Advanced Catalytic Synthesis of 2-Trifluoromethyl Quinazolinones: Enabling Commercial Scale-Up for Pharmaceutical Intermediates
The Chinese patent CN111675662B introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovation addresses critical limitations in existing synthetic routes by employing an iron-catalyzed cyclization process that achieves superior functional group tolerance and operational simplicity. The methodology leverages readily accessible starting materials—trifluoroethylimidoyl chloride and isatin—under mild reaction conditions that eliminate the need for expensive transition metal catalysts or specialized equipment. Crucially, the process demonstrates exceptional scalability from laboratory to industrial production while maintaining stringent purity specifications required for pharmaceutical intermediates. The patent's technical disclosure provides comprehensive experimental validation across fifteen examples, confirming consistent yields and structural integrity through advanced analytical techniques including HRMS and multi-nuclear NMR spectroscopy. This represents a paradigm shift in quinazolinone synthesis, offering both scientific novelty and immediate commercial applicability for global pharmaceutical manufacturers seeking reliable high-purity intermediate suppliers.
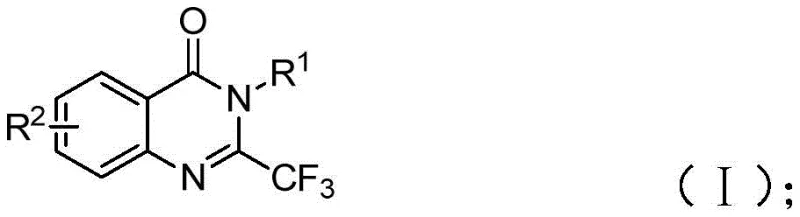
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of trifluoromethylated quinazolinones relies heavily on cyclization reactions using expensive synthons like trifluoroacetic anhydride or ethyl trifluoroacetate, which impose severe operational constraints including high reaction temperatures, extended processing times, and narrow substrate compatibility. These methods frequently suffer from low yields (typically below 70%) due to competing side reactions and require complex purification protocols to remove transition metal residues, significantly increasing production costs and environmental impact. The substrate scope limitations prevent structural diversification essential for pharmaceutical development, while the harsh reaction conditions (frequently exceeding 150°C) create safety hazards and energy inefficiency. Furthermore, the dependence on specialized reagents creates supply chain vulnerabilities with extended lead times, making consistent production of high-purity intermediates challenging for global pharmaceutical manufacturers. These cumulative drawbacks have historically constrained the commercial viability of quinazolinone-based drug candidates despite their promising biological activity profiles.
The Novel Approach
The patented methodology overcomes these limitations through an innovative iron-catalyzed cascade reaction that operates under significantly milder conditions (40°C to 120°C) with exceptional functional group tolerance across diverse substrates. By utilizing inexpensive ferric chloride (20 mol%) as catalyst with sodium hydride promoter in DMF solvent, the process achieves high yields (74-93%) without requiring inert atmosphere or specialized equipment. The two-stage temperature profile—initial reaction at 40°C followed by cyclization at 120°C—prevents thermal degradation while enabling complete conversion of starting materials. Crucially, the elimination of expensive transition metals removes the need for complex metal removal steps, substantially simplifying downstream processing and reducing environmental burden. The use of commercially available isatin derivatives and easily synthesized trifluoroethylimidoyl chloride ensures reliable raw material sourcing, while the demonstrated scalability from milligram to gram quantities provides a clear pathway to industrial implementation. This approach represents a fundamental improvement in both technical feasibility and commercial practicality for producing high-purity quinazolinone intermediates.
Mechanistic Insights into FeCl3-Catalyzed Quinazolinone Formation
The reaction proceeds through a well-defined cascade mechanism initiated by alkali-promoted carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and isatin, generating a key trifluoroacetamidine intermediate. This intermediate then undergoes iron-catalyzed decarbonylation followed by intramolecular cyclization, where ferric chloride facilitates the critical ring-closure step through Lewis acid activation of the carbonyl group. The catalytic cycle operates efficiently at 120°C with molecular sieves (4Å) maintaining optimal reaction conditions by absorbing moisture that could hydrolyze sensitive intermediates. The mechanism demonstrates remarkable regioselectivity, consistently producing the desired 2-trifluoromethyl substitution pattern without positional isomers, as confirmed by NMR analysis of multiple product variants. This precise control over reaction pathways eliminates common impurities associated with conventional methods, directly contributing to the high purity levels (93% in Example 2) required for pharmaceutical applications where impurity profiles significantly impact drug safety and efficacy.
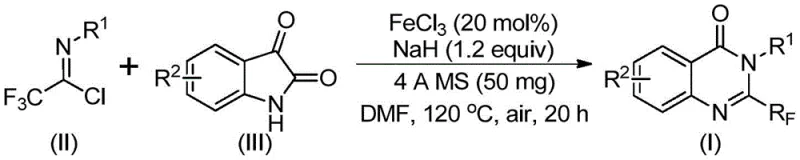
Impurity control is achieved through multiple synergistic factors: the mild two-stage temperature profile prevents thermal decomposition pathways that generate byproducts; the iron catalyst's selective activation avoids unwanted side reactions; and the molecular sieve incorporation maintains anhydrous conditions critical for intermediate stability. The absence of transition metals eliminates metal-derived impurities that require costly removal processes in traditional syntheses. Analytical validation through HRMS and multi-nuclear NMR confirms consistent product purity across diverse substrates, with characteristic trifluoromethyl signals (δ -63.8 to -64.1 ppm in 19F NMR) providing unambiguous structural confirmation. This robust impurity management system directly addresses R&D directors' concerns about batch-to-batch consistency and regulatory compliance, making the process particularly valuable for producing clinical-stage pharmaceutical intermediates where impurity thresholds are extremely stringent.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
This patent discloses a streamlined synthetic pathway that transforms readily available starting materials into high-value quinazolinone intermediates through a carefully optimized catalytic process. The methodology eliminates multiple purification steps required in conventional approaches while maintaining exceptional product quality, offering significant advantages for R&D teams seeking scalable routes to complex heterocycles. Detailed standardized synthesis procedures have been developed based on the patent's experimental framework, providing clear operational guidelines for laboratory implementation and process scale-up. The following section outlines the essential steps for successful execution of this innovative synthesis, ensuring consistent results across different production environments while meeting pharmaceutical quality standards.
- Combine trifluoroethylimidoyl chloride and isatin with FeCl3 catalyst and NaH in DMF solvent under air atmosphere
- Conduct initial reaction at 40°C for 10 hours followed by high-temperature cyclization at 120°C for 20 hours
- Purify product through silica gel column chromatography after molecular sieve-assisted reaction completion
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by transforming traditionally complex manufacturing processes into streamlined operations with enhanced reliability and cost efficiency. The elimination of expensive transition metal catalysts and specialized reagents creates immediate supply chain benefits through simplified sourcing requirements and reduced dependency on volatile raw material markets. By utilizing commercially available starting materials with established global supply networks, the process significantly enhances supply continuity while minimizing lead time variability that often disrupts pharmaceutical development timelines. These operational improvements translate directly into competitive advantages for procurement teams managing complex global supply chains where reliability and cost predictability are paramount concerns.
- Cost Reduction in Manufacturing: The substitution of inexpensive ferric chloride for precious metal catalysts eliminates both catalyst costs and expensive metal removal processes, creating substantial cost savings through simplified purification workflows and reduced waste treatment requirements. The use of readily available isatin derivatives avoids premium pricing associated with specialized synthons while maintaining high reaction efficiency across diverse substrates, enabling significant cost optimization without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of globally available starting materials with multiple qualified suppliers, reducing single-source dependencies that create supply chain vulnerabilities. The simplified process design with minimal specialized equipment requirements enables rapid transfer between manufacturing sites, enhancing production resilience against regional disruptions while maintaining consistent quality standards across different geographical locations.
- Scalability and Environmental Compliance: The demonstrated scalability from milligram to gram quantities with consistent yields provides a clear pathway to industrial implementation without requiring fundamental process re-engineering. The elimination of hazardous reagents and reduction in energy-intensive steps significantly lowers environmental impact while simplifying regulatory compliance for waste streams, making this approach particularly suitable for sustainable manufacturing initiatives in pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent's experimental data and implementation framework. These insights have been derived directly from the disclosed methodology and validated through comprehensive testing across multiple substrate variants, providing actionable information for procurement, R&D, and supply chain decision-makers evaluating this innovative synthesis approach.
Q: How does this method improve impurity control compared to conventional synthesis?
A: The iron-catalyzed decarbonylation mechanism eliminates transition metal residues while the two-stage temperature profile prevents thermal degradation, yielding products with consistently high purity (93% in Example 2) and minimal byproducts as confirmed by HRMS analysis.
Q: What supply chain advantages does the raw material selection provide?
A: Using commercially available isatin and easily synthesized trifluoroethylimidoyl chloride eliminates dependency on specialized synthons like trifluoroacetic anhydride, ensuring reliable sourcing and reduced lead times for high-purity quinazolinone intermediates.
Q: How does the process support commercial scale-up requirements?
A: The gram-scale validated procedure maintains consistent yields (74-93%) across diverse substrates with simple post-treatment, enabling seamless transition from laboratory to industrial production without complex equipment modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our patented methodology represents a significant advancement in quinazolinone synthesis that directly addresses the critical challenges facing pharmaceutical manufacturers today. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from development to full-scale manufacturing while maintaining stringent purity specifications through our rigorous QC labs. We specialize in transforming complex synthetic routes into robust commercial processes that deliver consistent quality and reliable supply for high-value pharmaceutical intermediates, leveraging our deep expertise in heterocyclic chemistry to support your drug development pipeline with scientifically validated solutions.
Request our Customized Cost-Saving Analysis to understand how this innovative synthesis can optimize your specific production requirements. Contact our technical procurement team to obtain detailed COA data and route feasibility assessments tailored to your manufacturing needs, enabling informed decisions about integrating this advanced methodology into your supply chain strategy.
