Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Multicomponent Carbonylation
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Multicomponent Carbonylation
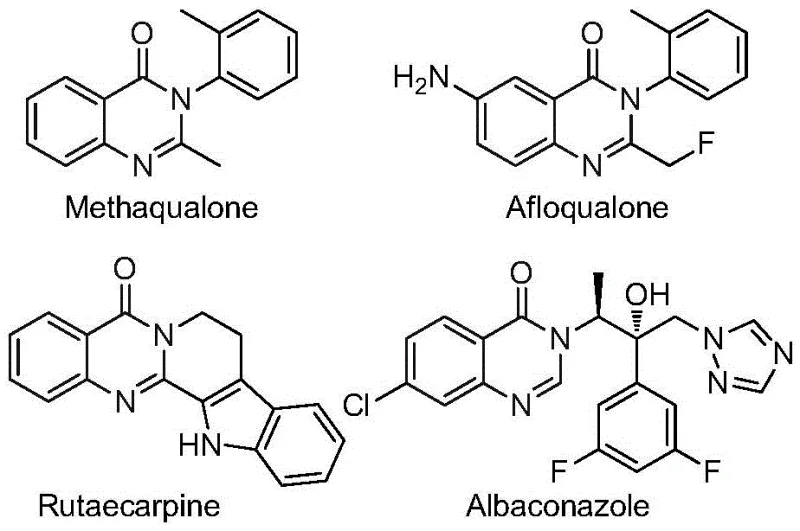
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting broad biological activity. Patent CN112480015B discloses a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a privileged structure found in numerous pharmaceutical agents. As illustrated in the structural diversity of bioactive molecules below, quinazolinone derivatives such as Methaqualone and Afloqualone possess significant therapeutic potential ranging from antifungal to anticancer properties. The introduction of a trifluoromethyl group further enhances these properties by improving metabolic stability and lipophilicity, making the efficient construction of this motif a high priority for process research and development teams.
This novel methodology addresses critical bottlenecks in the supply chain for high-purity pharmaceutical intermediates by utilizing readily available starting materials. The process leverages a transition metal palladium-catalyzed carbonylation cascade that merges trifluoroethylimidoyl chloride with nitro compounds. By avoiding the use of hazardous high-pressure carbon monoxide gas and replacing it with a solid surrogate, the technology offers a safer, more robust pathway for commercial scale-up of complex pharmaceutical intermediates. This report analyzes the technical merits and commercial implications of this invention for global procurement and R&D strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that present significant challenges for industrial application. Traditional synthetic routes often involve ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides, which notoriously require high-pressure carbon monoxide conditions. Such high-pressure operations necessitate specialized autoclave equipment and rigorous safety protocols, drastically increasing capital expenditure and operational complexity. Furthermore, alternative iron-catalyzed condensation reactions or palladium-catalyzed cyclizations using 2-bromoformylaniline often suffer from narrow substrate scope and low atom economy. These conventional methods frequently demand expensive, pre-activated substrates that are not commercially available off-the-shelf, leading to extended lead times for high-purity intermediates and complicating the sourcing strategy for procurement managers.
The Novel Approach
In stark contrast, the method described in Patent CN112480015B utilizes a streamlined multi-component coupling that bypasses these logistical hurdles. The core innovation lies in the direct use of cheap and abundant nitro compounds alongside trifluoroethylimidoyl chloride. As depicted in the general reaction scheme below, this transformation is facilitated by a palladium catalyst system in the presence of molybdenum hexacarbonyl, which serves as a safe, solid source of carbon monoxide. This eliminates the need for handling toxic CO gas cylinders, thereby reducing regulatory burdens and safety risks associated with the manufacturing process. The reaction proceeds efficiently in 1,4-dioxane at 120°C, demonstrating exceptional functional group tolerance that allows for the late-stage diversification of the quinazolinone scaffold without protecting group manipulations.
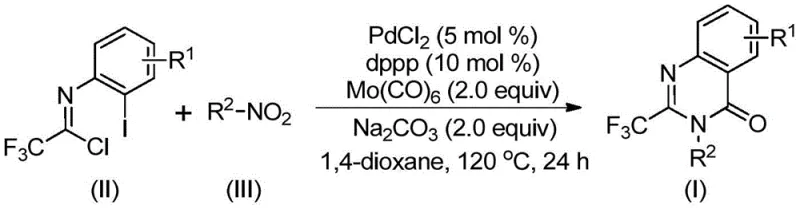
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the catalytic cycle is essential for R&D directors aiming to optimize impurity profiles and ensure batch-to-batch consistency. The reaction likely initiates with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which simultaneously releases carbon monoxide in situ. This generated amine then undergoes a base-promoted intermolecular carbon-nitrogen bond coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, generated from PdCl2 and the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride moiety, forming a reactive divalent palladium species. The released carbon monoxide then inserts into this carbon-palladium bond to generate an acyl-palladium intermediate, setting the stage for ring closure.
The final stages of the mechanism involve the promotion of palladium-nitrogen bond formation under basic conditions, leading to a seven-membered cyclic palladium intermediate. This strained intermediate subsequently undergoes reductive elimination to release the desired 2-trifluoromethyl substituted quinazolinone product and regenerate the active palladium catalyst. This intricate cascade ensures high reaction efficiency, with experimental data showing yields ranging from 69% to 96% across various substrates. The use of sodium carbonate as a mild base facilitates the deprotonation steps necessary for cyclization while minimizing side reactions such as hydrolysis of the sensitive imidoyl chloride. This mechanistic clarity allows process chemists to fine-tune reaction parameters to suppress specific byproducts, ensuring the delivery of high-purity OLED material or API intermediates that meet stringent quality specifications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this one-pot protocol makes it highly attractive for both laboratory discovery and pilot plant operations. The procedure involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and the two primary organic substrates in an aprotic solvent. The mixture is then heated to facilitate the cascade sequence, after which standard workup procedures involving filtration and silica gel chromatography yield the pure product. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrate classes, please refer to the step-by-step guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours under inert atmosphere.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for supply chain resilience and cost management. By shifting away from proprietary or hard-to-source precursors, manufacturers can leverage a commodity-based supply chain that is less susceptible to market volatility. The following points detail how this technology translates into tangible business value for stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing and supply chain reliability.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of nitro compounds, which are among the most inexpensive and widely available building blocks in the fine chemical industry. Unlike traditional routes that require pre-functionalized benzamides or acid anhydrides, this method starts from bulk chemicals, significantly lowering the raw material cost basis. Furthermore, the replacement of high-pressure carbon monoxide gas with solid Mo(CO)6 removes the need for expensive pressure-rated reactors and the associated safety infrastructure, leading to substantial capital expenditure savings. The high catalytic efficiency also means lower loading of precious palladium metals, contributing to a more sustainable and cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex intermediates. This method mitigates that risk by utilizing a modular approach where both the nitro component and the imidoyl chloride can be sourced from multiple global vendors or synthesized in-house with ease. The robustness of the reaction conditions, tolerating a wide range of substituents including halogens and alkyl groups, ensures that supply disruptions for one specific derivative can be managed by adjusting the synthetic design without changing the core process. This flexibility allows procurement managers to maintain steady inventory levels of reliable pharmaceutical intermediate supplier grades even during market fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen safety and waste disposal challenges. This one-pot carbonylation method is inherently scalable because it avoids the handling of toxic gases and minimizes the number of isolation steps, thereby reducing solvent consumption and waste generation. The use of a solid CO surrogate simplifies the engineering controls required for large-scale production, facilitating a smoother technology transfer from R&D to commercial manufacturing. Additionally, the high atom economy and reduced solvent usage align with green chemistry principles, helping companies meet increasingly strict environmental regulations and sustainability goals without compromising on output volume.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational parameters. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation.
Q: What are the advantages of using Mo(CO)6 over gaseous CO in this synthesis?
A: Using Mo(CO)6 as a solid carbon monoxide surrogate eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for scale-up.
Q: What is the substrate scope for the nitro compound in this reaction?
A: The method demonstrates excellent compatibility with various substituents including alkyl, halogen, and trifluoromethyl groups on the aromatic ring, allowing for diverse structural modifications.
Q: How does this method compare to traditional ruthenium-catalyzed cyclizations?
A: Unlike traditional methods requiring harsh conditions or expensive pre-activated substrates, this palladium-catalyzed approach utilizes cheap nitro compounds and operates under milder, one-pot conditions with higher efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory results translate into reliable industrial supply. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel quinazolinone derivatives or scale-up of existing routes, our infrastructure is designed to support your timeline and quality requirements.
We invite you to collaborate with us to explore how this innovative palladium-catalyzed carbonylation technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions for your next development phase.
