Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Intermediate Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and agrochemical industries continuously seek efficient pathways to access privileged heterocyclic scaffolds, among which quinazolinones stand out due to their profound biological activities ranging from antifungal to anticancer properties. A significant technological breakthrough in this domain is documented in Chinese Patent CN112480015B, which discloses a novel multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by leveraging a palladium-catalyzed carbonylation cascade that utilizes inexpensive nitro compounds as starting materials. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing processes for high-value intermediates. The introduction of the trifluoromethyl group is particularly strategic, as it enhances metabolic stability and lipophilicity, key parameters in modern drug design. By integrating this methodology into our production capabilities, we offer partners a reliable pharmaceutical intermediate supplier solution that balances high purity with operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been fraught with synthetic challenges that hinder large-scale adoption. Conventional routes often rely on the ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides, which notoriously require high-pressure carbon monoxide conditions. Such requirements impose severe safety risks and necessitate specialized, capital-intensive reactor infrastructure that many facilities lack. Furthermore, alternative methods involving iron-catalyzed condensation or palladium-catalyzed cyclization of 2-bromoformylaniline often suffer from narrow substrate scopes and the need for pre-activated, expensive starting materials. These legacy processes frequently result in low yields and generate substantial waste streams due to harsh reaction conditions and complex purification needs. The reliance on gaseous CO also introduces significant supply chain vulnerabilities and regulatory hurdles regarding hazardous gas handling. Consequently, the industry has long sought a safer, more atom-economical alternative that does not compromise on yield or structural diversity.
The Novel Approach
The methodology outlined in patent CN112480015B offers a transformative solution by employing a multi-component one-pot strategy that circumvents the need for external high-pressure gas. Instead of gaseous CO, the process utilizes molybdenum hexacarbonyl (Mo(CO)6) as a solid, safe, and controllable carbon monoxide substitute. This approach allows the reaction to proceed at a moderate temperature of 120°C in common organic solvents like 1,4-dioxane. The reaction initiates with the reduction of the nitro compound to an amine in situ, followed by coupling with trifluoroethylimidoyl chloride and subsequent cyclization. This telescoped sequence eliminates the isolation of unstable intermediates, thereby streamlining the workflow and reducing material loss. The use of cheap and readily available nitro compounds as the nitrogen source drastically lowers the raw material cost compared to pre-functionalized anilines or benzamides. This novel route not only simplifies the operational procedure but also expands the accessible chemical space for medicinal chemists.
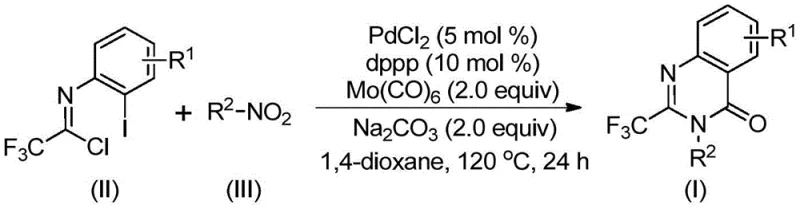
Mechanistically, this transformation is a masterpiece of organometallic chemistry, orchestrated by a palladium catalyst system comprising PdCl2 and the bidentate ligand 1,3-bis(diphenylphosphino)propane (dppp). The catalytic cycle likely begins with the reduction of the nitro group to the corresponding amine by Mo(CO)6, which simultaneously serves as the CO source. The generated amine then undergoes a base-promoted intermolecular carbon-nitrogen bond coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride moiety, forming a divalent palladium intermediate. As the reaction temperature rises, Mo(CO)6 releases carbon monoxide, which inserts into the carbon-palladium bond to generate an acylpalladium species. Under the influence of the base, an intramolecular nucleophilic attack facilitates the formation of a palladium-nitrogen bond, creating a seven-membered cyclic palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This intricate dance of coordination and insertion ensures high chemoselectivity, minimizing the formation of side products such as ureas or simple amides that often plague amidation reactions.
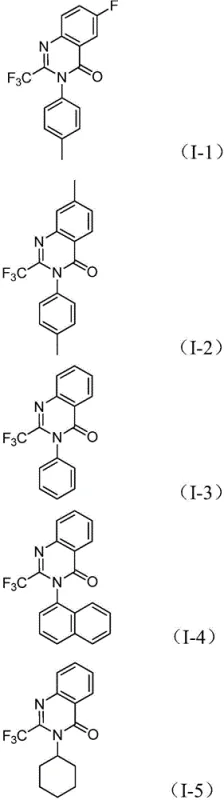
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. The one-pot nature of the reaction means that reactive intermediates are consumed as soon as they are formed, preventing their accumulation and subsequent degradation into hard-to-remove impurities. The specific choice of the dppp ligand stabilizes the palladium center, preventing the formation of palladium black and ensuring consistent catalytic activity throughout the 16 to 30-hour reaction window. Furthermore, the tolerance for various functional groups—such as halogens (fluorine, chlorine, bromine), alkyl chains, and electron-withdrawing trifluoromethyl groups—means that the process is robust against minor variations in feedstock quality. This robustness is crucial for maintaining a clean impurity profile, which simplifies downstream purification. The ability to synthesize diverse derivatives, as illustrated by the successful preparation of compounds I-1 through I-5, confirms that the electronic and steric properties of the substrates do not significantly derail the catalytic cycle, ensuring batch-to-batch consistency.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process is designed to be operationally simple, requiring standard Schlenk techniques or sealed tube reactors capable of withstanding 120°C. The key to success lies in the precise stoichiometric balance between the trifluoroethylimidoyl chloride, the nitro compound, and the catalytic system. Typically, a slight excess of the nitro compound (1.2 equivalents) is used to drive the reaction to completion, while the palladium catalyst is employed at a low loading of 5 mol% to maintain cost efficiency. The reaction mixture, dissolved in an aprotic solvent like 1,4-dioxane, must be heated uniformly to facilitate the slow release of CO from the molybdenum source. Detailed standardized synthetic steps for replicating this high-efficiency protocol are provided in the guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow for the carbonylation cascade and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from high-pressure gas protocols to a solid-state CO source fundamentally alters the risk profile and capital expenditure requirements of the manufacturing facility. By eliminating the need for specialized high-pressure autoclaves and the associated safety infrastructure for handling toxic carbon monoxide gas, companies can significantly reduce their operational overhead and insurance costs. This safety enhancement also accelerates the regulatory approval process for new manufacturing sites, as the hazards associated with the process are markedly lower. Furthermore, the reliance on commodity chemicals like nitro compounds and commercially available palladium salts ensures a stable and resilient supply chain, insulating production schedules from the volatility of specialized reagent markets.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive, pre-activated starting materials with cheap, bulk-available nitro compounds. In traditional routes, the synthesis of specific benzamides or anilines often requires multiple protection and deprotection steps, each adding cost and time. This one-pot method collapses these steps into a single operation, drastically reducing labor, solvent usage, and energy consumption. Additionally, the use of a solid CO surrogate removes the logistical costs and safety premiums associated with purchasing and storing high-pressure gas cylinders. The overall simplification of the workflow means that the cost per kilogram of the final API intermediate is substantially lowered, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of complex building blocks. This method mitigates that risk by utilizing nitro compounds, which are among the most abundant and widely produced organic chemicals globally. Whether sourcing 4-nitrotoluene or various halogenated nitrobenzenes, suppliers can easily find multiple vendors, preventing single-source bottlenecks. The robustness of the reaction conditions also means that production is less susceptible to delays caused by minor fluctuations in raw material specifications. This reliability allows for more accurate forecasting and inventory management, ensuring that downstream drug development programs are not stalled by intermediate shortages. The ability to scale this reaction from gram to multi-kilogram quantities without re-optimization further secures the supply line from clinical trials to commercial launch.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to minimize waste and hazardous emissions is a critical compliance metric. This synthesis generates fewer byproducts compared to multi-step alternatives, leading to a cleaner E-factor (mass of waste per mass of product). The absence of high-pressure gas vents reduces the risk of atmospheric contamination, and the use of standard organic solvents like dioxane allows for established recycling protocols. The scalability of the process is evidenced by its successful demonstration at the gram level with consistent yields, indicating a smooth path to ton-scale production. The simplified workup, involving basic filtration and chromatography, reduces the load on wastewater treatment facilities. This alignment with green chemistry principles not only satisfies regulatory bodies but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this method into their existing portfolios. The focus is on practical applicability, safety, and the breadth of chemical space accessible through this route.
Q: What are the primary advantages of this one-pot method over traditional quinazolinone synthesis?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap nitro compounds and operates under atmospheric pressure with solid CO sources, significantly improving safety and reducing equipment costs.
Q: Does this catalytic system tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility. It successfully accommodates various substituents including halogens (F, Cl, Br), alkyl groups, and trifluoromethyl groups on both the aromatic ring and the nitrogen substituent, allowing for broad library synthesis.
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly states the method can be expanded to the gram level and beyond. The use of commercially available reagents, simple workup procedures involving filtration and chromatography, and robust reaction conditions make it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the lab are faithfully reproduced on an industrial scale. We understand that for pharmaceutical intermediates, consistency is key; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards. Our facility is equipped to handle the specific thermal and catalytic requirements of this palladium-mediated process safely and efficiently, providing you with a secure source of high-quality materials for your drug development pipelines.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific route can optimize your bill of materials compared to your current supply chain. We encourage you to contact our technical procurement team to request specific COA data for related quinazolinone derivatives and to discuss route feasibility assessments tailored to your unique molecular targets. Let us help you accelerate your timeline to market with a supply partner committed to innovation, quality, and reliability.
