Scalable Visible Light Photocatalysis for High-Purity 3-Aryl-2H-Indazole Pharmaceutical Intermediates
Scalable Visible Light Photocatalysis for High-Purity 3-Aryl-2H-Indazole Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways to construct complex heterocyclic scaffolds that serve as the backbone of modern therapeutics. A significant breakthrough in this domain is detailed in Chinese Patent CN114805209A, which discloses a novel preparation method for visible light-induced 3-aryl-2H-indazole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed cross-coupling reactions to a greener, organic photocatalytic approach. By leveraging the unique reactivity of aryl sulfonium salts under blue light irradiation, this method enables the direct functionalization of the C3 position on the indazole ring with exceptional precision. For R&D directors and process chemists, this offers a compelling alternative that bypasses the limitations of noble metal catalysts while maintaining high purity standards essential for drug development.
Furthermore, the implications of this technology extend beyond mere academic interest; it addresses critical supply chain vulnerabilities associated with precious metal availability and pricing volatility. The use of 2,4,5,6-tetrakis(9-carbazolyl)-isophthalonitrile (4CzIPN) as a photocatalyst ensures that the process remains entirely metal-free, thereby simplifying the downstream purification processes and reducing the environmental footprint of the manufacturing operation. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative synthetic methodologies is crucial for maintaining competitiveness in the global market. The following analysis delves into the technical nuances and commercial viability of this visible light-induced transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the C3 position on 2H-indazoles has relied heavily on classical strategies involving transition metal-catalyzed cross-coupling reactions. These conventional methods typically necessitate the use of expensive noble metal catalysts such as palladium or copper, along with specialized ligands and additives to facilitate the reaction. Beyond the direct cost of these reagents, the presence of residual heavy metals in the final product poses a significant regulatory hurdle, requiring rigorous and costly purification steps to meet stringent pharmacopeial limits. Additionally, previous photocatalytic attempts often utilized aryl diazonium salts as radical precursors. While effective, aryl diazonium salts are notoriously unstable and carry a high risk of explosion, making them unsuitable for large-scale commercial production due to severe safety concerns.
The Novel Approach
In stark contrast, the methodology described in patent CN114805209A introduces a robust and safe alternative by employing aryl sulfonium salts as the aryl radical precursor. This innovation effectively mitigates the explosion risks associated with diazonium salts while expanding the substrate scope to include complex drug molecules. The reaction proceeds under mild conditions, specifically at room temperature under blue LED irradiation, which significantly reduces energy consumption compared to thermal methods requiring high heat. 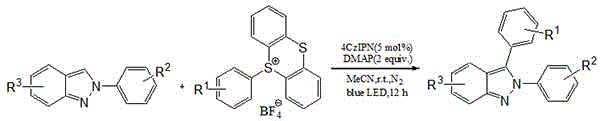 As illustrated in the reaction scheme, the process utilizes 4CzIPN as an organic photocatalyst and DMAP as an additive in acetonitrile solvent. This metal-free system not only lowers the raw material costs but also streamlines the workup procedure, as there is no need for extensive metal scavenging. The result is a cleaner reaction profile with yields consistently reported between 71% and 85%, demonstrating both high efficiency and operational simplicity suitable for industrial scale-up.
As illustrated in the reaction scheme, the process utilizes 4CzIPN as an organic photocatalyst and DMAP as an additive in acetonitrile solvent. This metal-free system not only lowers the raw material costs but also streamlines the workup procedure, as there is no need for extensive metal scavenging. The result is a cleaner reaction profile with yields consistently reported between 71% and 85%, demonstrating both high efficiency and operational simplicity suitable for industrial scale-up.
Mechanistic Insights into 4CzIPN-Catalyzed Radical Arylation
The core of this technological advancement lies in the photoredox catalytic cycle mediated by 4CzIPN. Upon absorption of blue light photons, the 4CzIPN catalyst enters an excited state capable of engaging in single-electron transfer (SET) processes. In this specific transformation, the excited photocatalyst interacts with the aryl sulfonium salt, facilitating the homolytic cleavage of the carbon-sulfur bond to generate a highly reactive aryl radical. This radical species then selectively attacks the electron-rich C3 position of the 2-aryl-2H-indazole substrate. The subsequent oxidation and deprotonation steps restore the aromaticity of the indazole system, yielding the desired 3-aryl substituted product. This mechanism avoids the formation of organometallic intermediates, which are often sensitive to moisture and oxygen, thus enhancing the robustness of the reaction.
From an impurity control perspective, this radical pathway offers distinct advantages over ionic mechanisms. The mild reaction conditions minimize side reactions such as polymerization or over-arylation, which are common pitfalls in harsher thermal environments. Furthermore, the use of DMAP as a base helps to neutralize any acidic byproducts generated during the reaction, ensuring a stable pH environment that protects the integrity of sensitive functional groups on the substrate. For quality control teams, this translates to a simpler impurity profile, making it easier to isolate the target compound with high purity through standard silica gel column chromatography using petroleum ether and ethyl acetate. The ability to tolerate a wide range of substituents, including esters, cyano groups, and halogens, further underscores the versatility of this mechanistic approach.
How to Synthesize 3-Aryl-2H-Indazole Efficiently
The practical implementation of this synthesis is straightforward and aligns well with standard laboratory and pilot plant operations. The protocol involves dissolving the 2-aryl-2H-indazole and the aryl sulfonium salt in acetonitrile, followed by the addition of the photocatalyst and base. The mixture is then subjected to blue light irradiation under a nitrogen atmosphere to prevent oxygen quenching of the excited catalyst. Detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the technical guide below to ensure reproducibility and optimal yield.
- Dissolve 2-aryl-2H-indazole and aryl sulfonium salt in acetonitrile solvent within a reaction vessel.
- Add the organic photocatalyst 4CzIPN (5 mol%) and DMAP additive to the reaction mixture under a nitrogen atmosphere.
- Irradiate the mixture with blue LEDs at room temperature for 12 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light-induced synthesis offers tangible strategic benefits that directly impact the bottom line and operational resilience. The elimination of noble metal catalysts removes a major cost driver and supply bottleneck, as the prices of palladium and rhodium are subject to extreme market fluctuations. Moreover, the reliance on organic photocatalysts like 4CzIPN, which can be synthesized from abundant starting materials, ensures a more stable and predictable supply chain. The mild reaction conditions also allow for the use of standard glass-lined reactors equipped with LED arrays, avoiding the need for specialized high-pressure or high-temperature equipment, thereby reducing capital expenditure.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost structure of producing 3-aryl-2H-indazole derivatives. By removing the requirement for expensive transition metals and their associated ligands, the direct material costs are significantly lowered. Furthermore, the absence of heavy metals simplifies the purification workflow, eliminating the need for costly metal scavenger resins and reducing solvent consumption during the cleaning process. This streamlined downstream processing leads to substantial overall cost savings per kilogram of the final product, enhancing profit margins for high-volume manufacturing.
- Enhanced Supply Chain Reliability: Utilizing aryl sulfonium salts as stable radical precursors mitigates the logistical challenges associated with hazardous reagents. Unlike explosive diazonium salts that require special handling and storage protocols, sulfonium salts are shelf-stable and can be sourced or synthesized reliably. This stability ensures continuous production schedules without the risk of shutdowns due to reagent degradation or safety incidents. Additionally, the broad substrate scope allows for the flexible sourcing of various aryl building blocks, providing procurement teams with multiple options to secure raw materials even during market disruptions.
- Scalability and Environmental Compliance: The scalability of photochemical reactions has historically been a concern, but advancements in LED technology and flow chemistry have made large-scale photoredox catalysis commercially viable. The low energy input required for blue LED irradiation compared to thermal heating contributes to a lower carbon footprint, aligning with corporate sustainability goals. Moreover, the use of acetonitrile, a common and recyclable solvent, combined with the generation of minimal hazardous waste, facilitates compliance with increasingly stringent environmental regulations. This green chemistry profile enhances the company's reputation and reduces the regulatory burden associated with waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the visible light-induced synthesis of 3-aryl-2H-indazoles. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the feasibility and advantages of this method for potential partners and stakeholders.
Q: What are the safety advantages of using aryl sulfonium salts over diazonium salts?
A: Unlike aryl diazonium salts which pose significant explosion risks during storage and handling, aryl sulfonium salts are thermally stable and safe to handle on a commercial scale, drastically reducing operational hazards in pharmaceutical manufacturing.
Q: Does this photocatalytic method require expensive transition metal catalysts?
A: No, this method utilizes 4CzIPN, an organic metal-free photocatalyst. This eliminates the need for costly palladium or copper catalysts and removes the regulatory burden of heavy metal residue testing in the final API.
Q: What is the typical yield range for this visible light induced synthesis?
A: According to the patent data, the reaction consistently achieves high yields ranging from 71% to 85% across various substrates, demonstrating robust efficiency suitable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aryl-2H-Indazole Supplier
The technological potential of visible light-induced C-H functionalization is immense, offering a pathway to complex molecular architectures that were previously difficult or expensive to access. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical clients. We are committed to delivering high-quality intermediates that adhere to the highest standards of safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Together, we can accelerate the delivery of life-saving medicines to the market through efficient and sustainable manufacturing solutions.
