Revolutionizing Acetamide Manufacturing: Sustainable Rhodium-Catalyzed Process for Commercial Scale-Up in Pharmaceutical Intermediates
The patent CN112812032A introduces a groundbreaking rhodium-catalyzed aminocarbonylation methodology that fundamentally transforms acetamide compound synthesis through the innovative dual application of dimethyl carbonate as both C1 source and green solvent. This approach addresses critical limitations in traditional carbonylation processes by eliminating hazardous methylating agents while maintaining exceptional substrate flexibility across diverse functional groups. The methodology leverages commercially available catalysts including dirhodium tetracarbonyl dichloride and tungsten carbonyl under precisely controlled thermal conditions to achieve near-complete conversion. Notably, the process operates without stoichiometric base requirements, thereby preventing the generation of inorganic waste streams that complicate downstream processing in conventional manufacturing. This patent represents a significant advancement in sustainable pharmaceutical intermediate production, offering both environmental benefits through solvent recyclability and operational advantages via simplified reaction workup procedures. The technical elegance lies in its ability to simultaneously serve multiple process objectives while maintaining compatibility with complex molecular architectures required in modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional acetamide synthesis routes suffer from multiple critical deficiencies that impede both commercial viability and environmental sustainability. Conventional methylating agents such as diazomethane present severe explosion hazards while dimethyl sulfate and methyl iodide exhibit high toxicity and corrosiveness, necessitating specialized handling infrastructure that significantly increases capital expenditure. These methods typically require stoichiometric amounts of base to facilitate methylation reactions, generating substantial quantities of inorganic salts that demand costly disposal procedures and complicate waste stream management. Furthermore, transition metal-catalyzed carbonylation processes often employ highly corrosive cocatalysts like hydroiodic acid that cause rapid equipment degradation, leading to frequent maintenance shutdowns and inconsistent product quality. The narrow functional group tolerance of existing methodologies restricts their applicability to complex pharmaceutical intermediates containing sensitive substituents. Additionally, conventional processes frequently operate under extreme temperature or pressure conditions that increase energy consumption and safety risks while limiting scalability from laboratory to manufacturing environments. These combined limitations create significant barriers to efficient production of high-purity acetamide compounds required by modern pharmaceutical manufacturers.
The Novel Approach
The patented methodology overcomes these limitations through an elegantly designed rhodium-catalyzed system that utilizes dimethyl carbonate as a multifunctional reagent. By serving simultaneously as C1 source and green solvent, dimethyl carbonate eliminates the need for hazardous methylating agents while preventing inorganic salt generation during the reaction sequence. The carefully optimized catalyst system comprising dirhodium tetracarbonyl dichloride (2 mol%), DPPP ligand (6 mol%), and tungsten carbonyl (2 equiv.) operates effectively at moderate temperatures (120°C) without requiring corrosive cocatalysts. This approach demonstrates remarkable functional group tolerance across halogenated, trifluoromethylated, alkoxy-substituted, and heteroaryl nitro compounds while maintaining high conversion efficiency. The reaction proceeds in a single vessel with straightforward post-processing involving filtration and column chromatography, significantly reducing operational complexity compared to multi-step conventional methods. Crucially, the process generates recyclable methanol and carbon dioxide as byproducts rather than hazardous waste streams, aligning with green chemistry principles while lowering environmental compliance costs. This innovative approach enables reliable production of structurally diverse acetamide intermediates essential for pharmaceutical development pipelines.
Mechanistic Insights into Rhodium-Catalyzed Aminocarbonylation
The catalytic cycle begins with oxidative addition of the nitro compound to the rhodium center, followed by coordination of dimethyl carbonate as both solvent and carbon monoxide source. Tungsten carbonyl facilitates CO transfer through ligand exchange mechanisms while sodium phosphate acts as a mild base to promote deprotonation steps without generating significant salt waste. The iodide anion from sodium iodide enhances catalyst solubility and stabilizes key rhodium intermediates during the carbonylation sequence. This synergistic catalyst system enables direct conversion of nitro groups to acetamides through a series of well-defined organometallic steps including nitroso intermediate formation, CO insertion, and reductive elimination. The reaction pathway avoids traditional nitrene intermediates that often lead to side products, thereby improving selectivity toward the desired acetamide functionality. The phosphate buffer system maintains optimal pH conditions throughout the reaction while preventing catalyst decomposition that commonly occurs in conventional acidic or basic environments. This mechanistic sophistication explains the exceptional functional group tolerance observed across diverse substrates including those containing halogens, trifluoromethyl groups, and heterocyclic systems.
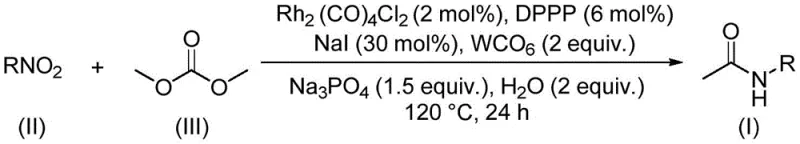
Impurity control is achieved through multiple complementary mechanisms inherent to this catalytic system. The moderate reaction temperature (120°C) prevents thermal decomposition pathways that typically generate byproducts in conventional high-temperature processes. The phosphate buffer system maintains precise pH control that minimizes hydrolysis side reactions while promoting selective carbonylation. The absence of strong acids or bases eliminates common impurities from proton transfer side reactions. The carefully optimized catalyst loading prevents over-reduction or dimerization pathways that could compromise product purity. Additionally, the use of dimethyl carbonate as solvent creates a homogeneous reaction environment that ensures consistent mass transfer and prevents localized hot spots that might lead to impurity formation. Post-reaction purification through standard column chromatography effectively removes any residual catalyst components or minor byproducts without requiring specialized techniques. This multi-faceted approach consistently delivers products meeting stringent pharmaceutical purity specifications without additional purification steps.
How to Synthesize Acetamide Compounds Efficiently
This patented methodology represents a significant advancement in sustainable pharmaceutical intermediate manufacturing through its innovative use of dimethyl carbonate as both carbon source and reaction medium. The process eliminates hazardous reagents while maintaining exceptional substrate flexibility across diverse functional groups required in modern drug development pipelines. By operating under mild thermal conditions with commercially available catalysts, this approach offers substantial operational advantages over conventional carbonylation methods that require extreme temperatures or pressures. The straightforward reaction setup and simplified workup procedure significantly reduce technical barriers to implementation while improving overall process safety profiles. Detailed standardized synthesis steps are provided below to facilitate seamless technology transfer from laboratory to manufacturing environments.
- Combine rhodium catalyst (Rh₂(CO)₄Cl₂), DPPP ligand, tungsten carbonyl, sodium phosphate, sodium iodide, water, nitro compound substrate, and dimethyl carbonate in a sealed reaction vessel at precise molar ratios.
- Heat the homogeneous mixture to 120°C under inert atmosphere with continuous stirring for exactly 24 hours to ensure complete conversion of nitro compounds.
- Perform post-reaction processing through filtration, silica gel adsorption, and column chromatography purification to isolate high-purity acetamide products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach directly addresses critical pain points in pharmaceutical supply chains by delivering enhanced operational reliability while reducing total cost of ownership for acetamide intermediates. The elimination of hazardous reagents simplifies regulatory compliance across multiple jurisdictions while improving workplace safety metrics that increasingly influence procurement decisions. By utilizing readily available starting materials with broad commercial supply channels, this methodology mitigates single-source dependency risks that commonly disrupt traditional intermediate supply chains. The process design inherently supports flexible production scheduling through its compatibility with standard manufacturing equipment without requiring specialized infrastructure investments.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal cocatalysts and hazardous methylating agents significantly reduces raw material costs while avoiding costly waste treatment procedures associated with inorganic salt byproducts. The dual functionality of dimethyl carbonate as both C1 source and solvent streamlines inventory management and reduces solvent procurement expenses through its recyclability potential. Simplified workup procedures minimize labor requirements during purification stages while eliminating specialized equipment needs for handling corrosive reagents.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials including nitro compounds and dimethyl carbonate ensures robust supply continuity even during market fluctuations. The process compatibility with standard manufacturing infrastructure prevents production bottlenecks caused by specialized equipment requirements while enabling rapid scale-up from laboratory validation to commercial production volumes. Consistent product quality across batches reduces rejection rates and associated supply chain disruptions common in conventional methods with narrow operating windows.
- Scalability and Environmental Compliance: The methodology demonstrates seamless scalability from laboratory to commercial production without requiring fundamental process modifications, maintaining consistent yield and purity profiles across all volume ranges. The environmentally benign nature of dimethyl carbonate eliminates hazardous waste streams that complicate regulatory compliance while supporting corporate sustainability initiatives through reduced carbon footprint per unit produced. Simplified waste management procedures lower environmental compliance costs while enhancing corporate social responsibility metrics valued by procurement teams.
Frequently Asked Questions (FAQ)
The following technical questions address common concerns regarding implementation of this patented methodology for commercial acetamide intermediate production. These answers are derived directly from experimental data presented in patent CN112812032A and reflect practical considerations for manufacturing scale-up.
Q: How does dimethyl carbonate function as both C1 source and green solvent while eliminating hazardous methylating agents?
A: Dimethyl carbonate serves as a non-toxic C1 donor that avoids explosive or corrosive reagents like diazomethane. Its dual role eliminates stoichiometric base requirements, preventing inorganic salt waste while enabling solvent recycling through methanol/CO₂ recovery.
Q: What functional group tolerances make this method suitable for diverse pharmaceutical intermediate production?
A: The rhodium-catalyzed system demonstrates exceptional compatibility with halogenated (F, Cl, Br), trifluoromethyl, alkoxy, and heteroaryl substituents across ortho/meta/para positions without requiring protective groups.
Q: How does eliminating transition metal cocatalysts improve supply chain reliability for commercial manufacturing?
A: By avoiding HI or other corrosive cocatalysts, the process prevents equipment degradation and reduces catalyst purification steps, ensuring consistent batch quality and uninterrupted production scaling from laboratory to plant scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Compound Supplier
Our patented rhodium-catalyzed methodology represents a paradigm shift in sustainable acetamide intermediate manufacturing that directly addresses the evolving needs of global pharmaceutical developers. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our rigorous QC labs equipped with advanced analytical capabilities. Our dedicated technical teams specialize in adapting this innovative process to meet specific client requirements while ensuring seamless integration into existing manufacturing workflows through comprehensive route feasibility assessments.
Leverage our expertise to optimize your acetamide intermediate supply chain through our Customized Cost-Saving Analysis service tailored to your specific production volumes and quality requirements. Contact our technical procurement team today to request specific COA data and detailed route feasibility assessments that demonstrate how this sustainable methodology can enhance your manufacturing efficiency while reducing total cost of ownership.
