Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
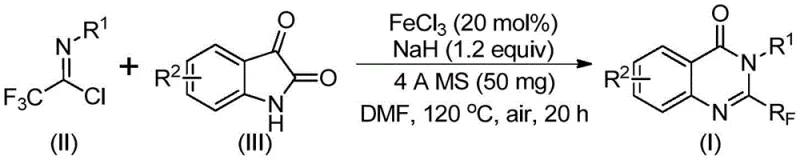
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access nitrogen-containing heterocycles, particularly quinazolinones, which serve as critical scaffolds in numerous bioactive molecules. Patent CN111675662B, published in late 2021, introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds that addresses long-standing challenges in synthetic efficiency and cost. This innovation leverages a readily available iron catalyst system to facilitate the cyclization of trifluoroethylimidoyl chloride with isatin derivatives, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production. The introduction of the trifluoromethyl group is strategically significant, as it enhances the electronegativity, metabolic stability, and lipophilicity of the target drug candidates, thereby improving their overall bioavailability and therapeutic potential. By shifting away from expensive precious metal catalysts and harsh reaction conditions typically associated with trifluoromethylation, this technology represents a pivotal advancement for manufacturers aiming to optimize their supply chains for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives bearing trifluoromethyl functionalities has been fraught with significant operational and economic hurdles that hinder efficient commercial manufacturing. Traditional routes often rely on the cyclization of synthons containing trifluoromethyl groups, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide or isatoic anhydride. These conventional methods are frequently plagued by severe reaction conditions that require stringent control over temperature and pressure, leading to increased energy consumption and safety risks in a plant setting. Furthermore, the starting materials employed in these legacy processes are often prohibitively expensive or difficult to source in bulk quantities, creating bottlenecks in the supply chain that can delay project timelines. The narrow substrate scope of many existing protocols also limits the ability of medicinal chemists to explore diverse chemical space, while low yields necessitate complex purification steps that generate substantial chemical waste, contradicting modern green chemistry principles.
The Novel Approach
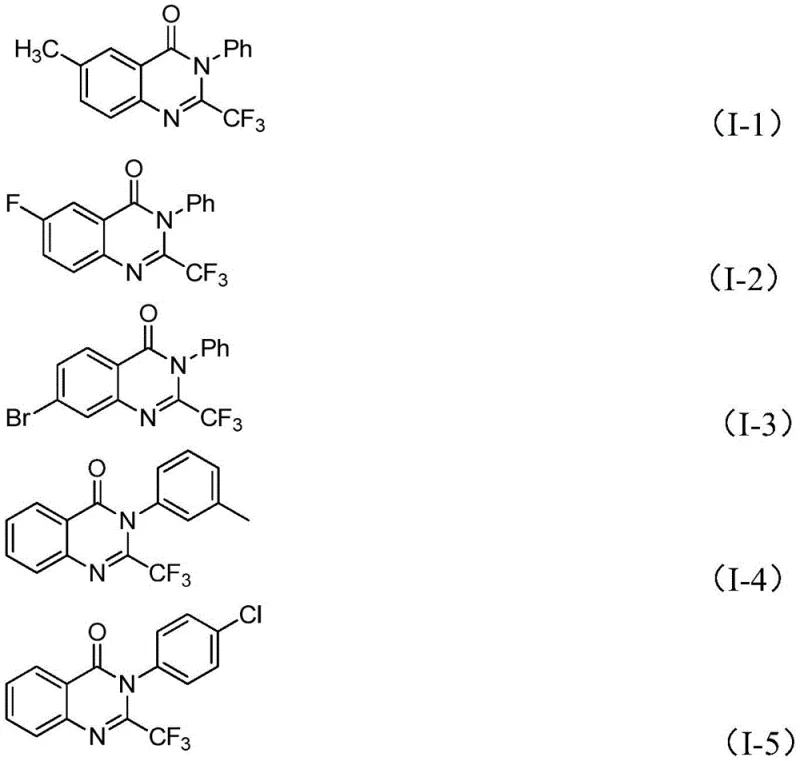
In stark contrast to these legacy issues, the novel approach detailed in the patent utilizes a highly efficient iron-catalyzed system that fundamentally reshapes the economics of producing these valuable heterocycles. By employing trifluoroethylimidoyl chloride and isatin as the primary building blocks, the method accesses the quinazolinone core through a streamlined sequence that avoids the need for exotic reagents. The use of ferric chloride as the catalyst is a game-changer, as iron is abundant, non-toxic, and significantly cheaper than palladium or rhodium alternatives often found in cross-coupling reactions. This new strategy not only simplifies the operational procedure but also expands the range of compatible functional groups, allowing for the synthesis of diverse derivatives with methyl, halogen, or methoxy substitutions without compromising yield. The reaction proceeds smoothly in common polar aprotic solvents like DMF, and the workup involves standard filtration and chromatography, making the transition from laboratory discovery to industrial production seamless and cost-effective.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway underpinning this transformation is a sophisticated interplay of base-promoted bond formation and transition metal catalysis that ensures high selectivity and conversion. Initially, the presence of sodium hydride facilitates the deprotonation of the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a crucial carbon-nitrogen bond, resulting in a trifluoroacetamidine intermediate that serves as the precursor for the subsequent cyclization event. Following this initial coupling, the ferric chloride catalyst plays a pivotal role in promoting a decarbonylation process, effectively removing the carbonyl oxygen from the isatin moiety to drive the ring closure. This iron-mediated step is critical for aromatizing the system and establishing the stable quinazolinone framework, distinguishing it from other pathways that might stall at the amidine stage. The synergy between the strong base and the Lewis acidic iron center creates a reactive environment that overcomes the kinetic barriers typically associated with forming fused heterocyclic rings under mild conditions.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that often plague multi-step syntheses. The specificity of the iron catalyst for the decarbonylation step reduces the formation of polymeric byproducts or incomplete cyclization species that are common in thermal-only processes. Additionally, the use of 4A molecular sieves in the reaction mixture helps to sequester moisture, which is essential for maintaining the activity of the sodium hydride and preventing the hydrolysis of the sensitive imidoyl chloride starting material. This careful management of reaction conditions ensures that the crude product profile is clean, significantly reducing the burden on downstream purification units. For R&D directors, understanding this mechanism highlights the robustness of the process, as the tolerance for various substituents on the aromatic rings suggests that electronic effects do not drastically inhibit the catalytic cycle, allowing for a wide array of analogues to be produced with consistent quality.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a practical setting requires precise attention to the stoichiometry and thermal profile outlined in the patent to maximize yield and purity. The process begins with the careful addition of ferric chloride and sodium hydride to a reaction vessel containing the organic solvent, followed by the introduction of the molecular sieves and substrates. It is critical to maintain the initial reaction temperature at 40°C for a defined period to allow the initial coupling to proceed without triggering premature decomposition of the reagents. Once this induction period is complete, the temperature is ramped to 120°C to drive the energetically demanding cyclization and decarbonylation steps to completion. Detailed standardized synthetic steps see the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C and continue reacting for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost stability and operational simplicity. The shift from precious metal catalysts to ferric chloride eliminates the volatility associated with the pricing of rare earth elements, providing a predictable cost structure for long-term manufacturing contracts. Furthermore, the reliance on isatin and aromatic amines as starting materials leverages a supply chain that is well-established and mature, ensuring that raw material availability will not become a bottleneck during scale-up phases. The simplified post-treatment process, which avoids complex extraction or distillation steps in favor of straightforward filtration and chromatography, translates directly into reduced labor hours and lower utility costs per kilogram of product. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market without compromising on margin or delivery reliability.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts with commodity-grade iron salts results in a drastic reduction in direct material costs, while the high atom economy of the reaction minimizes waste disposal fees. By avoiding the use of specialized ligands or inert atmosphere conditions required by other methods, the process further lowers the capital expenditure needed for reactor setup and maintenance. This economic efficiency allows manufacturers to offer competitive pricing for high-purity intermediates, enhancing the overall profitability of the drug development pipeline.
- Enhanced Supply Chain Reliability: Sourcing trifluoroethylimidoyl chloride and isatin derivatives is straightforward due to their widespread availability from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor fluctuations in environmental parameters or reagent quality. This reliability ensures consistent lead times for customers, enabling them to plan their own clinical trial material production or commercial launches with greater confidence and precision.
- Scalability and Environmental Compliance: The use of DMF as a solvent, while requiring proper handling, is a standard practice in the industry with well-defined recovery and recycling protocols that support environmental compliance goals. The absence of heavy metal residues in the final product simplifies the regulatory filing process, as there is no need for extensive testing to prove the removal of toxic catalyst traces. This scalability from gram to multi-kilogram batches without loss of efficiency makes the technology ideally suited for both early-stage development and full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of using FeCl3 for quinazolinone synthesis?
A: The use of ferric chloride offers a significant cost advantage over precious metal catalysts while maintaining high reaction efficiency and broad substrate tolerance, making it ideal for industrial scale-up.
Q: Can this method tolerate various functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substituents such as halogens, alkyl groups, and methoxy groups on both the isatin and imidoyl chloride components.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Absolutely. The method utilizes cheap, commercially available raw materials and simple post-treatment procedures like filtration and column chromatography, facilitating easy expansion from gram to kilogram scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, partnering with an experienced CDMO is essential for navigating the complexities of process chemistry. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our facility is equipped with stringent purity specifications and rigorous QC labs that guarantee every batch of 2-trifluoromethyl quinazolinone meets the highest international standards for pharmaceutical intermediates. We understand the critical nature of timeline and quality in drug development, and our team is dedicated to providing the technical support necessary to optimize this iron-catalyzed route for your specific needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this catalytic system for your specific target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will accelerate your path to market while maximizing value.
