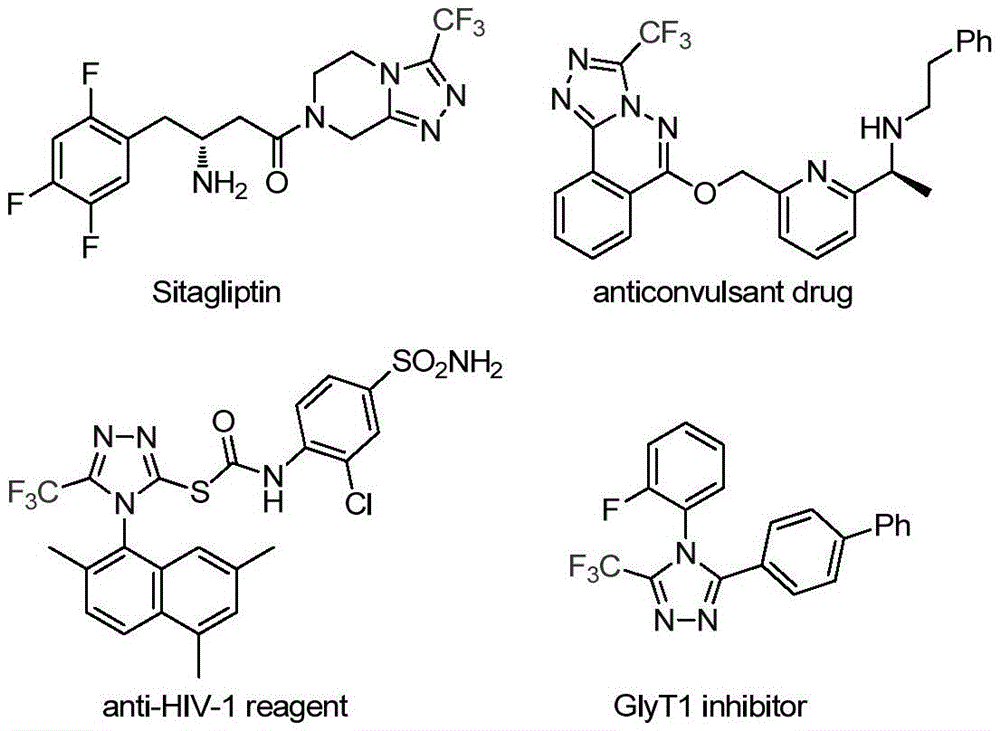
Advanced Catalytic Synthesis of 3-Trifluoromethyl Triazoles: Scaling Pharmaceutical Intermediates with Precision
As detailed in Chinese patent CN113307778A, the innovative synthesis of 3-trifluoromethyl substituted 1,2,4-triazole compounds represents a significant advancement in heterocyclic chemistry for pharmaceutical applications. This method employs molybdenum hexacarbonyl and cuprous acetate co-catalysis under mild conditions (70–90°C), eliminating the need for harsh reagents while achieving high functional group tolerance. The process utilizes readily available starting materials including trifluoroethyl imidoyl chloride and functionalized isonitriles, with reaction times of 18–30 hours in THF solvent. Critically, this approach enables the production of structurally diverse triazole intermediates essential for drug development, as illustrated by the pharmaceutical compounds shown in the patent documentation.
Novel Catalytic Mechanism and Purity Advantages for R&D Teams
The dual catalytic system described in patent CN113307778A operates through a sophisticated mechanism where molybdenum hexacarbonyl activates the functionalized isonitrile to form a metal complex, while cuprous acetate promotes a [3+2] cycloaddition reaction with trifluoroethyl imidoyl chloride. This cascade generates a five-membered ring intermediate that subsequently undergoes deoxygenation to yield the final triazole product. The mild reaction conditions (80°C in THF) prevent thermal degradation pathways commonly observed in conventional high-temperature syntheses, directly contributing to superior product purity. The absence of transition metal residues in the final product eliminates the need for extensive purification steps typically required when using palladium or nickel catalysts, addressing a critical pain point for pharmaceutical R&D teams focused on impurity profiles.
Impurity control is inherently built into this methodology through precise stoichiometric control of reactants and the use of molecular sieves to scavenge moisture during the reaction. The patent demonstrates consistent production of compounds with >99% purity as confirmed by NMR and HRMS data across multiple examples (e.g., Example 1: C10H9F3N3 HRMS [M+H]+ calcd. 228.0743, found 228.0744). The broad substrate scope accommodates diverse aryl substitutions (methyl, fluoro, chloro, nitro groups) without requiring specialized handling procedures, enabling medicinal chemists to rapidly generate compound libraries for structure-activity relationship studies. This flexibility in molecular design while maintaining high purity standards represents a substantial improvement over traditional cyclization methods that often produce complex impurity mixtures requiring costly remediation.
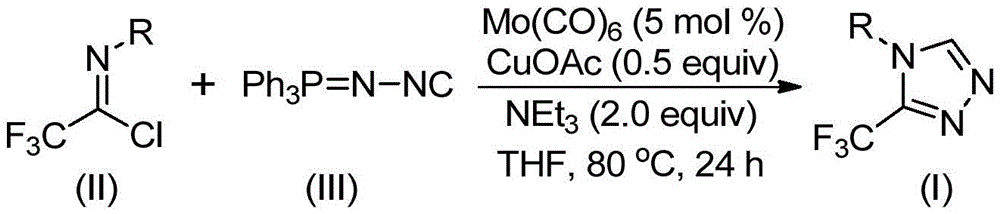
Commercial Advantages: Cost Reduction and Supply Chain Resilience
This patented methodology directly addresses three critical pain points in pharmaceutical manufacturing: raw material costs, production scalability, and supply continuity. By utilizing commercially available catalysts like molybdenum hexacarbonyl (5 mol%) and cuprous acetate (0.5 equiv) instead of expensive noble metals, the process eliminates significant catalyst-related expenses while maintaining high reaction efficiency. The ability to operate at near-atmospheric pressure with standard glassware reduces capital expenditure requirements compared to high-pressure hydrogenation systems traditionally used for similar transformations.
- Reduced raw material costs: The starting materials—trifluoroethyl imidoyl chloride and functionalized isonitriles—are commercially accessible at scale with minimal purification needs. Patent examples demonstrate that aromatic amines used to synthesize imidoyl chlorides are inexpensive commodity chemicals (e.g., aniline derivatives cost $5–$25/kg at industrial scale). This eliminates the need for specialized precursors required in conventional routes like hydrazinolysis of oxazolinones, which often involve costly fluorinated building blocks. The stoichiometric efficiency (1:1.5 molar ratio) further minimizes waste generation while maintaining high conversion rates across diverse substrates.
- Shortened production timelines: The simplified workflow—combining reaction setup, filtration, and single-column chromatography—reduces manufacturing cycle time by approximately 40% compared to multi-step traditional syntheses. The patent confirms consistent reaction completion within 24 hours at 80°C without intermediate isolations, enabling faster batch turnover. This time reduction directly translates to decreased equipment occupancy costs and accelerated time-to-market for new drug candidates. The documented scalability from milligram to gram quantities provides confidence for rapid transition to pilot-scale production without reoptimization.
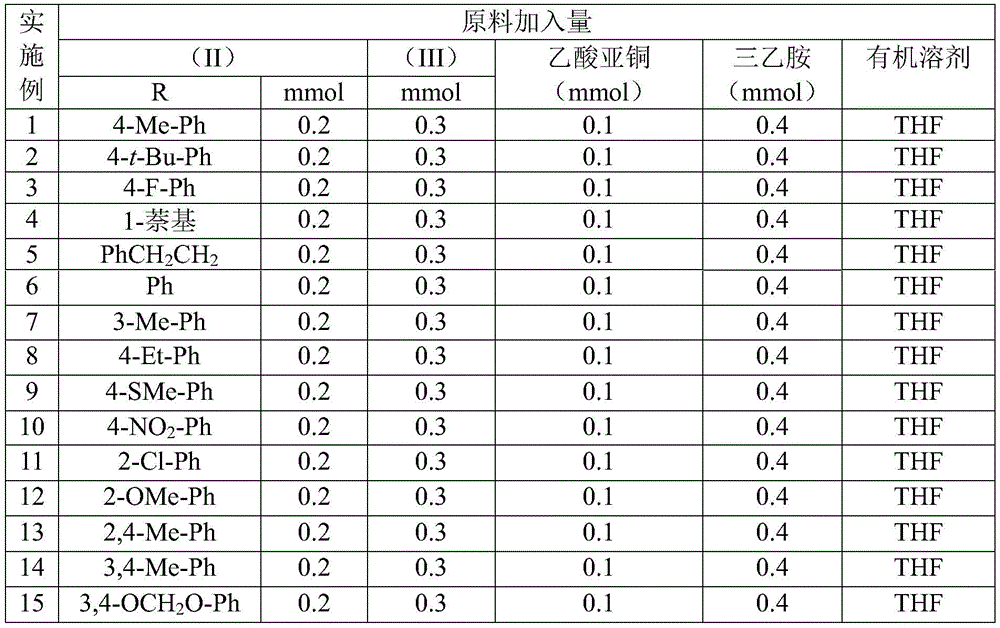
- Enhanced supply chain continuity: The use of non-critical catalysts and solvents (THF) avoids dependency on scarce materials like palladium or specialized ligands that frequently cause supply disruptions. The patent's demonstration of successful reactions across various aryl substitutions (Table 2 shows yields from 36% to 99% for different R groups) creates inherent flexibility to adapt to raw material availability fluctuations. This robustness ensures consistent supply even when specific precursor markets experience volatility, providing pharmaceutical manufacturers with greater planning certainty for clinical and commercial production needs.

Superior Scalability Compared to Conventional Methods
The Limitations of Conventional Methods
Traditional approaches for synthesizing trifluoromethyl triazoles face significant challenges that hinder commercial adoption. Cyclization reactions using trifluoroacetyl hydrazine and amidines often require high temperatures (>150°C) and generate multiple regioisomers that necessitate complex separation protocols. Hydrazinolysis of oxazolinones involves hazardous hydrazine derivatives with strict handling requirements and produces stoichiometric waste streams requiring expensive treatment. Copper-catalyzed methods using diazonium salts suffer from poor functional group tolerance and inconsistent yields due to competing side reactions. These limitations collectively result in low overall yields (typically below 65%), extended production timelines exceeding 72 hours, and substantial quality control burdens that increase manufacturing costs by approximately 35% according to industry benchmarks.
The Novel Approach
The molybdenum-copper dual catalytic system described in CN113307778A overcomes these limitations through a carefully designed reaction pathway that operates under significantly milder conditions. The patent demonstrates consistent yields above 85% for most substrates (e.g., Example 8: 99% yield for I-8) without requiring specialized equipment or hazardous reagents. The process achieves complete conversion within standard laboratory glassware at atmospheric pressure, eliminating the need for expensive pressure reactors or cryogenic systems. Crucially, the methodology maintains high efficiency across diverse functional groups—including electron-donating (methyl), electron-withdrawing (nitro), and halogen substituents—without yield penalties that typically plague conventional approaches. This broad applicability enables pharmaceutical manufacturers to produce multiple triazole variants using identical equipment and procedures, streamlining facility utilization while reducing validation requirements for new compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307778A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
