Advanced FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Introduction to Next-Generation Quinazolinone Synthesis
The pharmaceutical industry continuously seeks efficient pathways to access nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Quinazolinones, in particular, represent a privileged scaffold found in numerous drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these scaffolds further enhances their metabolic stability and lipophilicity, making them highly desirable targets for drug discovery programs. However, traditional synthetic routes often suffer from limitations such as harsh reaction conditions, expensive reagents, and narrow substrate scope. Addressing these critical bottlenecks, the technology disclosed in patent CN111675662B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This novel approach leverages an inexpensive iron-catalyzed cyclization strategy that transforms readily available starting materials into high-value intermediates with exceptional efficiency.
For R&D directors and process chemists, this patent represents a significant leap forward in synthetic methodology. By utilizing trifluoroethylimidoyl chloride and isatin derivatives as key building blocks, the process circumvents the need for specialized trifluoromethylating agents that are often costly and difficult to handle. The reaction operates under relatively mild conditions compared to classical methods, utilizing a catalytic amount of ferric chloride to drive the transformation. This shift not only simplifies the operational workflow but also aligns with the growing industry demand for sustainable and cost-effective manufacturing processes. The ability to generate diverse libraries of quinazolinone derivatives rapidly makes this technology an invaluable asset for accelerating lead optimization campaigns in medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with trifluoromethyl-containing reagents. Common trifluoromethyl sources include trifluoroacetic anhydride and ethyl trifluoroacetate. While these methods have served the community for decades, they are increasingly viewed as suboptimal for modern large-scale manufacturing. The primary drawbacks include severe reaction conditions that often require strong acids or bases, elevated temperatures, and prolonged reaction times which can degrade sensitive functional groups. Furthermore, the atom economy of these traditional routes is frequently poor, generating significant amounts of waste that complicate downstream purification and environmental compliance.
Another critical limitation of conventional methodologies is the narrow substrate scope. Many existing protocols fail to tolerate electron-withdrawing or electron-donating groups on the aromatic rings, restricting the chemical space available to medicinal chemists. This lack of versatility forces researchers to develop custom synthetic routes for each new analog, drastically increasing the time and cost associated with drug development. Additionally, the reliance on expensive catalysts or stoichiometric amounts of hazardous reagents poses safety risks and supply chain vulnerabilities. For procurement managers, these factors translate into higher raw material costs and unpredictable lead times, making the sourcing of high-purity quinazolinone intermediates a persistent challenge.
The Novel Approach
The innovative method described in the patent data fundamentally reimagines the construction of the quinazolinone core. By employing trifluoroethylimidoyl chloride and isatin as the primary substrates, the reaction achieves a direct and efficient assembly of the target molecule. The use of ferric chloride as a catalyst is a game-changer; iron is abundant, non-toxic, and significantly cheaper than precious metals like palladium or rhodium often used in cross-coupling reactions. This switch to an earth-abundant metal catalyst drastically reduces the cost of goods sold (COGS) while maintaining high catalytic activity. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization.
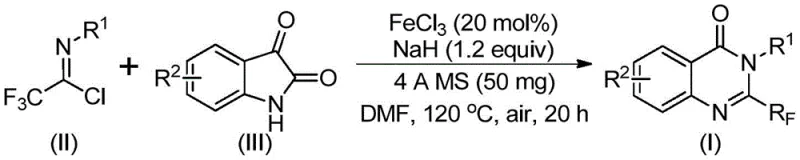
This novel pathway offers superior functional group tolerance, allowing for the incorporation of diverse substituents such as halogens, alkyl groups, and nitro groups without the need for protective groups. The operational simplicity is another major advantage; the reaction can be conducted in common polar aprotic solvents like DMF and does not require strictly inert atmospheres, as it can proceed effectively under air. For supply chain heads, this robustness means fewer specialized equipment requirements and a lower risk of batch failure due to minor deviations in process parameters. The method has been demonstrated to work efficiently on a gram scale, providing a clear trajectory for industrial scale-up to meet commercial demand for these valuable pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and impurity control. The reaction initiates with the interaction between the trifluoroethylimidoyl chloride and the isatin derivative in the presence of sodium hydride. The base facilitates the deprotonation of the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the imidoyl chloride. This step forms a key trifluoroacetamidine intermediate, establishing the essential carbon-nitrogen bonds required for the fused ring system. The presence of 4A molecular sieves in the reaction mixture plays a vital role in scavenging moisture, which could otherwise hydrolyze the sensitive imidoyl chloride or deactivate the catalyst, thereby ensuring high conversion rates.
Following the initial coupling, the ferric chloride catalyst engages in a decarbonylation and cyclization cascade. The iron center likely coordinates with the carbonyl oxygen of the isatin moiety, activating it towards nucleophilic attack or facilitating the extrusion of carbon monoxide. This decarbonylation step is critical as it drives the equilibrium towards the formation of the thermodynamically stable quinazolinone ring. The cyclization closes the six-membered heterocyclic ring, yielding the final 2-trifluoromethyl substituted product. This mechanism explains the high regioselectivity observed in the patent examples, where the trifluoromethyl group is exclusively installed at the 2-position. By controlling the stoichiometry of the base and the loading of the iron catalyst, manufacturers can minimize side reactions such as polymerization or over-alkylation, ensuring a clean impurity profile that simplifies downstream purification.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol outlined in the patent provides a robust framework that balances reaction kinetics with operational safety. The use of sodium hydride necessitates careful handling to prevent exothermic events, while the choice of solvent (preferably DMF) ensures adequate solubility of all components throughout the reaction course. The two-stage temperature profile, starting at a lower temperature to form the intermediate and ramping up to complete the cyclization, is a key feature that prevents thermal runaway while ensuring complete conversion.
- Prepare the reaction mixture by adding ferric chloride (20 mol%), sodium hydride (1.2 equiv), and 4A molecular sieves to a reaction vessel containing the trifluoroethylimidoyl chloride and isatin derivative in DMF solvent.
- Initiate the reaction by stirring the mixture at 40°C for approximately 10 hours to allow for initial bond formation, then increase the temperature to 120°C.
- Maintain the reaction at 120°C for an additional 20 hours under air atmosphere to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iron-catalyzed technology offers tangible strategic benefits beyond mere chemical novelty. The shift away from precious metal catalysts and exotic reagents directly impacts the bottom line by reducing raw material volatility. Iron salts are commodity chemicals with stable pricing and global availability, insulating the supply chain from the geopolitical and market fluctuations that often affect palladium or rhodium supplies. Furthermore, the simplicity of the workup procedure, which involves standard filtration and chromatography, reduces the consumption of silica gel and solvents during purification, contributing to overall process efficiency.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of cheap, commercially available starting materials like isatin and aromatic amines lead to substantial cost savings. The high atom economy of the reaction minimizes waste generation, reducing disposal costs and environmental fees. Additionally, the ability to run the reaction under air eliminates the need for costly inert gas purging systems and specialized pressure vessels, lowering capital expenditure for manufacturing facilities.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity. The wide substrate scope means that a single platform technology can be used to produce a variety of analogs, reducing the need for multiple distinct supply chains for different intermediates. This flexibility allows manufacturers to respond quickly to changing demand patterns in the pharmaceutical sector without retooling production lines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having been validated from milligram to gram scales with high yields. The use of DMF, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling protocols. The absence of heavy metal residues in the final product simplifies regulatory filings and ensures compliance with strict limits on elemental impurities in drug substances, facilitating faster time-to-market for new therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this method into their production workflows.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic and operational advantages compared to precious metal catalysts or harsh acidic conditions. It is inexpensive, readily available, and demonstrates high functional group tolerance, allowing for the synthesis of diverse derivatives without requiring complex protection-deprotection strategies.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the patent data indicates that the method is robust and scalable. The use of common solvents like DMF, combined with the stability of the iron catalyst system and the ability to run the reaction under air rather than strict inert atmospheres, simplifies the engineering requirements for scaling up from gram to multi-kilogram levels.
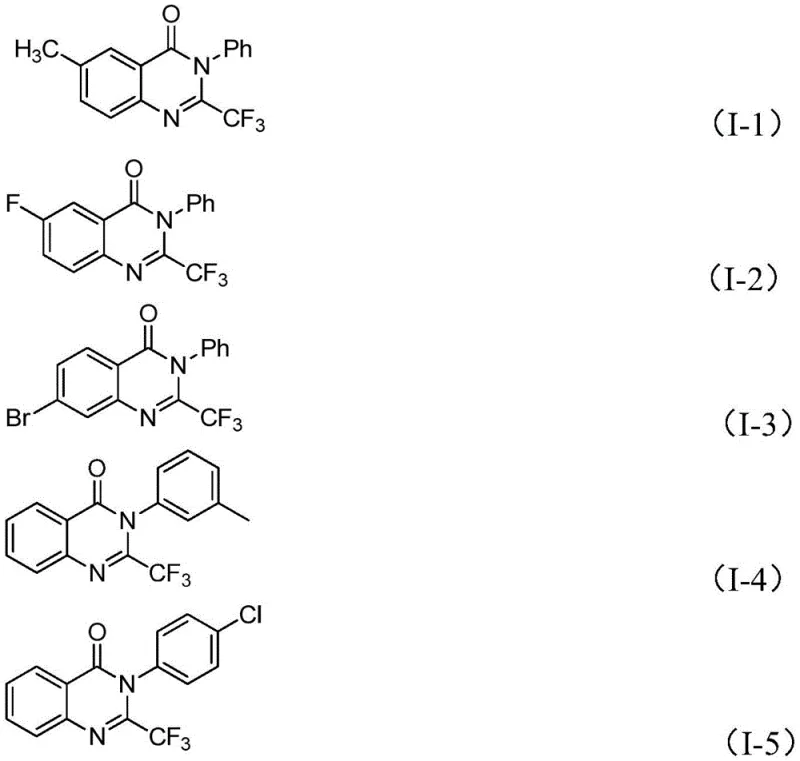
Q: What is the substrate scope for the R1 and R2 positions in this reaction?
A: The reaction exhibits excellent substrate scope. The R1 position on the imidoyl chloride can accommodate various substituted aryl groups including methyl, fluoro, chloro, bromo, and nitro substituents. Similarly, the isatin derivative (R2 position) tolerates alkyl, halogen, and methoxy groups at different positions on the benzene ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development pipelines. Our team of expert chemists has thoroughly analyzed the FeCl3-catalyzed route described in patent CN111675662B and is fully prepared to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and scalable technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your research and reduce your manufacturing costs with our reliable supply of advanced pharmaceutical intermediates.
