Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
Introduction to Next-Generation Quinolinone Synthesis
The quinolin-2(1H)one scaffold represents a cornerstone structure in modern medicinal chemistry, underpinning a vast array of biologically active compounds ranging from antibiotics and antiplatelet agents to potent antitumor drugs and endothelin receptor antagonists. As depicted in the structural diversity of bioactive molecules, the ability to efficiently construct this heterocyclic core is paramount for pharmaceutical innovation. 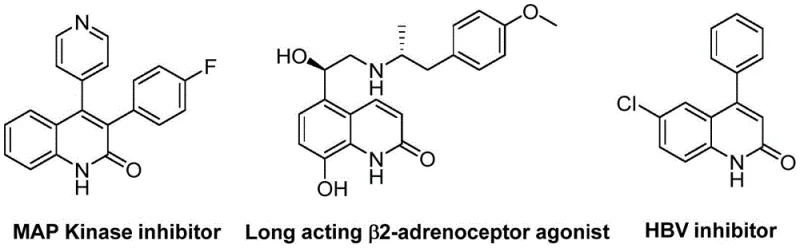 Recent advancements documented in patent CN113045489B introduce a transformative approach to accessing these valuable intermediates through a palladium-catalyzed aminocarbonylation strategy. This methodology not only addresses the synthetic challenges associated with traditional routes but also aligns perfectly with the industry's demand for reliable pharmaceutical intermediate supplier capabilities that prioritize efficiency and sustainability. By leveraging benzisoxazole as a unique dual-purpose reagent, this invention opens new avenues for the rapid assembly of complex heterocycles essential for drug discovery pipelines.
Recent advancements documented in patent CN113045489B introduce a transformative approach to accessing these valuable intermediates through a palladium-catalyzed aminocarbonylation strategy. This methodology not only addresses the synthetic challenges associated with traditional routes but also aligns perfectly with the industry's demand for reliable pharmaceutical intermediate supplier capabilities that prioritize efficiency and sustainability. By leveraging benzisoxazole as a unique dual-purpose reagent, this invention opens new avenues for the rapid assembly of complex heterocycles essential for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one framework has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these established protocols have served the community for decades, they are increasingly viewed as suboptimal for modern large-scale manufacturing due to inherent inefficiencies. Conventional routes often necessitate harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to significant degradation of sensitive functional groups and complicate downstream purification processes. Furthermore, many traditional strategies suffer from poor atom economy and require multi-step sequences to install the necessary carbonyl and nitrogen functionalities separately, resulting in increased waste generation and higher operational costs. The reliance on volatile or toxic reagents in some of these older methods also poses substantial safety hazards and environmental compliance burdens, making them less attractive for green chemistry initiatives in contemporary API manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the novel palladium-catalyzed aminocarbonylation reaction described in the patent offers a streamlined, one-pot solution that fundamentally reshapes the synthetic landscape. This innovative route utilizes readily available benzisoxazole derivatives and benzyl chloride compounds as starting materials, reacting them under mild conditions to directly yield the target 3-arylquinolin-2(1H)one derivatives. The brilliance of this approach lies in the dual functionality of benzisoxazole, which acts simultaneously as the nitrogen source and the formyl source, thereby collapsing multiple synthetic steps into a single transformation. This consolidation not only accelerates the synthesis timeline but also dramatically improves the overall yield and purity profile of the final product. The method's compatibility with a wide range of functional groups ensures that diverse chemical libraries can be generated rapidly, providing R&D teams with the flexibility needed to explore structure-activity relationships without being constrained by synthetic limitations.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological breakthrough is a sophisticated catalytic cycle driven by a palladium system ligated with chiral phosphines, specifically employing palladium acetate and (S)-1,1'-binaphthyl-2,2'-bisdiphenylphosphine ((S)-BINAP). The reaction mechanism initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species, forming an organopalladium intermediate. Crucially, the presence of molybdenum hexacarbonyl (Mo(CO)6) serves as a safe and effective solid surrogate for carbon monoxide gas, releasing CO in situ to facilitate the carbonylation step without the need for high-pressure gas infrastructure. 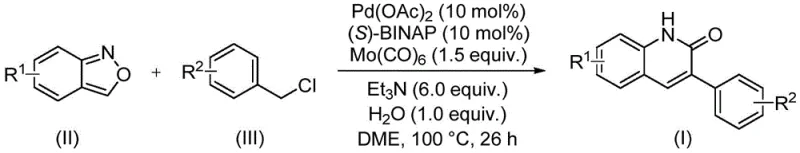 Following carbonyl insertion, the benzisoxazole molecule undergoes a ring-opening process, likely facilitated by the basic environment provided by triethylamine and the presence of water, which releases the necessary nitrogen and formyl components to close the quinolinone ring. This intricate dance of bond breaking and forming is meticulously balanced to ensure high turnover numbers and minimal side reactions, showcasing the robustness of the catalytic system.
Following carbonyl insertion, the benzisoxazole molecule undergoes a ring-opening process, likely facilitated by the basic environment provided by triethylamine and the presence of water, which releases the necessary nitrogen and formyl components to close the quinolinone ring. This intricate dance of bond breaking and forming is meticulously balanced to ensure high turnover numbers and minimal side reactions, showcasing the robustness of the catalytic system.
From an impurity control perspective, the choice of ligands and the specific reaction parameters play a pivotal role in maintaining product integrity. The use of (S)-BINAP helps stabilize the palladium center, preventing the formation of palladium black and other inactive species that could contaminate the final product. Additionally, the mild temperature range of 90 to 110°C prevents thermal decomposition of sensitive intermediates, ensuring a clean reaction profile. The inclusion of water as a co-reagent is particularly ingenious, as it assists in the hydrolysis steps required for ring closure while remaining compatible with the organometallic catalyst. This precise control over the reaction environment results in products with exceptional purity, reducing the burden on downstream purification units and enhancing the overall viability of the process for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to achieve the reported high yields ranging from 68% to 97%. The process begins by combining the palladium catalyst, chiral ligand, molybdenum carbonyl source, base, and water with the specific benzisoxazole and benzyl chloride substrates in a sealed vessel. Ethylene glycol dimethyl ether (DME) is utilized as the solvent of choice due to its ability to effectively dissolve the organic substrates while maintaining stability under the reaction conditions. The detailed standardized synthesis steps below outline the precise stoichiometry and operational sequence required to replicate these results successfully.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride compound.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture to ensure homogeneity before heating.
- Heat the reaction mixture to 100°C for 26 hours, then filter, mix with silica gel, and purify via column chromatography to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The strategic selection of starting materials and reagents directly addresses common pain points in the sourcing of high-purity pharmaceutical intermediates, creating a more resilient and cost-effective supply chain architecture. By eliminating the need for specialized high-pressure equipment and hazardous gas handling, the process lowers the barrier to entry for manufacturing partners and reduces the overall capital expenditure required for production facilities.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven primarily by the simplification of the synthetic route and the use of commodity chemicals. Since benzisoxazole serves as both the nitrogen and carbon source, there is no need to purchase and handle separate formylation reagents, which are often expensive and difficult to manage. Furthermore, the use of molybdenum hexacarbonyl as a solid CO source eliminates the logistical costs and safety premiums associated with transporting and storing high-pressure carbon monoxide cylinders. The high reaction efficiency and yields observed across various substrates mean that less raw material is wasted, leading to substantial cost savings in material consumption and waste disposal fees.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the commercial availability of the key starting materials. Both benzisoxazole derivatives and benzyl chloride compounds are widely produced commodity chemicals with stable global supply networks, reducing the risk of shortages that often plague specialized reagents. The robustness of the reaction conditions, which tolerate a broad spectrum of functional groups, allows manufacturers to source lower-grade or more abundant variants of substrates without compromising the final product quality. This flexibility ensures that production schedules remain uninterrupted even when specific grades of raw materials face temporary market volatility, providing a reliable buffer against supply chain disruptions.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the homogeneous nature of the reaction and the absence of extreme physical conditions. The operation at atmospheric pressure (within a sealed tube) and moderate temperatures simplifies reactor design and safety protocols, making it easier to transfer technology from R&D to full-scale commercial production. Additionally, the reduced generation of hazardous byproducts and the avoidance of toxic gases align with increasingly stringent environmental regulations. This green chemistry profile not only minimizes the environmental footprint but also streamlines the permitting process for new manufacturing lines, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity for technical teams evaluating this technology for adoption.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, eliminating the need for separate formylation reagents and simplifying the synthetic route significantly.
Q: How does this method improve safety compared to traditional carbonylation?
A: By utilizing molybdenum hexacarbonyl (Mo(CO)6) as a solid CO surrogate instead of high-pressure carbon monoxide gas, the process drastically reduces safety risks and infrastructure requirements for commercial production.
Q: What is the functional group tolerance of this catalytic system?
A: The protocol demonstrates excellent tolerance for various substituents including halogens, alkoxy groups, cyano, and trifluoromethyl groups, allowing for the synthesis of diverse pharmacological scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
As the pharmaceutical industry continues to demand more efficient and sustainable pathways for complex molecule synthesis, NINGBO INNO PHARMCHEM stands ready to leverage this cutting-edge technology for your project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and reliable. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-arylquinolin-2(1H)one derivatives meets the highest international standards for API intermediates.
We invite you to engage with our technical procurement team to discuss how this novel palladium-catalyzed route can optimize your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development programs.
