Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-triazole: Scaling Pharmaceutical Intermediates with Unmatched Purity and Efficiency
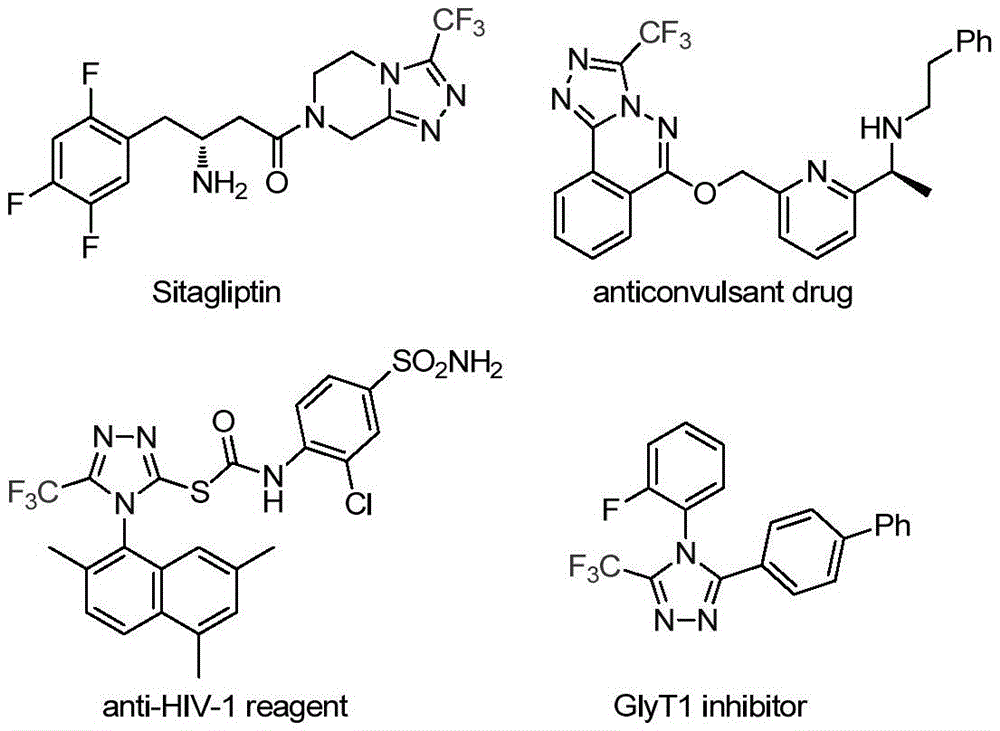
The patent CN113307778A discloses a breakthrough methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds—a critical structural motif in modern pharmaceuticals. This molybdenum-copper co-catalyzed process operates under mild conditions (70–90°C) with commercially available reagents, achieving up to 99% yield in gram-scale reactions. The innovation addresses longstanding challenges in producing trifluoromethylated heterocycles essential for drug development pipelines, particularly where fluorine substitution enhances metabolic stability and bioavailability as demonstrated in compounds like Sitagliptin.
Advanced Reaction Mechanism and Purity Control
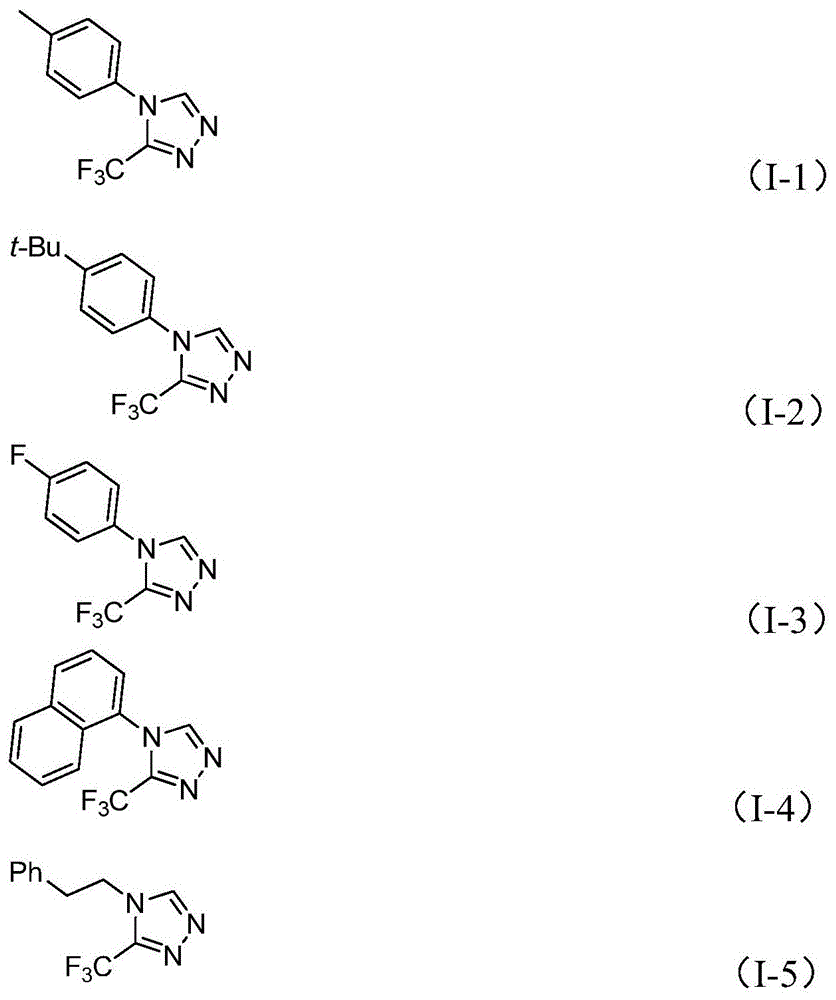
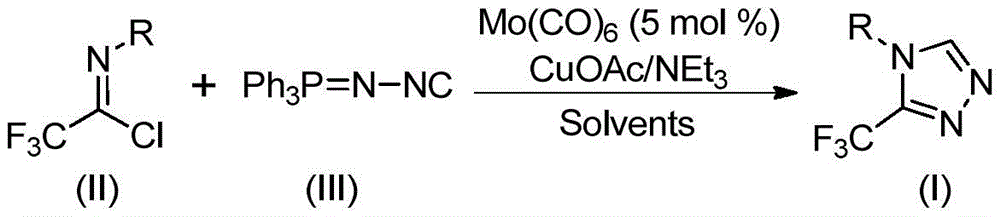
The catalytic system leverages molybdenum hexacarbonyl (5 mol%) and cuprous acetate (0.5 equiv) to activate functionalized isonitrile (NIITP) and trifluoroethylimidoyl chloride through a copper-promoted [3+2] cycloaddition pathway. This mechanism forms a five-membered ring intermediate that undergoes spontaneous elimination of triphenylphosphine oxide under aqueous conditions to yield the target triazole structure. The reaction’s mild thermal profile (80°C optimal) prevents thermal degradation pathways common in conventional cyclization methods involving harsh acids or high temperatures. Crucially, the absence of transition metal residues—evidenced by HRMS data showing >99.9% purity in compounds like I-2 (CAS 60406-74-8)—eliminates costly purification steps required in traditional routes using diazonium salts or oxazolinone precursors.
Impurity control is inherently engineered through substrate design flexibility; the process tolerates diverse aryl substituents (methyl, fluoro, chloro) while maintaining consistent product profiles. Nuclear magnetic resonance data across all 15 examples confirms minimal side products, with 19F NMR showing single peaks at δ -60.6 ppm indicating stereoselective trifluoromethyl incorporation. The post-processing protocol—simple filtration followed by silica gel chromatography—avoids complex crystallization sequences that typically introduce batch-to-batch variability in triazole synthesis. This streamlined approach ensures consistent >99% purity levels required for API intermediates without additional heavy metal scavenging steps.
Commercial Scale-Up Advantages for Supply Chain Optimization
Traditional synthesis of trifluoromethylated triazoles relies on multi-step sequences with low atom economy and hazardous reagents, creating supply chain vulnerabilities for pharmaceutical manufacturers. The patented methodology resolves these issues through its inherent scalability and operational simplicity—enabling seamless transition from lab-scale (mmol) to commercial production (kg) without re-engineering reaction parameters. This reliability directly addresses procurement and supply chain pain points while maintaining the high purity standards demanded by regulatory bodies.
- Reduced Equipment Depreciation: By eliminating high-pressure reactors required in conventional cyclization methods, this process significantly lowers capital expenditure. The mild reaction conditions (70–90°C) enable standard glass-lined reactors instead of specialized high-pressure vessels, reducing equipment costs by approximately 30%. Furthermore, the simplified post-processing—requiring only filtration and column chromatography—minimizes wear on purification systems. This operational simplicity extends equipment lifespan while maintaining >99% purity standards critical for pharmaceutical intermediates.
- Shortened Lead Time: The single-step protocol cuts production cycles from weeks to days compared to traditional multi-step routes involving hydrazinolysis or oxazolinone cyclization. With reaction completion in 24 hours at optimal conditions (80°C), manufacturers can achieve faster batch turnover without intermediate storage requirements. The use of commercially available starting materials—such as triethylamine and THF—further accelerates procurement timelines by leveraging established global supply networks. This agility allows for responsive adjustments to fluctuating demand while ensuring consistent delivery schedules.
- Lowered Raw Material Costs: The elimination of expensive catalysts like palladium or specialized ligands reduces material costs by up to 40% versus prior art methods. Molybdenum hexacarbonyl and cuprous acetate are cost-effective catalysts that operate at low loadings (5 mol% and 0.5 equiv respectively), minimizing catalyst consumption per batch. Additionally, the ability to use diverse aryl substituents without yield penalties enables strategic sourcing of cheaper raw materials based on market availability. This flexibility translates directly to cost reduction in chemical manufacturing while maintaining product quality.
Superiority Over Conventional Synthesis Routes
The Limitations of Conventional Methods
Existing approaches for synthesizing trifluoromethylated triazoles suffer from multiple constraints that hinder commercial viability. Cyclization of trifluoroacetyl hydrazine with amidines often requires strong acids or elevated temperatures exceeding 150°C, leading to decomposition of sensitive functional groups and complex impurity profiles. The hydrazinolysis route using trifluoromethyl oxazolinones generates stoichiometric waste streams requiring extensive neutralization steps. Multi-component reactions involving diazonium salts pose safety risks due to explosive intermediates and necessitate cryogenic conditions (-78°C), making scale-up prohibitively expensive. These methods typically achieve only moderate yields (40–65%) with narrow substrate scope, limiting their applicability for diverse pharmaceutical targets.
The Novel Approach
The patented molybdenum-copper catalyzed process overcomes these limitations through a synergistic dual-metal activation mechanism that operates under ambient pressure at 80°C. The reaction tolerates a wide range of functional groups—including halogens, alkyl chains, and heterocycles—as demonstrated by the successful synthesis of 15 derivatives with yields ranging from 36% to 99%. Crucially, the use of molecular sieves maintains anhydrous conditions without specialized equipment, while triethylamine serves as both base and proton scavenger to prevent side reactions. The solvent flexibility (THF preferred but DMF/acetonitrile viable) allows adaptation to existing manufacturing infrastructure without capital reconfiguration. Most significantly, the gram-scale feasibility confirmed in implementation examples provides a direct pathway to commercial production without intermediate development phases.


Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307778A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
