Advanced N-Acyl Indole Synthesis: Bridging Innovative Catalysis with Commercial-Scale Pharmaceutical Manufacturing
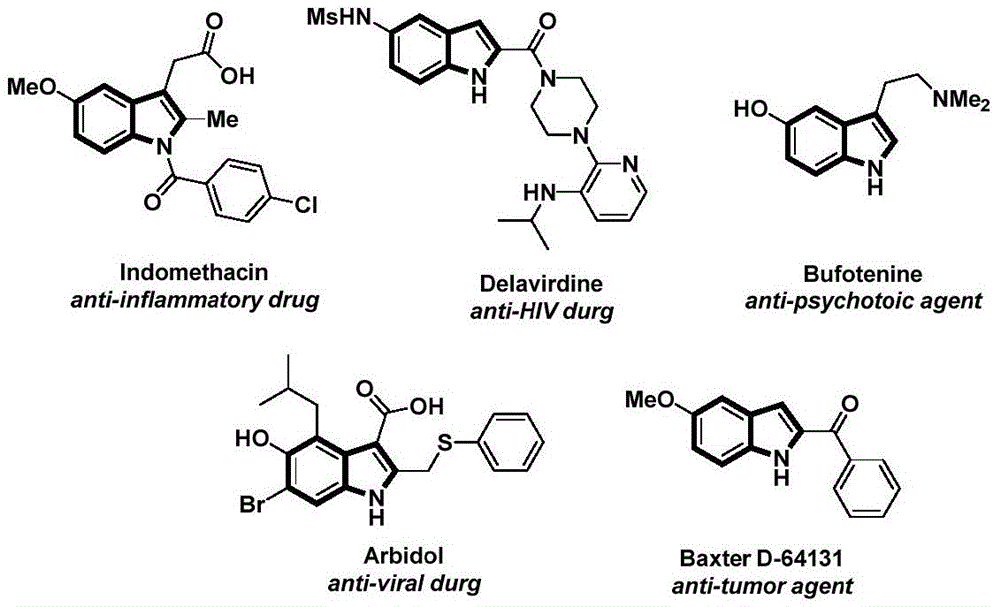
The innovative methodology detailed in Chinese patent CN112898192B presents a significant advancement in the synthesis of N-acyl indole compounds, which serve as critical building blocks in pharmaceutical development. This palladium-catalyzed carbonylation approach offers a streamlined pathway to these structurally complex molecules that are essential to numerous therapeutic agents, including anti-inflammatory, anti-HIV, and anti-tumor drugs as demonstrated by the molecular structures shown in Figure 1. The process eliminates traditional multi-step sequences while maintaining high substrate compatibility across diverse functional groups, addressing key challenges faced by pharmaceutical manufacturers seeking reliable API intermediate suppliers.
Advanced Catalytic Mechanism and Purity Control
The reaction mechanism begins with palladium insertion into the aryl iodide to form an aryl palladium intermediate, followed by carbon monoxide released from 1,3,5-tricarboxylate phenolate inserting into this intermediate to generate an acyl palladium species. Subsequently, 2-alkynylaniline adds to the acyl palladium intermediate, with reductive elimination yielding an amide compound that undergoes cyclization under silver oxide catalysis to form the final N-acyl indole product. This carefully orchestrated sequence avoids harsh reaction conditions typically required in conventional methods, operating at a moderate 60°C temperature that preserves sensitive functional groups while ensuring high conversion rates across various substrates as documented in the patent examples.
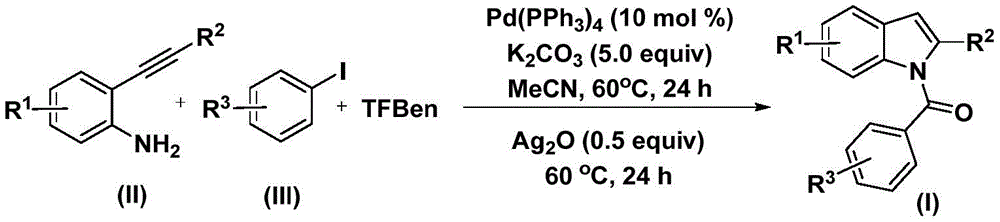
Impurity profile management is significantly enhanced through this methodology's inherent selectivity and mild reaction conditions, which minimize unwanted side reactions that commonly occur in traditional high-temperature carbonylation processes. The use of acetonitrile as solvent provides optimal solubility for all reaction components while facilitating straightforward purification through standard column chromatography techniques described in the patent. The documented NMR characterization data for multiple product variants confirms consistent high purity levels across diverse substitution patterns, with no detectable transition metal residues due to the efficient post-reaction filtration step that removes palladium catalysts before final purification. This inherent purity advantage directly addresses regulatory requirements for pharmaceutical intermediates while reducing the need for additional purification steps that typically increase manufacturing costs and timelines.
Traditional vs. Novel Synthesis Pathways for N-Acyl Indole Compounds
The Limitations of Conventional Methods
Traditional approaches to N-acyl indole synthesis often require multiple protection/deprotection steps and harsh reaction conditions that limit substrate scope and generate complex impurity profiles requiring extensive purification. Many existing methodologies employ high-pressure carbon monoxide gas systems that present significant safety hazards and require specialized equipment not readily available in standard pharmaceutical manufacturing facilities. The narrow functional group tolerance of conventional methods frequently necessitates custom synthetic routes for each derivative, substantially increasing development timelines and costs while creating supply chain vulnerabilities for pharmaceutical companies dependent on these critical intermediates. Furthermore, traditional processes often suffer from inconsistent yields across different substrate combinations, making scale-up unpredictable and complicating quality control procedures essential for pharmaceutical manufacturing.
The Novel Approach
The patented methodology overcomes these limitations through its innovative use of 1,3,5-tricarboxylate phenolate as a safe carbon monoxide substitute, eliminating the need for hazardous pressurized CO gas systems while maintaining high reaction efficiency. The single-pot process operates under mild conditions (60°C) with excellent functional group tolerance across diverse substitution patterns on both the alkyne and aryl iodide components, as demonstrated by the successful synthesis of fifteen different derivatives documented in the patent examples. This broad substrate compatibility enables pharmaceutical manufacturers to access multiple structural variants from a single standardized process platform, significantly reducing development timelines while ensuring consistent quality across different product variants. The straightforward workup procedure involving simple filtration followed by column chromatography provides a scalable purification pathway that maintains high purity without requiring specialized equipment or additional processing steps.
Commercial Advantages and Supply Chain Optimization
This innovative synthesis pathway delivers substantial commercial benefits that directly address critical pain points for pharmaceutical manufacturers seeking reliable API intermediate suppliers with cost-effective production capabilities. The process eliminates expensive high-pressure reaction equipment while utilizing readily available starting materials and catalysts, creating significant opportunities for cost reduction in pharmaceutical manufacturing without compromising product quality or purity standards required for clinical applications.
- Cost Reduction: The elimination of specialized high-pressure CO equipment reduces capital expenditure requirements while the use of commercially available starting materials at optimal stoichiometric ratios minimizes raw material costs. The single-pot methodology reduces solvent consumption and processing time compared to traditional multi-step sequences, with the documented reaction efficiency enabling higher throughput per manufacturing batch. This streamlined approach significantly lowers the cost per kilogram of final product while maintaining the high purity standards required for pharmaceutical intermediates, providing substantial economic advantages over conventional synthesis routes that require additional purification steps to achieve comparable quality levels.
- Reduced Lead Time: The simplified process flow with only two sequential reaction steps followed by straightforward purification reduces overall manufacturing cycle time compared to traditional multi-step approaches. The documented compatibility with diverse substrates allows for rapid production of multiple structural variants without requiring process revalidation, enabling faster response to changing development needs. The elimination of specialized equipment requirements means production can be initiated more quickly without lengthy installation and qualification periods, while the robust nature of the process ensures consistent first-time-right manufacturing that minimizes batch failures and associated delays in the supply chain.
- Scalability and Continuous Supply: The moderate reaction conditions (60°C) and standard equipment requirements make this process inherently scalable from laboratory to commercial production volumes without requiring significant process re-engineering. The documented success across fifteen different substrate combinations demonstrates the process robustness needed for reliable commercial supply of complex intermediates under varying production demands. The use of readily available catalysts and reagents ensures supply chain resilience against material shortages, while the straightforward purification protocol maintains consistent product quality across different production scales, supporting continuous supply requirements for both clinical and commercial pharmaceutical manufacturing operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112898192B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
