Advanced Pd-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrol-2-One Production
Advanced Pd-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrol-2-One Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A recent breakthrough detailed in patent CN112694430B introduces a novel preparation method for 1,5-dihydro-2H-pyrrol-2-one compounds, a privileged structural backbone found in numerous bioactive natural products and drugs. This technology leverages a palladium-catalyzed bis-carbonylation strategy that replaces hazardous gaseous carbon monoxide with a solid surrogate, fundamentally altering the safety profile and operational simplicity of the synthesis. The significance of this scaffold cannot be overstated, as it serves as the core structure for potent antibiotics like Althiomycin, hypoglycemic agents such as Glimepiride, and anticancer candidates like Isomalyngamide A.
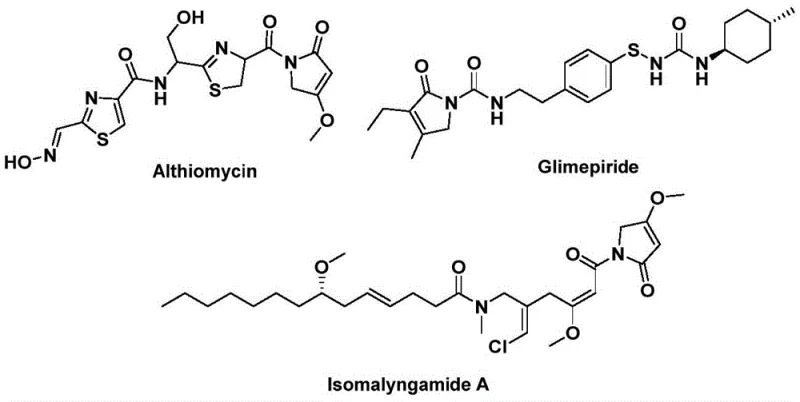
For R&D directors and process chemists, the ability to access these complex heterocycles in a single step from readily available starting materials represents a significant strategic advantage. The patent outlines a robust protocol that tolerates a wide variety of functional groups, ensuring that diverse analog libraries can be generated rapidly for structure-activity relationship (SAR) studies. By integrating this methodology into existing pipelines, organizations can accelerate the discovery phase while maintaining rigorous safety standards, positioning themselves as a reliable pharmaceutical intermediate supplier capable of delivering high-quality building blocks for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of carbonyl-containing heterocycles like pyrrolones has relied heavily on direct carbonylation using carbon monoxide gas. While effective, this approach presents severe logistical and safety challenges that hinder its widespread adoption in standard laboratory and manufacturing settings. The requirement for high-pressure equipment to handle toxic CO gas necessitates specialized infrastructure, extensive safety protocols, and highly trained personnel, all of which drive up operational costs and limit accessibility. Furthermore, conventional multi-step syntheses often involve harsh reagents, protecting group manipulations, and low atom economy, leading to significant waste generation and reduced overall yields. These inefficiencies create bottlenecks in the supply chain, extending lead times for critical intermediates and complicating the scale-up process for commercial production.
The Novel Approach
In stark contrast, the method disclosed in CN112694430B offers a transformative solution by utilizing 1,3,5-triacetoxybenzene (TFBen) as a solid carbon monoxide surrogate. This innovation allows the carbonylation reaction to proceed under atmospheric pressure conditions, effectively removing the hazards associated with gaseous CO. The reaction couples propargyl amines and benzyl chlorides in a one-pot process, streamlining the workflow and minimizing purification steps. This novel approach not only enhances operator safety but also improves the environmental footprint of the synthesis by reducing energy consumption and waste. For procurement managers, this translates to a more resilient supply chain where raw materials are cheaper and easier to source, and the manufacturing process is less susceptible to regulatory hurdles regarding hazardous gas handling.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The success of this synthesis lies in the intricate dance of the palladium catalytic cycle, which orchestrates the formation of two carbon-carbon bonds and the incorporation of two carbonyl units with high precision. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of the TFBen surrogate, inserts into this intermediate to form an acyl-palladium species. This acyl complex then undergoes nucleophilic attack by the propargyl amine, triggering a cyclization event that constructs the five-membered nitrogen-containing ring.
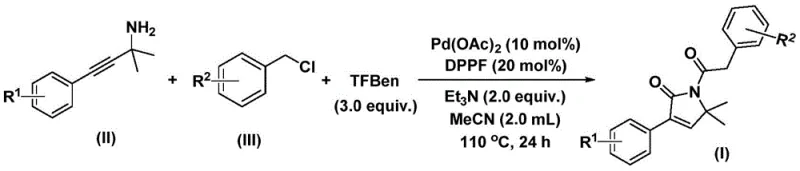
Following the initial cyclization, a second molecule of carbon monoxide inserts into the organopalladium species, expanding the coordination sphere before a final reductive elimination step releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst. This mechanistic pathway is highly tolerant of electronic variations on the aromatic rings, as evidenced by the successful synthesis of derivatives bearing electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, and trifluoromethyl. Understanding this mechanism is crucial for process optimization, as it highlights the importance of ligand selection (DPPP) and base choice (triethylamine) in stabilizing the key intermediates and driving the reaction to completion with minimal side product formation.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction conditions to maximize yield and purity. The protocol specifies the use of palladium acetate as the precatalyst and 1,1'-bis(diphenylphosphino)ferrocene (DPPP) as the ligand, dissolved in acetonitrile solvent. The reaction is heated to 110°C for 24 hours, ensuring sufficient energy for the CO surrogate decomposition and the subsequent catalytic turnover. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistency and quality control.
- Charge a reaction vessel with palladium acetate catalyst, DPPP ligand, triethylamine base, and the solid carbon monoxide substitute (1,3,5-triacetoxybenzene) in acetonitrile solvent.
- Add the propargyl amine substrate and benzyl chloride derivative to the mixture under inert atmosphere conditions.
- Heat the reaction mixture to 110°C and stir for 24 hours to ensure complete conversion, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of modern chemical procurement and supply chain management. The shift from hazardous gas handling to solid reagents significantly lowers the barrier to entry for manufacturing, allowing for production in facilities that may not be equipped for high-pressure carbonylations. This flexibility enhances supply chain reliability by diversifying the potential manufacturing base and reducing dependency on specialized gas suppliers. Moreover, the use of commodity chemicals like benzyl chlorides and propargyl amines ensures a stable and cost-effective raw material supply, insulating the production process from volatile market fluctuations associated with exotic reagents.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO gas infrastructure results in substantial capital expenditure savings and reduced operational overhead. By avoiding the need for specialized autoclaves and gas monitoring systems, manufacturers can allocate resources more efficiently. Additionally, the high reaction efficiency and one-step nature of the process minimize solvent usage and waste disposal costs, contributing to a leaner and more profitable manufacturing model that supports cost reduction in API manufacturing without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the commercial availability of all starting materials contribute to a highly reliable supply chain. Unlike processes dependent on unstable or hard-to-source reagents, this method utilizes bench-stable solids and liquids that can be stockpiled easily. This stability reduces the risk of production delays caused by raw material shortages, ensuring consistent delivery schedules for downstream customers who rely on timely access to high-purity pharmaceutical intermediates for their own drug development programs.
- Scalability and Environmental Compliance: The mild reaction conditions and simple workup procedure make this process exceptionally scalable from laboratory benchtop to industrial reactor sizes. The absence of toxic gas emissions aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, appealing to partners and investors who prioritize green chemistry principles in their supply chain decisions.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common questions regarding its implementation and performance. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this route into their existing portfolios.
Q: What is the primary safety advantage of this synthesis method?
A: This method utilizes 1,3,5-triacetoxybenzene (TFBen) as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous high-pressure CO gas cylinders typically required for carbonylation reactions.
Q: What is the typical yield range for these pyrrolone derivatives?
A: According to the patent data, the reaction demonstrates high efficiency with isolated yields ranging from 70% to 92% across various substrates, including those with electron-donating and electron-withdrawing groups.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses commercially available starting materials, operates at moderate temperatures (110°C), and employs a simple workup procedure involving filtration and chromatography, making it highly amenable to scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into viable commercial realities. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging technologies like the Pd-catalyzed bis-carbonylation described in CN112694430B, we offer our partners a competitive edge through superior quality and process efficiency.
We invite you to explore how our expertise can support your specific project needs. Whether you require custom synthesis of complex pyrrolone derivatives or optimization of existing routes, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your unique requirements. Let us collaborate to bring your next generation of therapeutic agents to market faster and more efficiently.
