Advanced Synthesis of Rucaparib Key Intermediate for Commercial Scale-up
Advanced Synthesis of Rucaparib Key Intermediate for Commercial Scale-up
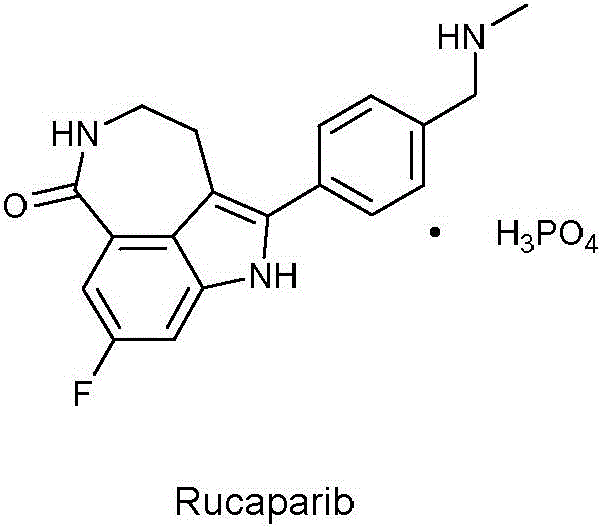
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with Poly (ADP-ribose) polymerase (PARP) inhibitors like Rucaparib playing a pivotal role in treating advanced ovarian cancer. As detailed in patent CN107954919B, a novel preparation method for the key intermediate of Rucaparib has been disclosed, offering a transformative approach to manufacturing this critical therapeutic agent. This patent introduces a streamlined synthetic route that bypasses the hazardous and costly steps associated with traditional methods, utilizing a robust Fischer indole synthesis followed by a controlled nitroaldol condensation. For global procurement and R&D teams, understanding this technological shift is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials. The structural complexity of Rucaparib, as shown below, demands precise synthetic control to ensure efficacy and safety in the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
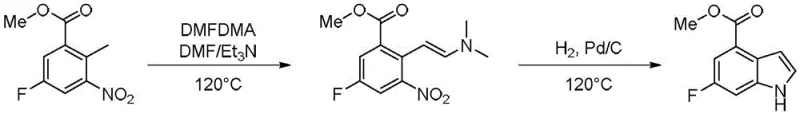
The Limitations of Conventional Methods
Historically, the synthesis of the indole core required for Rucaparib has relied on routes involving N,N-dimethylformamide dimethyl acetal (DMFDMA) and subsequent catalytic hydrogenation. As illustrated in the reaction scheme below, this conventional pathway necessitates harsh reaction conditions, specifically temperatures reaching 120°C, which poses significant thermal safety risks during scale-up. Furthermore, the reliance on palladium on carbon (Pd/C) for the cyclization step introduces substantial cost volatility due to the fluctuating prices of noble metals and creates stringent purification challenges to remove trace heavy metals from the final API. The violent heat release associated with the DMFDMA reaction step complicates process control, often leading to inconsistent yields and the formation of difficult-to-remove impurities that compromise the overall quality of the intermediate.
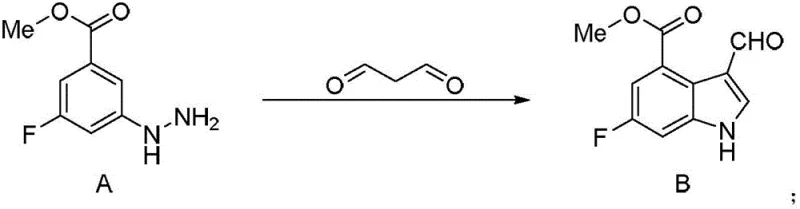
The Novel Approach
In stark contrast, the methodology outlined in patent CN107954919B employs a greener and more efficient strategy centered on Fischer indole synthesis. This innovative route utilizes malondialdehyde and a hydrazino-benzoate precursor in an aqueous buffered system, drastically lowering the reaction temperature to a mild 40°C. By eliminating the need for high-energy inputs and expensive transition metal catalysts, this approach not only enhances operational safety but also simplifies the downstream purification process. The subsequent conversion to the nitrovinyl derivative is achieved under similarly mild conditions using nitromethane and ammonium acetate, ensuring a smooth transformation with minimal byproduct formation. This rational design represents a significant leap forward in cost reduction in API manufacturing, providing a scalable solution that aligns with modern green chemistry principles.
Mechanistic Insights into Fischer Indole Cyclization
The core of this technological advancement lies in the meticulous optimization of the Fischer indole synthesis mechanism. In this process, the hydrazine group of the starting material undergoes condensation with the aldehyde groups of malondialdehyde to form a hydrazone intermediate. Under the catalytic influence of a mild acid buffer system, typically formic acid with sodium hydrogen phosphate, this hydrazone undergoes a [3,3]-sigmatropic rearrangement followed by aromatization to construct the indole ring. The use of a buffered aqueous medium is critical here, as it maintains the pH within a narrow optimal range that promotes cyclization while preventing the hydrolysis of the sensitive ester moiety. This precise control over the reaction environment ensures that the regioselectivity favors the formation of the desired 6-fluoro-3-formyl-1H-indole scaffold, minimizing the generation of regioisomers that could act as persistent impurities in later stages.
Furthermore, the second step involving the nitroaldol condensation is engineered to maximize stereochemical purity. The reaction between the aldehyde intermediate and nitromethane, catalyzed by ammonium acetate, proceeds through a well-defined transition state that favors the formation of the (E)-isomer of the nitrovinyl group. This stereoselectivity is paramount because the geometry of the double bond influences the subsequent cyclization steps required to form the final azepino-indole core of Rucaparib. By avoiding strong bases or harsh dehydrating agents, the process preserves the integrity of the fluorine substituent and the methyl ester, which are susceptible to nucleophilic attack under more aggressive conditions. This mechanistic elegance translates directly into higher crude purity, reducing the burden on crystallization and chromatography units during commercial production.
How to Synthesize 6-fluoro-3-[(E)-2-nitrovinyl]-1H-indole-4-carboxylic acid methyl ester Efficiently
To implement this superior synthetic route in a laboratory or pilot plant setting, operators must adhere to specific protocol parameters regarding stoichiometry and thermal management. The process begins with the careful preparation of the aqueous buffer system, ensuring that the molar ratios of the hydrazine precursor, malondialdehyde, and catalyst are maintained within the patented ranges to achieve optimal conversion. Detailed standard operating procedures for the isolation of the intermediate aldehyde and its subsequent conversion to the nitrovinyl product are critical for reproducibility. For a comprehensive guide on the exact experimental conditions, reagent grades, and workup procedures validated by the patent examples, please refer to the standardized synthesis protocol provided below.
- Perform Fischer indole synthesis by reacting 3-fluoro-5-hydrazino-benzoic acid methyl ester with malondialdehyde in an aqueous buffered system at 40°C.
- Isolate the intermediate 6-fluoro-3-formyl-1H-indole-4-carboxylic acid methyl ester through extraction and drying.
- Conduct nitroaldol condensation with nitromethane and ammonium acetate at 60°C to yield the final nitrovinyl intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of palladium catalysts removes a major cost driver and supply bottleneck, as the availability and price of precious metals can be highly unpredictable in the global market. Additionally, the shift from organic solvents to water as the primary reaction medium in the first step significantly reduces the environmental footprint of the manufacturing process, lowering the costs associated with solvent recovery and hazardous waste disposal. These factors combined create a more resilient supply chain capable of withstanding regulatory pressures and raw material fluctuations, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers without interruption.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts like Pd/C and the replacement of high-boiling organic solvents with water leads to substantial cost savings in raw material procurement. The mild reaction temperatures also result in drastically simplified energy requirements, as there is no need for extensive heating or cooling infrastructure to manage exothermic events. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, thereby improving the overall mass balance and reducing the cost of goods sold per kilogram of finished intermediate.
- Enhanced Supply Chain Reliability: By utilizing commercially available and stable raw materials such as malondialdehyde and simple hydrazines, the risk of supply disruption is significantly mitigated compared to relying on specialized reagents like DMFDMA. The robustness of the aqueous reaction system allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in ambient conditions or reagent quality. This stability ensures that production timelines can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing partners to plan their inventory levels with greater confidence and accuracy.
- Scalability and Environmental Compliance: The inherent safety of operating at 40°C in an aqueous environment makes this process exceptionally suitable for commercial scale-up of complex pharmaceutical intermediates. The absence of violent exotherms reduces the engineering controls required for large reactors, facilitating a smoother transition from pilot plant to multi-ton production scales. Moreover, the reduced generation of hazardous organic waste aligns with increasingly stringent global environmental regulations, positioning manufacturers as responsible corporate citizens and avoiding potential fines or shutdowns related to non-compliance with emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new method improve upon the conventional DMFDMA route?
A: The new method replaces the high-temperature (120°C) DMFDMA reaction and expensive Pd/C hydrogenation with a mild Fischer indole synthesis at 40°C, significantly reducing energy consumption and eliminating noble metal residues.
Q: What are the purity levels achievable with this process?
A: According to patent data, the process yields intermediates with HPLC purity exceeding 95%, reaching up to 98.3% in optimized examples, ensuring high quality for downstream API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of water as a primary solvent, mild reaction temperatures, and commercially available raw materials makes this route highly scalable and safer for industrial manufacturing compared to the exothermic prior art.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rucaparib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical application. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Rucaparib intermediate performs flawlessly in your downstream synthesis operations.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can drive efficiency and value for your organization.
