Advanced Palladium-Catalyzed Synthesis of Bioactive 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Bioactive 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds for Commercial Scale-Up
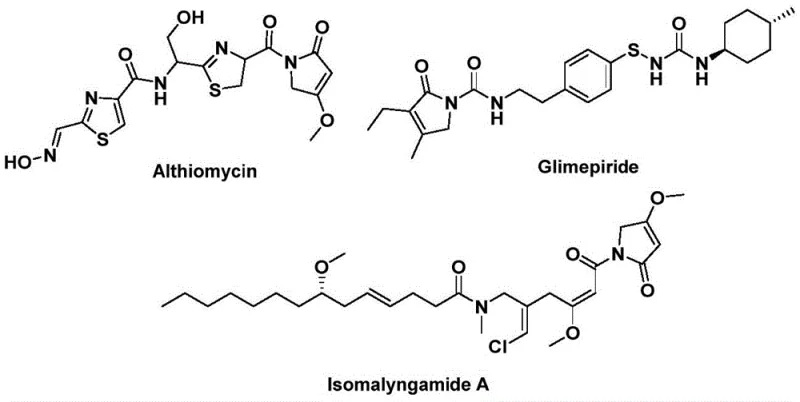
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for a diverse array of bioactive natural products and synthetic pharmaceuticals. Prominent examples include althiomycin, known for its significant antibacterial properties, glimepiride, a widely prescribed hypoglycemic agent for diabetes management, and isomalyngamide A, which exhibits potential anticancer activity. The strategic construction of this heterocyclic ring system is therefore of paramount importance to the global pharmaceutical supply chain. Recently, Chinese Patent CN112694430B disclosed a groundbreaking preparation method that utilizes a palladium-catalyzed bis-carbonylation strategy to access these valuable compounds efficiently. This technology leverages readily available propargylamines and benzyl chlorides, coupled with a solid carbon monoxide surrogate, to achieve high-yielding synthesis under relatively mild conditions, marking a significant advancement over traditional multi-step approaches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,5-dihydro-2H-pyrrol-2-one derivatives has relied on classical condensation reactions or multi-step cyclization protocols that often suffer from significant drawbacks in an industrial setting. Traditional routes frequently require harsh reaction conditions, such as strong acids or bases at elevated temperatures, which can lead to poor atom economy and the generation of substantial chemical waste. Furthermore, many conventional methods utilize gaseous carbon monoxide directly, which poses severe safety hazards due to its toxicity and flammability, necessitating expensive high-pressure reactors and rigorous safety infrastructure. These factors collectively contribute to increased operational expenditures and complex regulatory compliance burdens, making the commercial scale-up of complex pharmaceutical intermediates challenging and costly for procurement teams seeking reliable supply chains.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN112694430B introduces a streamlined, one-pot catalytic process that elegantly circumvents these historical bottlenecks. By employing a palladium catalyst system in conjunction with a solid phenol ester (TFBen) as a safe and controllable carbon monoxide source, this novel approach enables the direct assembly of the pyrrolone ring from simple starting materials. The reaction proceeds through a sophisticated bis-carbonylation cascade, effectively inserting two carbonyl units into the molecular framework in a single operational step. This not only drastically reduces the number of unit operations required but also enhances the overall reaction efficiency and substrate compatibility. The ability to tolerate a wide range of functional groups, including halogens and trifluoromethyl groups, allows for the rapid generation of diverse chemical libraries, providing R&D directors with a powerful tool for lead optimization without the need for extensive protecting group strategies.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the precision of modern organometallic catalysis, initiated by the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate. This critical step generates a reactive benzylpalladium intermediate, which subsequently undergoes migratory insertion with carbon monoxide liberated in situ from the thermal decomposition of the 1,3,5-tricarboxylic acid phenol ester (TFBen). The resulting acylpalladium species then engages with the propargylamine component, triggering a cyclization event that forms a five-membered ring palladium intermediate. Following this, a second molecule of carbon monoxide is inserted into the palladium-carbon bond, expanding the coordination sphere to form a six-membered ring acylpalladium intermediate. The catalytic cycle is finally closed by a reductive elimination step, which releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium catalyst for subsequent turnover.
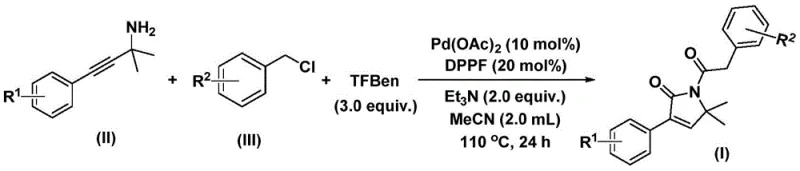
From an impurity control perspective, the use of the DPPF ligand plays a crucial role in stabilizing the palladium center and directing the regioselectivity of the carbonyl insertions. The steric and electronic properties of the ferrocene-based diphosphine ligand help suppress competing side reactions, such as homocoupling of the benzyl chloride or polymerization of the alkyne moiety, which are common pitfalls in palladium-catalyzed carbonylations. Furthermore, the controlled release of carbon monoxide from the solid surrogate ensures that the concentration of CO in the reaction medium remains optimal for the sequential insertion steps, preventing the formation of dicarbonyl byproducts or incomplete carbonylation species. This high level of chemoselectivity translates directly into a cleaner crude reaction profile, significantly reducing the burden on downstream purification processes and ensuring that the final API intermediates meet stringent quality standards.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
The practical execution of this synthesis is designed for robustness and scalability, utilizing standard laboratory equipment that can be easily adapted for pilot plant operations. The protocol involves charging a reaction vessel with palladium acetate, the DPPF ligand, triethylamine as a base, the solid CO surrogate TFBen, the specific propargylamine derivative, and the corresponding benzyl chloride in acetonitrile solvent. The mixture is then heated to 110°C and maintained for 24 hours to drive the reaction to completion. Detailed standardized synthesis steps, including precise molar ratios and workup procedures for specific derivatives like compounds I-1 through I-5, are provided in the technical guide below.
- Charge a reaction vessel with palladium acetate catalyst, DPPF ligand, triethylamine base, solid CO surrogate (TFBen), propargylamine, and benzyl chloride in acetonitrile solvent.
- Heat the reaction mixture to 110°C and maintain stirring for 24 hours to ensure complete conversion via the bis-carbonylation cascade.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity pyrrolone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from hazardous gaseous reagents to stable, solid surrogates fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility opens up a broader network of potential contract manufacturing organizations (CMOs), thereby enhancing supply chain resilience and reducing the risk of single-source bottlenecks. Additionally, the use of commodity chemicals like benzyl chloride and simple propargylamines as starting materials ensures a stable and cost-effective raw material supply, insulating the production process from the volatility often associated with specialized fine chemical precursors.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure results in significant capital expenditure savings, as there is no longer a need for specialized autoclaves or complex gas delivery systems. Furthermore, the one-pot nature of the reaction reduces solvent consumption, energy usage for heating and cooling between steps, and labor costs associated with multiple isolation and purification stages. The high atom economy of the bis-carbonylation process ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs and maximizing the value derived from each kilogram of input material.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against moisture and air, combined with the use of shelf-stable solid reagents, simplifies inventory management and reduces the risk of batch failures due to reagent degradation. The broad substrate scope means that a single manufacturing platform can be utilized to produce a wide variety of analogs by simply swapping the starting aryl halides or alkynes, allowing for agile response to changing market demands or clinical trial requirements without the need for extensive process re-validation.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer and more environmentally benign, aligning with green chemistry principles by avoiding toxic gases and reducing waste generation. This facilitates smoother regulatory approvals and environmental impact assessments, accelerating the timeline from process development to commercial launch. The simplicity of the post-treatment, involving basic filtration and chromatography, ensures that the process can be scaled from gram to ton quantities with minimal engineering challenges, supporting the seamless transition from R&D to full-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, based on the experimental data and mechanistic understanding provided in the patent literature. These insights are intended to assist technical teams in evaluating the feasibility of this method for their specific project needs.
Q: What are the primary advantages of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-trimethoxybenzene-derived phenol esters (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas cylinders and specialized containment equipment, drastically improving operational safety and simplifying the reaction setup for scale-up.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens (F, Cl, Br) and trifluoromethyl moieties on both the propargylamine and benzyl chloride substrates.
Q: How does this method impact the purity profile of the final intermediate?
A: The high chemoselectivity of the Pd/DPPF catalytic system minimizes side reactions and byproduct formation, resulting in crude reaction mixtures that are easier to purify, thereby ensuring stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development lifecycle. Our team of expert chemists has extensively evaluated the palladium-catalyzed bis-carbonylation technology described in Patent CN112694430B and confirmed its potential for delivering high-purity pyrrolone derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,5-dihydro-2H-pyrrol-2-one compound adheres to the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your path to market with confidence.
