, please refer to the standardized synthesis guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route addresses several critical pain points that procurement managers and supply chain directors face when sourcing complex heterocyclic intermediates. The primary advantage lies in the substantial cost reduction in API manufacturing enabled by the use of low-cost, commodity-grade starting materials. Benzisoxazoles and substituted benzyl chlorides are produced on a multi-ton scale globally, ensuring price stability and availability even during market fluctuations. By eliminating the need for custom-synthesized precursors or hazardous gaseous reagents, the overall bill of materials is significantly optimized. Furthermore, the operational simplicity of the process translates directly into lower manufacturing costs, as it does not require high-pressure reactors or extensive safety infrastructure associated with handling toxic gases.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the dual functionality of benzisoxazole, which acts as both a reactant and a reagent, effectively reducing the number of distinct chemical inputs required. This consolidation of reagents leads to a drastic simplification of the supply chain logistics and inventory management. Additionally, the use of a solid carbon monoxide source like molybdenum hexacarbonyl removes the capital intensity associated with gas handling systems, allowing for production in standard glass-lined or stainless steel reactors. The high yields reported across a broad substrate scope mean that less raw material is wasted, further driving down the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Securing a consistent supply of high-quality intermediates is a top priority for any pharmaceutical company. This method relies on feedstocks that are widely available from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures that production schedules are not easily disrupted by minor variations in raw material quality or environmental factors. Moreover, the scalability of the process from gram to kilogram scale has been demonstrated, providing confidence that supply can be ramped up quickly to meet clinical trial demands or commercial launch volumes without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: In an era of increasing environmental scrutiny, the green chemistry attributes of this synthesis offer a distinct competitive advantage. The reaction generates minimal hazardous waste compared to traditional methods that utilize stoichiometric amounts of toxic reagents. The solvent system is compatible with standard recovery and recycling protocols, reducing the environmental footprint of the manufacturing process. The absence of heavy metal contaminants in the final product, thanks to the efficient catalytic turnover and straightforward purification, simplifies the regulatory filing process. This alignment with sustainability goals not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What are the primary advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl group donor, eliminating the need for separate carbonylation reagents and simplifying the reaction stoichiometry significantly.
Q: Is this method suitable for large-scale pharmaceutical production?
A: Yes, the process utilizes commercially available starting materials like benzyl chlorides and operates under relatively mild thermal conditions (100°C), making it highly amenable to commercial scale-up of complex pharmaceutical intermediates.
Q: How does this method compare to traditional Friedlander reactions?
A: Unlike traditional methods that often require harsh acidic conditions or pre-functionalized substrates, this palladium-catalyzed approach offers broader functional group tolerance and avoids the use of corrosive reagents, enhancing safety and purity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN113045489B for accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest standards of safety and efficiency, while our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting requirements of the global pharmaceutical industry. We are committed to delivering high-purity 3-arylquinolin-2(1H)one derivatives that serve as the foundation for your next breakthrough therapy.
We invite you to leverage our technical expertise to optimize your supply chain and reduce time-to-market. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable network of chemical innovation and manufacturing excellence dedicated to supporting your long-term success in the competitive healthcare market.
